| Record Information |
|---|
| Version | 1.0 |
|---|
| Created at | 2020-04-17 18:41:03 UTC |
|---|
| Updated at | 2022-12-13 23:36:29 UTC |
|---|
| CannabisDB ID | CDB004829 |
|---|
| Secondary Accession Numbers | Not Available |
|---|
| Cannabis Compound Identification |
|---|
| Common Name | Pyruvic acid |
|---|
| Description | Pyruvic acid, also known as pyroracemic acid or 2-oxopropanoate, belongs to the class of organic compounds known as alpha-keto acids and derivatives. These are organic compounds containing an aldehyde substituted with a keto group on the adjacent carbon. Pyruvic acid is a drug which is used for nutritional supplementation, also for treating dietary shortage or imbalance. Pyruvic acid is a very hydrophobic molecule, practically insoluble (in water), and relatively neutral. Pyruvic acid exists in all living species, ranging from bacteria to humans. Outside of the human body, Pyruvic acid has been detected, but not quantified in, several different foods, such as ohelo berries, cottonseeds, chinese cinnamons, common sages, and vanilla. This could make pyruvic acid a potential biomarker for the consumption of these foods. A 2-oxo monocarboxylic acid that is the 2-keto derivative of propionic acid. Pyruvic acid is a potentially toxic compound. Pyruvic acid is expected to be in Cannabis as all living plants are known to produce and metabolize it. |
|---|
| Structure | |
|---|
| Synonyms | | Value | Source |
|---|
| 2-Ketopropionic acid | ChEBI | | 2-Oxopropanoic acid | ChEBI | | 2-Oxopropansaeure | ChEBI | | 2-Oxopropionsaeure | ChEBI | | Acetylformic acid | ChEBI | | Acide pyruvique | ChEBI | | alpha-Ketopropionic acid | ChEBI | | alpha-Oxopropionsaeure | ChEBI | | Brenztraubensaeure | ChEBI | | BTS | ChEBI | | CH3COCOOH | ChEBI | | Pyroracemic acid | ChEBI | | 2-Oxopropanoate | Kegg | | 2-Ketopropionate | Generator | | Acetylformate | Generator | | a-Ketopropionate | Generator | | a-Ketopropionic acid | Generator | | alpha-Ketopropionate | Generator | | Α-ketopropionate | Generator | | Α-ketopropionic acid | Generator | | a-Oxopropionsaeure | Generator | | Α-oxopropionsaeure | Generator | | Pyroracemate | Generator | | Pyruvate | Generator | | 2-Oxopropionate | HMDB | | 2-Oxopropionic acid | HMDB | | Acid, pyruvic | MeSH, HMDB | | Pyruvic acid | HMDB | | alpha-Ketopropanoic acid | HMDB | | α-Ketopropanoic acid | HMDB |
|
|---|
| Chemical Formula | C3H4O3 |
|---|
| Average Molecular Weight | 88.06 |
|---|
| Monoisotopic Molecular Weight | 88.016 |
|---|
| IUPAC Name | 2-oxopropanoic acid |
|---|
| Traditional Name | pyruvic acid |
|---|
| CAS Registry Number | 127-17-3 |
|---|
| SMILES | CC(=O)C(O)=O |
|---|
| InChI Identifier | InChI=1S/C3H4O3/c1-2(4)3(5)6/h1H3,(H,5,6) |
|---|
| InChI Key | LCTONWCANYUPML-UHFFFAOYSA-N |
|---|
| Chemical Taxonomy |
|---|
| Description | Belongs to the class of organic compounds known as alpha-keto acids and derivatives. These are organic compounds containing an aldehyde substituted with a keto group on the adjacent carbon. |
|---|
| Kingdom | Organic compounds |
|---|
| Super Class | Organic acids and derivatives |
|---|
| Class | Keto acids and derivatives |
|---|
| Sub Class | Alpha-keto acids and derivatives |
|---|
| Direct Parent | Alpha-keto acids and derivatives |
|---|
| Alternative Parents | |
|---|
| Substituents | - Alpha-keto acid
- Alpha-hydroxy ketone
- Ketone
- Monocarboxylic acid or derivatives
- Carboxylic acid
- Carboxylic acid derivative
- Organic oxygen compound
- Organic oxide
- Hydrocarbon derivative
- Organooxygen compound
- Carbonyl group
- Aliphatic acyclic compound
|
|---|
| Molecular Framework | Aliphatic acyclic compounds |
|---|
| External Descriptors | |
|---|
| Ontology |
|---|
|
| Physiological effect | Health effect: |
|---|
| Disposition | Route of exposure: Source: Biological location: |
|---|
| Role | Industrial application: Biological role: |
|---|
| Physical Properties |
|---|
| State | Liquid |
|---|
| Experimental Properties | | Property | Value | Reference |
|---|
| Melting Point | 13.8 °C | Not Available | | Boiling Point | 165 °C | Wikipedia | | Water Solubility | 1000 mg/mL | Not Available | | logP | Not Available | Not Available |
|
|---|
| Predicted Properties | [] |
|---|
| Spectra |
|---|
| EI-MS/GC-MS | | Type | Description | Splash Key | View |
|---|
| EI-MS | Mass Spectrum (Electron Ionization) | splash10-0006-9000000000-f315d0752893e7d0c657 | 2014-09-20 | View Spectrum | | GC-MS | Pyruvic acid, non-derivatized, GC-MS Spectrum | splash10-00dr-4900000000-f26ef76666e40ab9fe61 | Spectrum | | GC-MS | Pyruvic acid, 1 MEOX; 1 TMS, GC-MS Spectrum | splash10-00di-5900000000-b8e81f82572d4796e944 | Spectrum | | GC-MS | Pyruvic acid, 2 TMS, GC-MS Spectrum | splash10-014i-5970000000-154bf9ad168a12593fcc | Spectrum | | GC-MS | Pyruvic acid, non-derivatized, GC-MS Spectrum | splash10-0006-9000000000-a2cf85a5e1d2379d26df | Spectrum | | GC-MS | Pyruvic acid, non-derivatized, GC-MS Spectrum | splash10-00dr-4900000000-f26ef76666e40ab9fe61 | Spectrum | | GC-MS | Pyruvic acid, non-derivatized, GC-MS Spectrum | splash10-00di-5900000000-b8e81f82572d4796e944 | Spectrum | | GC-MS | Pyruvic acid, non-derivatized, GC-MS Spectrum | splash10-014i-5970000000-154bf9ad168a12593fcc | Spectrum | | GC-MS | Pyruvic acid, non-derivatized, GC-MS Spectrum | splash10-00dr-5900000000-5b1f470d4ff91420618c | Spectrum | | Predicted GC-MS | Pyruvic acid, non-derivatized, Predicted GC-MS Spectrum - 70eV, Positive | splash10-0006-9000000000-5417b44aa241a7ba27e8 | Spectrum | | Predicted GC-MS | Pyruvic acid, 1 TMS, Predicted GC-MS Spectrum - 70eV, Positive | splash10-00dm-9400000000-6db65a709bdc47e3adf7 | Spectrum | | Predicted GC-MS | Pyruvic acid, non-derivatized, Predicted GC-MS Spectrum - 70eV, Positive | Not Available | Spectrum | | Predicted GC-MS | Pyruvic acid, non-derivatized, Predicted GC-MS Spectrum - 70eV, Positive | Not Available | Spectrum | | Predicted GC-MS | Pyruvic acid, TMS_1_2, Predicted GC-MS Spectrum - 70eV, Positive | Not Available | Spectrum | | Predicted GC-MS | Pyruvic acid, TBDMS_1_1, Predicted GC-MS Spectrum - 70eV, Positive | Not Available | Spectrum | | Predicted GC-MS | Pyruvic acid, TBDMS_1_2, Predicted GC-MS Spectrum - 70eV, Positive | Not Available | Spectrum |
|
|---|
| MS/MS | | Type | Description | Splash Key | View |
|---|
| MS/MS | LC-MS/MS Spectrum - EI-B (HITACHI M-80B) , Positive | splash10-0006-9000000000-a2cf85a5e1d2379d26df | 2012-08-31 | View Spectrum | | MS/MS | LC-MS/MS Spectrum - LC-ESI-QQ (API3000, Applied Biosystems) 10V, Negative | splash10-000i-9000000000-dd49835da8355fb6e625 | 2012-08-31 | View Spectrum | | MS/MS | LC-MS/MS Spectrum - LC-ESI-QQ (API3000, Applied Biosystems) 20V, Negative | splash10-000i-9000000000-f09d8e3d7a774b255d89 | 2012-08-31 | View Spectrum | | MS/MS | LC-MS/MS Spectrum - LC-ESI-QQ (API3000, Applied Biosystems) 30V, Negative | splash10-0006-9000000000-7d91f6f626cab1a366fd | 2012-08-31 | View Spectrum | | MS/MS | LC-MS/MS Spectrum - LC-ESI-QQ (API3000, Applied Biosystems) 40V, Negative | splash10-0006-9000000000-8ae98cdb3e142034e52a | 2012-08-31 | View Spectrum | | MS/MS | LC-MS/MS Spectrum - LC-ESI-QQ (API3000, Applied Biosystems) 50V, Negative | splash10-0006-9000000000-e04e6c68013983e1b6dc | 2012-08-31 | View Spectrum | | MS/MS | LC-MS/MS Spectrum - LC-ESI-QQ , negative | splash10-000i-9000000000-dd49835da8355fb6e625 | 2017-09-14 | View Spectrum | | MS/MS | LC-MS/MS Spectrum - LC-ESI-QQ , negative | splash10-000i-9000000000-f09d8e3d7a774b255d89 | 2017-09-14 | View Spectrum | | MS/MS | LC-MS/MS Spectrum - LC-ESI-QQ , negative | splash10-0006-9000000000-7d91f6f626cab1a366fd | 2017-09-14 | View Spectrum | | MS/MS | LC-MS/MS Spectrum - LC-ESI-QQ , negative | splash10-0006-9000000000-8ae98cdb3e142034e52a | 2017-09-14 | View Spectrum | | MS/MS | LC-MS/MS Spectrum - LC-ESI-QQ , negative | splash10-0006-9000000000-e04e6c68013983e1b6dc | 2017-09-14 | View Spectrum | | MS/MS | LC-MS/MS Spectrum - 10V, Negative | splash10-000f-9000000000-f24c93ecfd3928827154 | 2021-09-20 | View Spectrum | | MS/MS | LC-MS/MS Spectrum - 40V, Negative | splash10-0udj-9000000000-fc3b9ad0c57f44261fba | 2021-09-20 | View Spectrum | | MS/MS | LC-MS/MS Spectrum - 20V, Negative | splash10-014i-9000000000-f3444f8b94ee5a0a9f74 | 2021-09-20 | View Spectrum | | MS/MS | LC-MS/MS Spectrum - 33V, Negative | splash10-0avr-9000000000-dc40a6a1b9b166d6e68a | 2021-09-20 | View Spectrum | | MS/MS | LC-MS/MS Spectrum - 33V, Negative | splash10-016r-9000000000-efac7b176bb77118ecb8 | 2021-09-20 | View Spectrum | | Predicted MS/MS | Predicted LC-MS/MS Spectrum - 10V, Positive | splash10-000i-9000000000-d0defa72b09503c6d6d1 | 2016-09-12 | View Spectrum | | Predicted MS/MS | Predicted LC-MS/MS Spectrum - 20V, Positive | splash10-000f-9000000000-c25fa150e9c490319a2a | 2016-09-12 | View Spectrum | | Predicted MS/MS | Predicted LC-MS/MS Spectrum - 40V, Positive | splash10-0006-9000000000-ccb42b4c05ddd001990f | 2016-09-12 | View Spectrum | | Predicted MS/MS | Predicted LC-MS/MS Spectrum - 10V, Negative | splash10-000i-9000000000-faf36ff70d6205370270 | 2016-09-12 | View Spectrum | | Predicted MS/MS | Predicted LC-MS/MS Spectrum - 20V, Negative | splash10-000i-9000000000-60c1a02aabf80f51050f | 2016-09-12 | View Spectrum | | Predicted MS/MS | Predicted LC-MS/MS Spectrum - 40V, Negative | splash10-000f-9000000000-ca5f4a2f06787d8b62a0 | 2016-09-12 | View Spectrum | | Predicted MS/MS | Predicted LC-MS/MS Spectrum - 10V, Positive | splash10-0006-9000000000-0eb1fb2cdd24bdc78601 | 2021-09-22 | View Spectrum | | Predicted MS/MS | Predicted LC-MS/MS Spectrum - 20V, Positive | splash10-0006-9000000000-87bbaed151efac084591 | 2021-09-22 | View Spectrum | | Predicted MS/MS | Predicted LC-MS/MS Spectrum - 40V, Positive | splash10-0006-9000000000-87bbaed151efac084591 | 2021-09-22 | View Spectrum |
|
|---|
| NMR | | Type | Description | | View |
|---|
| 1D NMR | 13C NMR Spectrum (1D, 125 MHz, H2O, experimental) | | Spectrum | | 1D NMR | 1H NMR Spectrum (1D, 500 MHz, H2O, experimental) | | Spectrum | | 1D NMR | 1H NMR Spectrum (1D, 300 MHz, CDCl3, experimental) | | Spectrum | | 1D NMR | 13C NMR Spectrum (1D, 25.16 MHz, CDCl3, experimental) | | Spectrum | | 1D NMR | 1H NMR Spectrum (1D, D2O, experimental) | | Spectrum | | 1D NMR | 1H NMR Spectrum (1D, D2O, experimental) | | Spectrum | | 1D NMR | 1H NMR Spectrum (1D, D2O, experimental) | | Spectrum | | 1D NMR | 13C NMR Spectrum (1D, D2O, experimental) | | Spectrum | | 1D NMR | 1H NMR Spectrum (1D, 100 MHz, D2O, predicted) | | Spectrum | | 1D NMR | 13C NMR Spectrum (1D, 100 MHz, D2O, predicted) | | Spectrum | | 1D NMR | 1H NMR Spectrum (1D, 1000 MHz, D2O, predicted) | | Spectrum | | 1D NMR | 13C NMR Spectrum (1D, 1000 MHz, D2O, predicted) | | Spectrum | | 1D NMR | 1H NMR Spectrum (1D, 200 MHz, D2O, predicted) | | Spectrum | | 1D NMR | 13C NMR Spectrum (1D, 200 MHz, D2O, predicted) | | Spectrum | | 1D NMR | 1H NMR Spectrum (1D, 300 MHz, D2O, predicted) | | Spectrum | | 1D NMR | 13C NMR Spectrum (1D, 300 MHz, D2O, predicted) | | Spectrum | | 1D NMR | 1H NMR Spectrum (1D, 400 MHz, D2O, predicted) | | Spectrum | | 1D NMR | 13C NMR Spectrum (1D, 400 MHz, D2O, predicted) | | Spectrum | | 1D NMR | 1H NMR Spectrum (1D, 500 MHz, D2O, predicted) | | Spectrum | | 1D NMR | 13C NMR Spectrum (1D, 500 MHz, D2O, predicted) | | Spectrum | | 1D NMR | 1H NMR Spectrum (1D, 600 MHz, D2O, predicted) | | Spectrum | | 1D NMR | 13C NMR Spectrum (1D, 600 MHz, D2O, predicted) | | Spectrum | | 1D NMR | 1H NMR Spectrum (1D, 700 MHz, D2O, predicted) | | Spectrum | | 1D NMR | 13C NMR Spectrum (1D, 700 MHz, D2O, predicted) | | Spectrum | | 1D NMR | 1H NMR Spectrum (1D, 800 MHz, D2O, predicted) | | Spectrum | | 2D NMR | [1H, 13C]-HSQC NMR Spectrum (2D, 600 MHz, H2O, experimental) | | Spectrum |
|
|---|
| Pathways |
|---|
| Pathways | | Name | SMPDB/Pathwhiz | KEGG | | Urea Cycle | 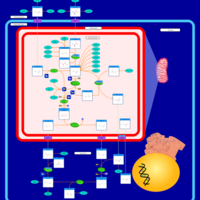 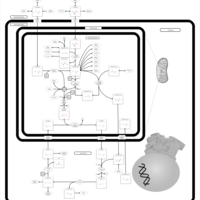 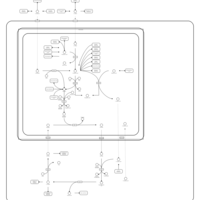 |  | | Glucose-Alanine Cycle | 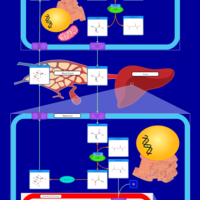 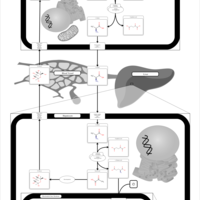  | Not Available | | Glycine and Serine Metabolism | 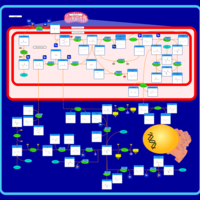 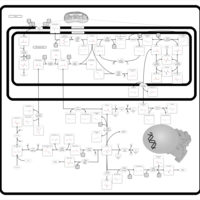  |  | | Pyruvate Metabolism |    | 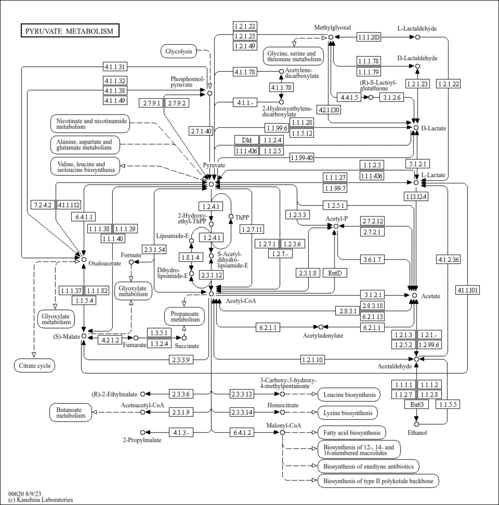 | | Alanine Metabolism |    |  |
|
|---|
| Protein Targets |
|---|
| Enzymes | |
| Pyruvate dehydrogenase E1 component subunit beta, mitochondrial | PDHB | 3p21.1-p14.2 | P11177 | details | | Pyruvate carboxylase, mitochondrial | PC | 11q13.4-q13.5 | P11498 | details | | Pyruvate dehydrogenase E1 component subunit alpha, somatic form, mitochondrial | PDHA1 | Xp22.1 | P08559 | details | | Pyruvate dehydrogenase E1 component subunit alpha, testis-specific form, mitochondrial | PDHA2 | 4q22-q23 | P29803 | details | | Dihydrolipoyl dehydrogenase, mitochondrial | DLD | 7q31-q32 | P09622 | details | | Dihydrolipoyllysine-residue acetyltransferase component of pyruvate dehydrogenase complex, mitochondrial | DLAT | 11q23.1 | P10515 | details | | D-amino-acid oxidase | DAO | 12q24 | P14920 | details | | 4-aminobutyrate aminotransferase, mitochondrial | ABAT | 16p13.2 | P80404 | details | | Alanine--glyoxylate aminotransferase 2, mitochondrial | AGXT2 | 5p13 | Q9BYV1 | details | | S-adenosylmethionine decarboxylase proenzyme | AMD1 | 6q21 | P17707 | details | | Kynurenine--oxoglutarate transaminase 1 | CCBL1 | 9q34.11 | Q16773 | details | | Cystathionine gamma-lyase | CTH | 1p31.1 | P32929 | details | | L-serine dehydratase/L-threonine deaminase | SDS | 12q24.13 | P20132 | details | | Adenylate cyclase type 7 | ADCY7 | 16q12.1 | P51828 | details | | Adenylate cyclase type 4 | ADCY4 | 14q12 | Q8NFM4 | details | | Adenylate cyclase type 6 | ADCY6 | 12q12-q13 | O43306 | details | | Adenylate cyclase type 5 | ADCY5 | 3q21.1 | O95622 | details | | Adenylate cyclase type 8 | ADCY8 | 8q24 | P40145 | details | | Adenylate cyclase type 9 | ADCY9 | 16p13.3 | O60503 | details | | Adenylate cyclase type 3 | ADCY3 | 2p23.3 | O60266 | details | | Adenylate cyclase type 1 | ADCY1 | 7p13-p12 | Q08828 | details | | Pyruvate kinase isozymes M1/M2 | PKM | 15q22 | P14618 | details | | Pyruvate kinase isozymes R/L | PKLR | 1q21 | P30613 | details | | 3-mercaptopyruvate sulfurtransferase | MPST | 22q13.1 | P25325 | details | | Serine--pyruvate aminotransferase | AGXT | 2q37.3 | P21549 | details | | Alanine aminotransferase 1 | GPT | 8q24.3 | P24298 | details | | Histidine decarboxylase | HDC | 15q21-q22 | P19113 | details | | L-lactate dehydrogenase A-like 6A | LDHAL6A | 11p15.1 | Q6ZMR3 | details | | Adenylate cyclase type 2 | ADCY2 | 5p15.3 | Q08462 | details | | Thiosulfate sulfurtransferase | TST | 22q13.1 | Q16762 | details | | Monocarboxylate transporter 3 | SLC16A8 | 22q12.3-q13.2 | O95907 | details | | Monocarboxylate transporter 7 | SLC16A6 | 17q24.2 | O15403 | details | | Monocarboxylate transporter 6 | SLC16A5 | 17q25.1 | O15375 | details | | Monocarboxylate transporter 5 | SLC16A4 | 1p13.3 | O15374 | details | | Phosphatidylserine decarboxylase proenzyme | PISD | 22q12.2 | Q9UG56 | details | | L-lactate dehydrogenase B chain | LDHB | 12p12.2-p12.1 | P07195 | details | | L-lactate dehydrogenase C chain | LDHC | 11p15.1 | P07864 | details | | L-lactate dehydrogenase A chain | LDHA | 11p15.4 | P00338 | details | | L-lactate dehydrogenase A-like 6B | LDHAL6B | 15q22.2 | Q9BYZ2 | details | | Probable D-lactate dehydrogenase, mitochondrial | LDHD | 16q23.1 | Q86WU2 | details | | Alanine aminotransferase 2 | GPT2 | 16q12.1 | Q8TD30 | details | | Selenocysteine lyase | SCLY | 2q37.3 | Q96I15 | details | | N-acetylneuraminate lyase | NPL | 1q25 | Q9BXD5 | details | | Acetolactate synthase-like protein | ILVBL | 19p13.1 | A1L0T0 | details | | Malic enzyme | ME3 | 11cen-q22.3 | Q8TBJ0 | details | | Acylpyruvase FAHD1, mitochondrial | FAHD1 | 16p13.3 | Q6P587 | details | | Kynurenine--oxoglutarate transaminase 3 | CCBL2 | 1p22.2 | Q6YP21 | details | | Serine dehydratase-like | SDSL | 12q24.13 | Q96GA7 | details | | Serine racemase | SRR | 17p13 | Q9GZT4 | details | | Probable 4-hydroxy-2-oxoglutarate aldolase, mitochondrial | HOGA1 | 10q24.2 | Q86XE5 | details |
|
|---|
| Transporters | |
|---|
| Metal Bindings | |
|---|
| Receptors | |
|---|
| Transcriptional Factors | |
|---|
| Concentrations Data |
|---|
| |
| Alien Dawg | Detected and Quantified | 0.00178 mg/g dry wt | | details | | Gabriola | Detected and Quantified | 0.00245 mg/g dry wt | | details | | Island Honey | Detected and Quantified | 0.00260 mg/g dry wt | | details | | Quadra | Detected and Quantified | 0.00238 mg/g dry wt | | details | | Sensi Star | Detected and Quantified | 0.00340 mg/g dry wt | | details | | Tangerine Dream | Detected and Quantified | 0.00271 mg/g dry wt | | details |
|
|---|
| External Links |
|---|
| HMDB ID | HMDB0000243 |
|---|
| DrugBank ID | DB00119 |
|---|
| Phenol Explorer Compound ID | Not Available |
|---|
| FoodDB ID | FDB031141 |
|---|
| KNApSAcK ID | C00001200 |
|---|
| Chemspider ID | Not Available |
|---|
| KEGG Compound ID | C00022 |
|---|
| BioCyc ID | PYRUVATE |
|---|
| BiGG ID | Not Available |
|---|
| Wikipedia Link | Pyruvic_acid |
|---|
| METLIN ID | Not Available |
|---|
| PubChem Compound | 1060 |
|---|
| PDB ID | Not Available |
|---|
| ChEBI ID | 32816 |
|---|
| References |
|---|
| General References | Not Available |
|---|



