| Record Information |
|---|
| Version | 1.0 |
|---|
| Created at | 2020-04-17 18:49:45 UTC |
|---|
| Updated at | 2020-12-07 19:11:15 UTC |
|---|
| CannabisDB ID | CDB004912 |
|---|
| Secondary Accession Numbers | Not Available |
|---|
| Cannabis Compound Identification |
|---|
| Common Name | Niacinamide |
|---|
| Description | Niacinamide, also known as nicotinamid or nicovit, belongs to the class of organic compounds known as nicotinamides. These are heterocyclic aromatic compounds containing a pyridine ring substituted at position 3 by a carboxamide group. Niacinamide is a strong basic compound (based on its pKa). Niacinamide exists in all living species, ranging from bacteria to humans. niacinamide and phosphoribosyl pyrophosphate can be converted into nicotinic acid mononucleotide and phosphate through the action of the enzyme nicotinamide phosphoribosyltransferase. In humans, niacinamide is involved in the metabolic disorder called the nad+ signalling pathway (cancer). Niacinamide is an odorless tasting compound. Outside of the human body, Niacinamide is found, on average, in the highest concentration within a few different foods, such as common sages, milk (cow), and cocoa beans and in a lower concentration in common pea and beers. Niacinamide has also been detected, but not quantified in, several different foods, such as pineappple sages, boysenberries, cashew nuts, macadamia nut (m. tetraphylla), and sweet marjorams. This could make niacinamide a potential biomarker for the consumption of these foods. Niacinamide is a potentially toxic compound. A pyridinecarboxamide that is pyridine in which the hydrogen at position 3 is replaced by a carboxamide group. Niacinamide is expected to be in Cannabis as all living plants are known to produce and metabolize it. |
|---|
| Structure | |
|---|
| Synonyms | | Value | Source |
|---|
| 3-Pyridinecarboxamide | ChEBI | | beta-Pyridinecarboxamide | ChEBI | | Niacin | ChEBI | | Nicotinamid | ChEBI | | Nicotinic acid amide | ChEBI | | Nicotinsaeureamid | ChEBI | | Nikotinamid | ChEBI | | Nikotinsaeureamid | ChEBI | | Vitamin b3 | ChEBI | | Vitamin PP | ChEBI | | Nicotinamide | Kegg | | b-Pyridinecarboxamide | Generator | | Β-pyridinecarboxamide | Generator | | Nicotinate amide | Generator | | 3-Carbamoylpyridine | HMDB | | 3-Pyridinecarboxylic acid amide | HMDB | | Acid amide | HMDB | | Amid kyseliny nikotinove | HMDB | | Amide PP | HMDB | | Aminicotin | HMDB | | Amixicotyn | HMDB | | Amnicotin | HMDB | | Austrovit PP | HMDB | | Benicot | HMDB | | Delonin amide | HMDB | | Dipegyl | HMDB | | Dipigyl | HMDB | | Endobion | HMDB | | Factor PP | HMDB | | Hansamid | HMDB | | Inovitan PP | HMDB | | m-(Aminocarbonyl)pyridine | HMDB | | Mediatric | HMDB | | NAM | HMDB | | Nandervit-N | HMDB | | Niacevit | HMDB | | Niamide | HMDB | | Niavit PP | HMDB | | Nicamide | HMDB | | Nicamina | HMDB | | Nicamindon | HMDB | | Nicasir | HMDB | | Nicobion | HMDB | | Nicofort | HMDB | | Nicogen | HMDB | | Nicomidol | HMDB | | Nicosan 2 | HMDB | | Nicosylamide | HMDB | | Nicota | HMDB | | Nicotamide | HMDB | | Nicotilamide | HMDB | | Nicotililamido | HMDB | | Nicotinamida | HMDB | | Nicotinamidum | HMDB | | Nicotine acid amide | HMDB | | Nicotine amide | HMDB | | Nicotinic amide | HMDB | | Nicotinsaureamid | HMDB | | Nicotol | HMDB | | Nicotylamide | HMDB | | Nicotylamidum | HMDB | | Nicovel | HMDB | | Nicovit | HMDB | | Nicovitina | HMDB | | Nicovitol | HMDB | | Nicozymin | HMDB | | Nictoamide | HMDB | | Niko-tamin | HMDB | | Niocinamide | HMDB | | Niozymin | HMDB | | Papulex | HMDB | | Pelmin | HMDB | | Pelmine | HMDB | | Pelonin amide | HMDB | | PP-Faktor | HMDB | | Propamine a | HMDB | | Pyridine-3-carboxylic acid amide | HMDB | | Savacotyl | HMDB | | Vi-nicotyl | HMDB | | Vi-noctyl | HMDB | | Witamina PP | HMDB | | 3 Pyridinecarboxamide | HMDB | | Astra brand OF niacinamide | HMDB | | b 3, Vitamin | HMDB | | Niacinamide merck brand | HMDB | | Pharmagenix brand OF niacinamide | HMDB | | b3, Vitamin | HMDB | | Enduramide | HMDB | | Jenapharm, nicotinsäureamid | HMDB | | Niacinamide jenapharm brand | HMDB | | Niacinamide pharmagenix brand | HMDB | | Jenapharm brand OF niacinamide | HMDB | | Nicotinsäureamid jenapharm | HMDB | | Vitamin b 3 | HMDB | | Merck brand OF niacinamide | HMDB | | Niacinamide astra brand | HMDB | | Niacinamide | ChEBI |
|
|---|
| Chemical Formula | C6H6N2O |
|---|
| Average Molecular Weight | 122.12 |
|---|
| Monoisotopic Molecular Weight | 122.048 |
|---|
| IUPAC Name | pyridine-3-carboxamide |
|---|
| Traditional Name | nicotinamide |
|---|
| CAS Registry Number | 98-92-0 |
|---|
| SMILES | NC(=O)C1=CC=CN=C1 |
|---|
| InChI Identifier | InChI=1S/C6H6N2O/c7-6(9)5-2-1-3-8-4-5/h1-4H,(H2,7,9) |
|---|
| InChI Key | DFPAKSUCGFBDDF-UHFFFAOYSA-N |
|---|
| Chemical Taxonomy |
|---|
| Description | Belongs to the class of organic compounds known as nicotinamides. These are heterocyclic aromatic compounds containing a pyridine ring substituted at position 3 by a carboxamide group. |
|---|
| Kingdom | Organic compounds |
|---|
| Super Class | Organoheterocyclic compounds |
|---|
| Class | Pyridines and derivatives |
|---|
| Sub Class | Pyridinecarboxylic acids and derivatives |
|---|
| Direct Parent | Nicotinamides |
|---|
| Alternative Parents | |
|---|
| Substituents | - Nicotinamide
- Heteroaromatic compound
- Primary carboxylic acid amide
- Carboxamide group
- Azacycle
- Carboxylic acid derivative
- Organic nitrogen compound
- Organic oxygen compound
- Organopnictogen compound
- Organic oxide
- Hydrocarbon derivative
- Organooxygen compound
- Organonitrogen compound
- Aromatic heteromonocyclic compound
|
|---|
| Molecular Framework | Aromatic heteromonocyclic compounds |
|---|
| External Descriptors | |
|---|
| Ontology |
|---|
|
| Physiological effect | Health effect: |
|---|
| Disposition | Route of exposure: Source: Biological location: |
|---|
| Role | Biological role: Industrial application: |
|---|
| Physical Properties |
|---|
| State | Solid |
|---|
| Experimental Properties | | Property | Value | Reference |
|---|
| Melting Point | 130 °C | Not Available | | Boiling Point | Not Available | Not Available | | Water Solubility | 500 mg/mL at 25 °C | Not Available | | logP | -0.37 | HANSCH,C ET AL. (1995) |
|
|---|
| Predicted Properties | [] |
|---|
| Spectra |
|---|
| EI-MS/GC-MS | | Type | Description | Splash Key | View |
|---|
| EI-MS | Mass Spectrum (Electron Ionization) | splash10-0kor-9600000000-2b1c5667d92bc1b80b36 | 2014-09-20 | View Spectrum | | GC-MS | Niacinamide, 1 TMS, GC-MS Spectrum | splash10-004i-0900000000-acb6a21304b0c09c8472 | Spectrum | | GC-MS | Niacinamide, non-derivatized, GC-MS Spectrum | splash10-004r-0900000000-ce86dcff0153b66dd538 | Spectrum | | GC-MS | Niacinamide, 1 TMS, GC-MS Spectrum | splash10-004i-9500000000-790385e574240b8d39de | Spectrum | | GC-MS | Niacinamide, 2 TMS, GC-MS Spectrum | splash10-0udi-2910000000-d760b65bb76d6464038e | Spectrum | | GC-MS | Niacinamide, 1 TMS, GC-MS Spectrum | splash10-004i-4900000000-47c6f72d1bb5465c1376 | Spectrum | | GC-MS | Niacinamide, non-derivatized, GC-MS Spectrum | splash10-0kor-9800000000-1809e537780b814af0c9 | Spectrum | | GC-MS | Niacinamide, non-derivatized, GC-MS Spectrum | splash10-0kor-9800000000-c4a5ff5285417499eff8 | Spectrum | | GC-MS | Niacinamide, non-derivatized, GC-MS Spectrum | splash10-004i-0900000000-0e84a60cc2a2d44704f4 | Spectrum | | GC-MS | Niacinamide, non-derivatized, GC-MS Spectrum | splash10-004i-0900000000-acb6a21304b0c09c8472 | Spectrum | | GC-MS | Niacinamide, non-derivatized, GC-MS Spectrum | splash10-004r-0900000000-ce86dcff0153b66dd538 | Spectrum | | GC-MS | Niacinamide, non-derivatized, GC-MS Spectrum | splash10-004i-9500000000-790385e574240b8d39de | Spectrum | | GC-MS | Niacinamide, non-derivatized, GC-MS Spectrum | splash10-0udi-2910000000-d760b65bb76d6464038e | Spectrum | | GC-MS | Niacinamide, non-derivatized, GC-MS Spectrum | splash10-004i-4900000000-47c6f72d1bb5465c1376 | Spectrum | | GC-MS | Niacinamide, non-derivatized, GC-MS Spectrum | splash10-004i-0900000000-23a567506bfbc259e77c | Spectrum | | Predicted GC-MS | Niacinamide, non-derivatized, Predicted GC-MS Spectrum - 70eV, Positive | splash10-05fr-8900000000-931c139355be3ef594f2 | Spectrum | | Predicted GC-MS | Niacinamide, non-derivatized, Predicted GC-MS Spectrum - 70eV, Positive | Not Available | Spectrum | | Predicted GC-MS | Niacinamide, non-derivatized, Predicted GC-MS Spectrum - 70eV, Positive | Not Available | Spectrum |
|
|---|
| MS/MS | | Type | Description | Splash Key | View |
|---|
| MS/MS | LC-MS/MS Spectrum - Quattro_QQQ 10V, Positive (Annotated) | splash10-00di-2900000000-72a7c92eb2667f5b6c3f | 2012-07-24 | View Spectrum | | MS/MS | LC-MS/MS Spectrum - Quattro_QQQ 25V, Positive (Annotated) | splash10-001i-9000000000-6486d230a4a2fd4bab0e | 2012-07-24 | View Spectrum | | MS/MS | LC-MS/MS Spectrum - Quattro_QQQ 40V, Positive (Annotated) | splash10-001i-9000000000-6486d230a4a2fd4bab0e | 2012-07-24 | View Spectrum | | MS/MS | LC-MS/MS Spectrum - EI-B (HITACHI M-80) , Positive | splash10-0kor-9800000000-89d56b6cb7e033699d24 | 2012-08-31 | View Spectrum | | MS/MS | LC-MS/MS Spectrum - LC-ESI-QQ (API3000, Applied Biosystems) 10V, Positive | splash10-00di-0900000000-eed71abbb10ba926c13d | 2012-08-31 | View Spectrum | | MS/MS | LC-MS/MS Spectrum - LC-ESI-QQ (API3000, Applied Biosystems) 20V, Positive | splash10-00di-3900000000-0c62aa6aba6e23b6c195 | 2012-08-31 | View Spectrum | | MS/MS | LC-MS/MS Spectrum - LC-ESI-QQ (API3000, Applied Biosystems) 30V, Positive | splash10-001i-9100000000-4357a8833611d1756c59 | 2012-08-31 | View Spectrum | | MS/MS | LC-MS/MS Spectrum - LC-ESI-QQ (API3000, Applied Biosystems) 40V, Positive | splash10-001i-9000000000-c9685717817d0081fe6d | 2012-08-31 | View Spectrum | | MS/MS | LC-MS/MS Spectrum - LC-ESI-QQ (API3000, Applied Biosystems) 50V, Positive | splash10-0fc0-9000000000-ec0ccca306f38cf72a78 | 2012-08-31 | View Spectrum | | MS/MS | LC-MS/MS Spectrum - LC-ESI-QTOF (UPLC Q-Tof Premier, Waters) , Positive | splash10-00di-4900000000-f6d71a6c467f84ddd6ab | 2012-08-31 | View Spectrum | | MS/MS | LC-MS/MS Spectrum - LC-ESI-QTOF (UPLC Q-Tof Premier, Waters) 30V, Positive | splash10-001i-9100000000-62c4bd312efbfa37d1a1 | 2012-08-31 | View Spectrum | | MS/MS | LC-MS/MS Spectrum - LC-ESI-QTOF (UPLC Q-Tof Premier, Waters) , Positive | splash10-00di-4900000000-05714b528dc84942b9db | 2012-08-31 | View Spectrum | | MS/MS | LC-MS/MS Spectrum - LC-ESI-QTOF (UPLC Q-Tof Premier, Waters) 30V, Positive | splash10-001i-9100000000-edb6a77c0dfdcbdacc5d | 2012-08-31 | View Spectrum | | MS/MS | LC-MS/MS Spectrum - LC-ESI-QQ , positive | splash10-00di-0900000000-eed71abbb10ba926c13d | 2017-09-14 | View Spectrum | | MS/MS | LC-MS/MS Spectrum - LC-ESI-QQ , positive | splash10-00di-3900000000-0c62aa6aba6e23b6c195 | 2017-09-14 | View Spectrum | | MS/MS | LC-MS/MS Spectrum - LC-ESI-QQ , positive | splash10-001i-9100000000-4357a8833611d1756c59 | 2017-09-14 | View Spectrum | | MS/MS | LC-MS/MS Spectrum - LC-ESI-QQ , positive | splash10-001i-9000000000-c9685717817d0081fe6d | 2017-09-14 | View Spectrum | | MS/MS | LC-MS/MS Spectrum - LC-ESI-QQ , positive | splash10-0fc0-9000000000-ec0ccca306f38cf72a78 | 2017-09-14 | View Spectrum | | MS/MS | LC-MS/MS Spectrum - LC-ESI-ITFT , positive | splash10-00di-0900000000-960acce7ead55399b5db | 2017-09-14 | View Spectrum | | Predicted MS/MS | Predicted LC-MS/MS Spectrum - 10V, Positive | splash10-00di-0900000000-2c7ec333d35eb740520a | 2015-05-27 | View Spectrum | | Predicted MS/MS | Predicted LC-MS/MS Spectrum - 20V, Positive | splash10-0089-9600000000-466f98885e2efba73575 | 2015-05-27 | View Spectrum | | Predicted MS/MS | Predicted LC-MS/MS Spectrum - 40V, Positive | splash10-0ue9-9200000000-6ef8d7e5edcdd0b02758 | 2015-05-27 | View Spectrum | | Predicted MS/MS | Predicted LC-MS/MS Spectrum - 10V, Negative | splash10-00di-1900000000-f38177aa475e58941088 | 2015-05-27 | View Spectrum | | Predicted MS/MS | Predicted LC-MS/MS Spectrum - 20V, Negative | splash10-00fr-9800000000-6a883d82fcd7c450f6ce | 2015-05-27 | View Spectrum | | Predicted MS/MS | Predicted LC-MS/MS Spectrum - 40V, Negative | splash10-0006-9000000000-882d0c59d946a5087b9f | 2015-05-27 | View Spectrum |
|
|---|
| NMR | | Type | Description | | View |
|---|
| 1D NMR | 13C NMR Spectrum (1D, 125 MHz, H2O, experimental) | | Spectrum | | 1D NMR | 1H NMR Spectrum (1D, 600 MHz, H2O, experimental) | | Spectrum | | 1D NMR | 1H NMR Spectrum (1D, 400 MHz, DMSO-d6, experimental) | | Spectrum | | 1D NMR | 13C NMR Spectrum (1D, 25.16 MHz, D2O, experimental) | | Spectrum | | 1D NMR | 1H NMR Spectrum (1D, D2O, experimental) | | Spectrum | | 1D NMR | 13C NMR Spectrum (1D, D2O, experimental) | | Spectrum | | 1D NMR | 1H NMR Spectrum (1D, 100 MHz, D2O, predicted) | | Spectrum | | 1D NMR | 13C NMR Spectrum (1D, 100 MHz, D2O, predicted) | | Spectrum | | 1D NMR | 1H NMR Spectrum (1D, 1000 MHz, D2O, predicted) | | Spectrum | | 1D NMR | 13C NMR Spectrum (1D, 1000 MHz, D2O, predicted) | | Spectrum | | 1D NMR | 1H NMR Spectrum (1D, 200 MHz, D2O, predicted) | | Spectrum | | 1D NMR | 13C NMR Spectrum (1D, 200 MHz, D2O, predicted) | | Spectrum | | 1D NMR | 1H NMR Spectrum (1D, 300 MHz, D2O, predicted) | | Spectrum | | 1D NMR | 13C NMR Spectrum (1D, 300 MHz, D2O, predicted) | | Spectrum | | 1D NMR | 1H NMR Spectrum (1D, 400 MHz, D2O, predicted) | | Spectrum | | 1D NMR | 13C NMR Spectrum (1D, 400 MHz, D2O, predicted) | | Spectrum | | 1D NMR | 1H NMR Spectrum (1D, 500 MHz, D2O, predicted) | | Spectrum | | 1D NMR | 13C NMR Spectrum (1D, 500 MHz, D2O, predicted) | | Spectrum | | 1D NMR | 1H NMR Spectrum (1D, 600 MHz, D2O, predicted) | | Spectrum | | 1D NMR | 13C NMR Spectrum (1D, 600 MHz, D2O, predicted) | | Spectrum | | 1D NMR | 1H NMR Spectrum (1D, 700 MHz, D2O, predicted) | | Spectrum | | 1D NMR | 13C NMR Spectrum (1D, 700 MHz, D2O, predicted) | | Spectrum | | 1D NMR | 1H NMR Spectrum (1D, 800 MHz, D2O, predicted) | | Spectrum | | 1D NMR | 13C NMR Spectrum (1D, 800 MHz, D2O, predicted) | | Spectrum | | 1D NMR | 1H NMR Spectrum (1D, 900 MHz, D2O, predicted) | | Spectrum | | 2D NMR | [1H, 1H]-TOCSY. Unexported temporarily by An Chi on Oct 15, 2021 until json or nmrML file is generated. 2D NMR Spectrum (experimental) | | Spectrum | | 2D NMR | [1H, 13C]-HSQC NMR Spectrum (2D, 600 MHz, H2O, experimental) | | Spectrum |
|
|---|
| Pathways |
|---|
| Pathways | | Name | SMPDB/Pathwhiz | KEGG | | Nicotinate and Nicotinamide Metabolism | 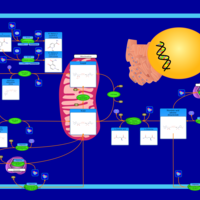 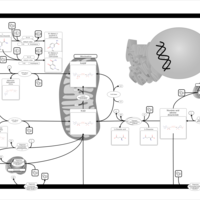  | 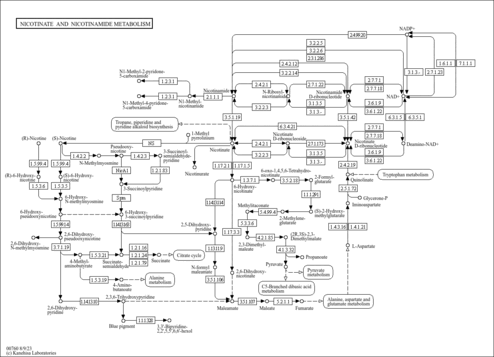 |
|
|---|
| Protein Targets |
|---|
| Enzymes | |
|---|
| Transporters | Not Available |
|---|
| Metal Bindings | |
|---|
| Receptors | |
|---|
| Transcriptional Factors | |
|---|
| Concentrations Data |
|---|
| Not Available |
|---|
| External Links |
|---|
| HMDB ID | HMDB0001406 |
|---|
| DrugBank ID | DB02701 |
|---|
| Phenol Explorer Compound ID | Not Available |
|---|
| FoodDB ID | FDB012485 |
|---|
| KNApSAcK ID | C00000209 |
|---|
| Chemspider ID | 911 |
|---|
| KEGG Compound ID | C00153 |
|---|
| BioCyc ID | NIACINAMIDE |
|---|
| BiGG ID | 34058 |
|---|
| Wikipedia Link | Nicotinamide |
|---|
| METLIN ID | 1497 |
|---|
| PubChem Compound | 936 |
|---|
| PDB ID | Not Available |
|---|
| ChEBI ID | 17154 |
|---|
| References |
|---|
| General References | Not Available |
|---|
