| Record Information |
|---|
| Version | 1.0 |
|---|
| Created at | 2020-04-17 18:40:01 UTC |
|---|
| Updated at | 2020-12-07 19:11:01 UTC |
|---|
| CannabisDB ID | CDB004819 |
|---|
| Secondary Accession Numbers | Not Available |
|---|
| Cannabis Compound Identification |
|---|
| Common Name | Oxoglutaric acid |
|---|
| Description | Oxoglutaric acid, also known as oxoglutarate or alpha-ketoglutarate, belongs to the class of organic compounds known as gamma-keto acids and derivatives. These are organic compounds containing an aldehyde substituted with a keto group on the C4 carbon atom. Oxoglutaric acid is an extremely weak basic (essentially neutral) compound (based on its pKa). Oxoglutaric acid exists in all living species, ranging from bacteria to humans. Within humans, oxoglutaric acid participates in a number of enzymatic reactions. In particular, oxoglutaric acid can be biosynthesized from isocitric acid; which is mediated by the enzyme isocitrate dehydrogenase. In addition, oxoglutaric acid can be converted into succinyl-CoA; which is mediated by the enzyme oxoglutarate dehydrogenase complex. In humans, oxoglutaric acid is involved in the oncogenic action of fumarate. Oxoglutaric acid is an odorless tasting compound. Outside of the human body, Oxoglutaric acid is found, on average, in the highest concentration within star fruits and milk (cow). Oxoglutaric acid has also been detected, but not quantified in, several different foods, such as common chokecherries, red beetroots, adzuki beans, buffalo currants, and grapefruit/pummelo hybrids. This could make oxoglutaric acid a potential biomarker for the consumption of these foods. Oxoglutaric acid is a potentially toxic compound. An oxo dicarboxylic acid that consists of glutaric acid bearing an oxo substituent at position 2. Oxoglutaric acid is expected to be in Cannabis as all living plants are known to produce and metabolize it. |
|---|
| Structure | |
|---|
| Synonyms | | Value | Source |
|---|
| 2-Ketoglutaric acid | ChEBI | | alpha-Ketoglutaric acid | ChEBI | | 2-Ketoglutarate | Generator | | a-Ketoglutarate | Generator | | a-Ketoglutaric acid | Generator | | alpha-Ketoglutarate | Generator | | Α-ketoglutarate | Generator | | Α-ketoglutaric acid | Generator | | Oxoglutarate | Generator | | 2-oxo-1,5-Pentanedioate | HMDB | | 2-oxo-1,5-Pentanedioic acid | HMDB | | 2-Oxoglutarate | HMDB | | 2-Oxoglutaric acid | HMDB | | 2-Oxopentanedioate | HMDB | | 2-Oxopentanedioic acid | HMDB | | alpha-Oxoglutarate | HMDB | | Oxogluric acid | HMDB | | 2 Oxoglutaric acid | HMDB | | Ketoglutaric acid | HMDB | | alpha Ketoglutaric acid | HMDB | | 2 Ketoglutaric acid | HMDB | | alpha Ketoglutarate | HMDB | | alpha Oxoglutarate | HMDB | | 2 Ketoglutarate | HMDB | | 2 Oxoglutarate | HMDB | | alpha-Oxoglutaric acid | HMDB | | alpha-Oxopentanedioic acid | HMDB | | alpha-Keto-glutaric acid | HMDB | | Α-oxoglutaric acid | HMDB | | Α-oxopentanedioic acid | HMDB | | Α-keto-glutaric acid | HMDB |
|
|---|
| Chemical Formula | C5H6O5 |
|---|
| Average Molecular Weight | 146.1 |
|---|
| Monoisotopic Molecular Weight | 146.0215 |
|---|
| IUPAC Name | 2-oxopentanedioic acid |
|---|
| Traditional Name | oxoglutarate |
|---|
| CAS Registry Number | 328-50-7 |
|---|
| SMILES | OC(=O)CCC(=O)C(O)=O |
|---|
| InChI Identifier | InChI=1S/C5H6O5/c6-3(5(9)10)1-2-4(7)8/h1-2H2,(H,7,8)(H,9,10) |
|---|
| InChI Key | KPGXRSRHYNQIFN-UHFFFAOYSA-N |
|---|
| Chemical Taxonomy |
|---|
| Description | Belongs to the class of organic compounds known as gamma-keto acids and derivatives. These are organic compounds containing an aldehyde substituted with a keto group on the C4 carbon atom. |
|---|
| Kingdom | Organic compounds |
|---|
| Super Class | Organic acids and derivatives |
|---|
| Class | Keto acids and derivatives |
|---|
| Sub Class | Gamma-keto acids and derivatives |
|---|
| Direct Parent | Gamma-keto acids and derivatives |
|---|
| Alternative Parents | |
|---|
| Substituents | - Gamma-keto acid
- Short-chain keto acid
- Dicarboxylic acid or derivatives
- Alpha-keto acid
- Alpha-hydroxy ketone
- Ketone
- Carboxylic acid
- Carboxylic acid derivative
- Organic oxygen compound
- Organic oxide
- Hydrocarbon derivative
- Organooxygen compound
- Carbonyl group
- Aliphatic acyclic compound
|
|---|
| Molecular Framework | Aliphatic acyclic compounds |
|---|
| External Descriptors | |
|---|
| Ontology |
|---|
|
| Physiological effect | Health effect: |
|---|
| Disposition | Route of exposure: Source: Biological location: |
|---|
| Role | Biological role: Industrial application: |
|---|
| Physical Properties |
|---|
| State | Solid |
|---|
| Experimental Properties | | Property | Value | Reference |
|---|
| Melting Point | 115.5 °C | Not Available | | Boiling Point | Not Available | Not Available | | Water Solubility | 541.5 mg/mL | Not Available | | logP | Not Available | Not Available |
|
|---|
| Predicted Properties | [] |
|---|
| Spectra |
|---|
| EI-MS/GC-MS | | Type | Description | Splash Key | View |
|---|
| EI-MS | Mass Spectrum (Electron Ionization) | splash10-0fba-9200000000-4e367c66e94c51eb1a1b | 2014-09-20 | View Spectrum | | GC-MS | Oxoglutaric acid, 2 TMS, GC-MS Spectrum | splash10-000b-3910000000-4097b7eb52c2910a4277 | Spectrum | | GC-MS | Oxoglutaric acid, non-derivatized, GC-MS Spectrum | splash10-0002-1910000000-3277227b9c4da4baa1f7 | Spectrum | | GC-MS | Oxoglutaric acid, 2 TMS; 1 MEOX, GC-MS Spectrum | splash10-00di-9300000000-974bce546caa4fbe6b0f | Spectrum | | GC-MS | Oxoglutaric acid, 1 MEOX; 2 TMS, GC-MS Spectrum | splash10-000i-9710000000-d0aea11e947b042571f9 | Spectrum | | GC-MS | Oxoglutaric acid, 1 MEOX; 2 TMS, GC-MS Spectrum | splash10-052s-4920000000-bf7ed92c68fd4c8dbb6b | Spectrum | | GC-MS | Oxoglutaric acid, non-derivatized, GC-MS Spectrum | splash10-000b-3910000000-4097b7eb52c2910a4277 | Spectrum | | GC-MS | Oxoglutaric acid, non-derivatized, GC-MS Spectrum | splash10-0002-1910000000-3277227b9c4da4baa1f7 | Spectrum | | GC-MS | Oxoglutaric acid, non-derivatized, GC-MS Spectrum | splash10-00di-9300000000-974bce546caa4fbe6b0f | Spectrum | | GC-MS | Oxoglutaric acid, non-derivatized, GC-MS Spectrum | splash10-000i-9710000000-d0aea11e947b042571f9 | Spectrum | | GC-MS | Oxoglutaric acid, non-derivatized, GC-MS Spectrum | splash10-052s-4920000000-bf7ed92c68fd4c8dbb6b | Spectrum | | GC-MS | Oxoglutaric acid, non-derivatized, GC-MS Spectrum | splash10-000b-2900000000-e514f7afea2490bbe9ed | Spectrum | | Predicted GC-MS | Oxoglutaric acid, non-derivatized, Predicted GC-MS Spectrum - 70eV, Positive | splash10-0kfx-9200000000-0ee77a39e9862659f8a7 | Spectrum | | Predicted GC-MS | Oxoglutaric acid, 2 TMS, Predicted GC-MS Spectrum - 70eV, Positive | splash10-00di-9730000000-5e17bd8cfd944963c79f | Spectrum | | Predicted GC-MS | Oxoglutaric acid, non-derivatized, Predicted GC-MS Spectrum - 70eV, Positive | Not Available | Spectrum | | Predicted GC-MS | Oxoglutaric acid, TMS_1_1, Predicted GC-MS Spectrum - 70eV, Positive | Not Available | Spectrum | | Predicted GC-MS | Oxoglutaric acid, TMS_1_2, Predicted GC-MS Spectrum - 70eV, Positive | Not Available | Spectrum | | Predicted GC-MS | Oxoglutaric acid, TMS_1_3, Predicted GC-MS Spectrum - 70eV, Positive | Not Available | Spectrum | | Predicted GC-MS | Oxoglutaric acid, TMS_2_2, Predicted GC-MS Spectrum - 70eV, Positive | Not Available | Spectrum | | Predicted GC-MS | Oxoglutaric acid, TMS_2_3, Predicted GC-MS Spectrum - 70eV, Positive | Not Available | Spectrum | | Predicted GC-MS | Oxoglutaric acid, TBDMS_1_1, Predicted GC-MS Spectrum - 70eV, Positive | Not Available | Spectrum | | Predicted GC-MS | Oxoglutaric acid, TBDMS_1_2, Predicted GC-MS Spectrum - 70eV, Positive | Not Available | Spectrum | | Predicted GC-MS | Oxoglutaric acid, TBDMS_1_3, Predicted GC-MS Spectrum - 70eV, Positive | Not Available | Spectrum | | Predicted GC-MS | Oxoglutaric acid, TBDMS_2_1, Predicted GC-MS Spectrum - 70eV, Positive | Not Available | Spectrum | | Predicted GC-MS | Oxoglutaric acid, TBDMS_2_2, Predicted GC-MS Spectrum - 70eV, Positive | Not Available | Spectrum | | Predicted GC-MS | Oxoglutaric acid, TBDMS_2_3, Predicted GC-MS Spectrum - 70eV, Positive | Not Available | Spectrum |
|
|---|
| MS/MS | | Type | Description | Splash Key | View |
|---|
| MS/MS | LC-MS/MS Spectrum - Quattro_QQQ 10V, Negative (Annotated) | splash10-0zfr-6900000000-7fc343a243ce5d2a4f39 | 2012-07-24 | View Spectrum | | MS/MS | LC-MS/MS Spectrum - Quattro_QQQ 25V, Negative (Annotated) | splash10-0a4i-9200000000-1a7af9946604baa8b3d2 | 2012-07-24 | View Spectrum | | MS/MS | LC-MS/MS Spectrum - Quattro_QQQ 40V, Negative (Annotated) | splash10-0a4l-9800000000-30495b2afdd3526854e7 | 2012-07-24 | View Spectrum | | MS/MS | LC-MS/MS Spectrum - LC-ESI-ITFT (LTQ Orbitrap XL, Thermo Scientfic) , Negative | splash10-0002-0925200000-af33449c9638276466c2 | 2012-08-31 | View Spectrum | | MS/MS | LC-MS/MS Spectrum - LC-ESI-ITFT (LTQ Orbitrap XL, Thermo Scientfic) , Negative | splash10-0a4i-9000000000-6e43f767847790112b65 | 2012-08-31 | View Spectrum | | MS/MS | LC-MS/MS Spectrum - LC-ESI-ITFT (LTQ Orbitrap XL, Thermo Scientfic) , Negative | splash10-03di-0900000000-4cacc41705b7b385e91e | 2012-08-31 | View Spectrum | | MS/MS | LC-MS/MS Spectrum - LC-ESI-ITFT (LTQ Orbitrap XL, Thermo Scientfic) , Negative | splash10-0udi-0900000000-1367eeeb6ad64c67a0aa | 2012-08-31 | View Spectrum | | MS/MS | LC-MS/MS Spectrum - LC-ESI-ITFT (LTQ Orbitrap XL, Thermo Scientfic) , Negative | splash10-000t-0915051100-91cda631ee54800322b8 | 2012-08-31 | View Spectrum | | MS/MS | LC-MS/MS Spectrum - LC-ESI-ITFT (LTQ Orbitrap XL, Thermo Scientfic) , Negative | splash10-0a4i-9000000000-4f842c3673925d29c402 | 2012-08-31 | View Spectrum | | MS/MS | LC-MS/MS Spectrum - LC-ESI-ITFT (LTQ Orbitrap XL, Thermo Scientfic) , Negative | splash10-0udi-0900000000-5e751351656f6eb62de3 | 2012-08-31 | View Spectrum | | MS/MS | LC-MS/MS Spectrum - LC-ESI-ITFT (LTQ Orbitrap XL, Thermo Scientfic) , Negative | splash10-001i-0910000000-7729faa24733fbad77b7 | 2012-08-31 | View Spectrum | | MS/MS | LC-MS/MS Spectrum - LC-ESI-QQ (API3000, Applied Biosystems) 10V, Negative | splash10-0002-0900000000-c3c62f83bff99f44f7ab | 2012-08-31 | View Spectrum | | MS/MS | LC-MS/MS Spectrum - LC-ESI-QQ (API3000, Applied Biosystems) 20V, Negative | splash10-0zfr-6900000000-b6d8379db3e3f1480b14 | 2012-08-31 | View Spectrum | | MS/MS | LC-MS/MS Spectrum - LC-ESI-QQ (API3000, Applied Biosystems) 30V, Negative | splash10-0a4i-9300000000-4fde39bb93cde5a27f23 | 2012-08-31 | View Spectrum | | MS/MS | LC-MS/MS Spectrum - LC-ESI-QQ (API3000, Applied Biosystems) 40V, Negative | splash10-00i0-9000000000-4343832f3a55842423a9 | 2012-08-31 | View Spectrum | | Predicted MS/MS | Predicted LC-MS/MS Spectrum - 10V, Positive | splash10-004i-2900000000-5aa3d6d620db1d733a65 | 2015-05-26 | View Spectrum | | Predicted MS/MS | Predicted LC-MS/MS Spectrum - 20V, Positive | splash10-0m30-7900000000-fc77f69ad43f915bf23e | 2015-05-26 | View Spectrum | | Predicted MS/MS | Predicted LC-MS/MS Spectrum - 40V, Positive | splash10-0a6r-9000000000-c597f1f5e416037aa82d | 2015-05-26 | View Spectrum | | Predicted MS/MS | Predicted LC-MS/MS Spectrum - 10V, Positive | splash10-004i-2900000000-5aa3d6d620db1d733a65 | 2015-05-26 | View Spectrum | | Predicted MS/MS | Predicted LC-MS/MS Spectrum - 20V, Positive | splash10-0m30-7900000000-fc77f69ad43f915bf23e | 2015-05-26 | View Spectrum | | Predicted MS/MS | Predicted LC-MS/MS Spectrum - 40V, Positive | splash10-0a6r-9000000000-c597f1f5e416037aa82d | 2015-05-26 | View Spectrum | | Predicted MS/MS | Predicted LC-MS/MS Spectrum - 10V, Negative | splash10-0002-1900000000-f2b463076a150a514f0c | 2015-05-27 | View Spectrum | | Predicted MS/MS | Predicted LC-MS/MS Spectrum - 20V, Negative | splash10-0f9t-8900000000-b2d3fa8d7ab01a220d9a | 2015-05-27 | View Spectrum | | Predicted MS/MS | Predicted LC-MS/MS Spectrum - 40V, Negative | splash10-053r-9100000000-554bfbd6839bc376cba0 | 2015-05-27 | View Spectrum | | Predicted MS/MS | Predicted LC-MS/MS Spectrum - 10V, Negative | splash10-0002-1900000000-f2b463076a150a514f0c | 2015-05-27 | View Spectrum |
|
|---|
| NMR | | Type | Description | | View |
|---|
| 1D NMR | 1H NMR Spectrum (1D, 600 MHz, H2O, experimental) | | Spectrum | | 1D NMR | 13C NMR Spectrum (1D, 125 MHz, H2O, experimental) | | Spectrum | | 1D NMR | 1H NMR Spectrum (1D, 400 MHz, DMSO-d6, experimental) | | Spectrum | | 1D NMR | 13C NMR Spectrum (1D, 100.54 MHz, DMSO-d6, experimental) | | Spectrum | | 1D NMR | 1H NMR Spectrum (1D, D2O, experimental) | | Spectrum | | 1D NMR | 13C NMR Spectrum (1D, 100 MHz, D2O, predicted) | | Spectrum | | 1D NMR | 1H NMR Spectrum (1D, 100 MHz, D2O, predicted) | | Spectrum | | 1D NMR | 13C NMR Spectrum (1D, 1000 MHz, D2O, predicted) | | Spectrum | | 1D NMR | 1H NMR Spectrum (1D, 1000 MHz, D2O, predicted) | | Spectrum | | 1D NMR | 13C NMR Spectrum (1D, 200 MHz, D2O, predicted) | | Spectrum | | 1D NMR | 1H NMR Spectrum (1D, 200 MHz, D2O, predicted) | | Spectrum | | 1D NMR | 13C NMR Spectrum (1D, 300 MHz, D2O, predicted) | | Spectrum | | 1D NMR | 1H NMR Spectrum (1D, 300 MHz, D2O, predicted) | | Spectrum | | 1D NMR | 13C NMR Spectrum (1D, 400 MHz, D2O, predicted) | | Spectrum | | 1D NMR | 1H NMR Spectrum (1D, 400 MHz, D2O, predicted) | | Spectrum | | 1D NMR | 13C NMR Spectrum (1D, 500 MHz, D2O, predicted) | | Spectrum | | 1D NMR | 1H NMR Spectrum (1D, 500 MHz, D2O, predicted) | | Spectrum | | 1D NMR | 13C NMR Spectrum (1D, 600 MHz, D2O, predicted) | | Spectrum | | 1D NMR | 1H NMR Spectrum (1D, 600 MHz, D2O, predicted) | | Spectrum | | 1D NMR | 13C NMR Spectrum (1D, 700 MHz, D2O, predicted) | | Spectrum | | 1D NMR | 1H NMR Spectrum (1D, 700 MHz, D2O, predicted) | | Spectrum | | 1D NMR | 13C NMR Spectrum (1D, 800 MHz, D2O, predicted) | | Spectrum | | 1D NMR | 1H NMR Spectrum (1D, 800 MHz, D2O, predicted) | | Spectrum | | 1D NMR | 13C NMR Spectrum (1D, 900 MHz, D2O, predicted) | | Spectrum | | 1D NMR | 1H NMR Spectrum (1D, 900 MHz, D2O, predicted) | | Spectrum | | 2D NMR | [1H, 13C]-HSQC NMR Spectrum (2D, 600 MHz, H2O, experimental) | | Spectrum |
|
|---|
| Pathways |
|---|
| Pathways | | Name | SMPDB/Pathwhiz | KEGG | | Citric Acid Cycle | 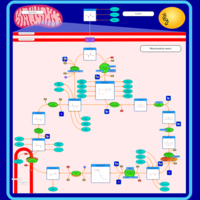 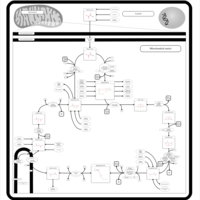 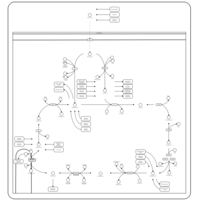 | 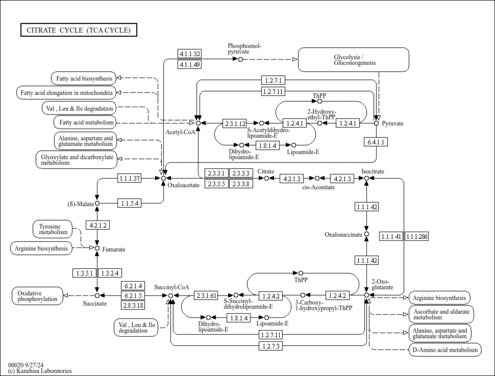 | | Gluconeogenesis | 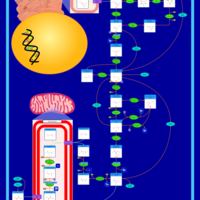 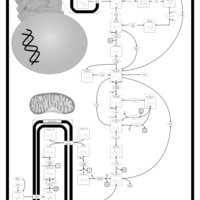 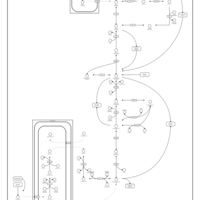 | 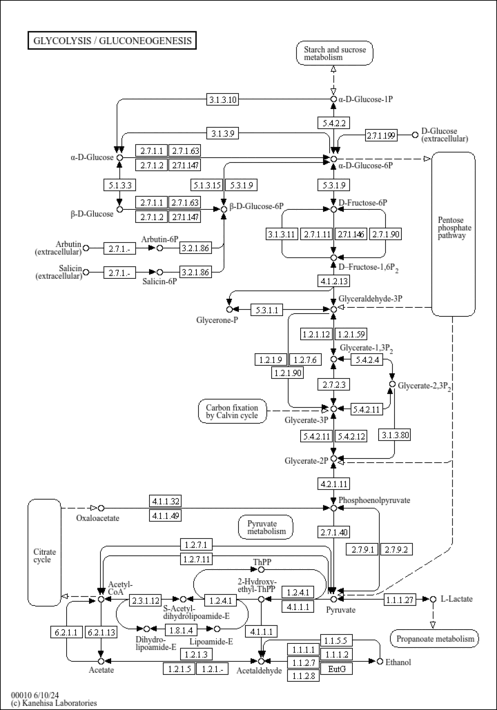 | | Glutamate Metabolism | 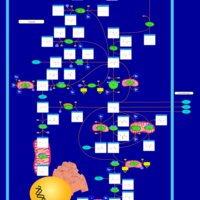 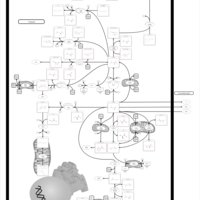 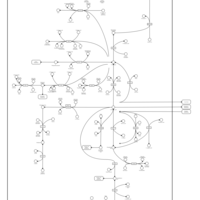 |  | | Urea Cycle | 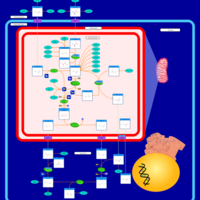 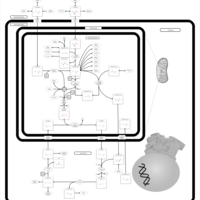 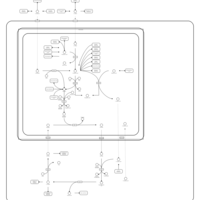 |  | | Glucose-Alanine Cycle | 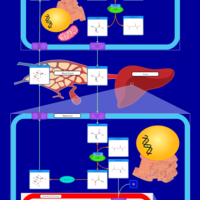 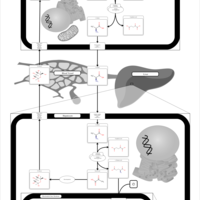 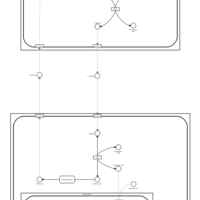 | Not Available |
|
|---|
| Protein Targets |
|---|
| Enzymes | |
| Dihydrolipoyl dehydrogenase, mitochondrial | DLD | 7q31-q32 | P09622 | details | | 4-aminobutyrate aminotransferase, mitochondrial | ABAT | 16p13.2 | P80404 | details | | Phytanoyl-CoA dioxygenase, peroxisomal | PHYH | 10p13 | O14832 | details | | Tyrosine aminotransferase | TAT | 16q22.1 | P17735 | details | | Aspartate aminotransferase, cytoplasmic | GOT1 | 10q24.1-q25.1 | P17174 | details | | Aspartate aminotransferase, mitochondrial | GOT2 | 16q21 | P00505 | details | | Aldehyde dehydrogenase, mitochondrial | ALDH2 | 12q24.2 | P05091 | details | | Fatty aldehyde dehydrogenase | ALDH3A2 | 17p11.2 | P51648 | details | | Aldehyde dehydrogenase X, mitochondrial | ALDH1B1 | 9p11.1 | P30837 | details | | Branched-chain-amino-acid aminotransferase, cytosolic | BCAT1 | 12p12.1 | P54687 | details | | Prolyl 4-hydroxylase subunit alpha-2 | P4HA2 | 5q31 | O15460 | details | | Prolyl 4-hydroxylase subunit alpha-1 | P4HA1 | 10q21.3-q23.1 | P13674 | details | | Kynurenine/alpha-aminoadipate aminotransferase, mitochondrial | AADAT | 4q33 | Q8N5Z0 | details | | Gamma-butyrobetaine dioxygenase | BBOX1 | 11p14.2 | O75936 | details | | Alanine--glyoxylate aminotransferase 2, mitochondrial | AGXT2 | 5p13 | Q9BYV1 | details | | Ornithine aminotransferase, mitochondrial | OAT | 10q26 | P04181 | details | | 2-oxoglutarate dehydrogenase, mitochondrial | OGDH | 7p14-p13 | Q02218 | details | | Procollagen-lysine,2-oxoglutarate 5-dioxygenase 1 | PLOD1 | 1p36.22 | Q02809 | details | | Procollagen-lysine,2-oxoglutarate 5-dioxygenase 2 | PLOD2 | 3q24 | O00469 | details | | Procollagen-lysine,2-oxoglutarate 5-dioxygenase 3 | PLOD3 | 7q22 | O60568 | details | | Kynurenine--oxoglutarate transaminase 1 | CCBL1 | 9q34.11 | Q16773 | details | | Branched-chain-amino-acid aminotransferase, mitochondrial | BCAT2 | 19q13 | O15382 | details | | Glutamate dehydrogenase 2, mitochondrial | GLUD2 | Xq24-q25 | P49448 | details | | Glutamate dehydrogenase 1, mitochondrial | GLUD1 | 10q23.3 | P00367 | details | | Serine--pyruvate aminotransferase | AGXT | 2q37.3 | P21549 | details | | Alanine aminotransferase 1 | GPT | 8q24.3 | P24298 | details | | Phosphoserine aminotransferase | PSAT1 | 9q21.2 | Q9Y617 | details | | Alpha-aminoadipic semialdehyde synthase, mitochondrial | AASS | 7q31.3 | Q9UDR5 | details | | Isocitrate dehydrogenase [NADP], mitochondrial | IDH2 | 15q26.1 | P48735 | details | | Isocitrate dehydrogenase [NAD] subunit beta, mitochondrial | IDH3B | 20p13 | O43837 | details | | Isocitrate dehydrogenase [NADP] cytoplasmic | IDH1 | 2q33.3 | O75874 | details | | Isocitrate dehydrogenase [NAD] subunit alpha, mitochondrial | IDH3A | 15q25.1-q25.2 | P50213 | details | | Isocitrate dehydrogenase [NAD] subunit gamma, mitochondrial | IDH3G | Xq28 | P51553 | details | | D-3-phosphoglycerate dehydrogenase | PHGDH | 1p12 | O43175 | details | | Aspartyl/asparaginyl beta-hydroxylase | ASPH | 8q12.1 | Q12797 | details | | Mitochondrial 2-oxoglutarate/malate carrier protein | SLC25A11 | 17p13.3 | Q02978 | details | | D-2-hydroxyglutarate dehydrogenase, mitochondrial | D2HGDH | 2q37.3 | Q8N465 | details | | L-2-hydroxyglutarate dehydrogenase, mitochondrial | L2HGDH | 14q21.3 | Q9H9P8 | details | | Aminoadipate aminotransferase, isoform CRA_b | AADAT | 4q33 | Q4W5N8 | details | | Trimethyllysine dioxygenase, mitochondrial | TMLHE | Xq28 | Q9NVH6 | details | | Alanine aminotransferase 2 | GPT2 | 16q12.1 | Q8TD30 | details | | Hydroxyacid-oxoacid transhydrogenase, mitochondrial | ADHFE1 | 8q13.1 | Q8IWW8 | details | | Aminoadipate-semialdehyde synthase | AASS | 7q31.3 | A4D0W4 | details | | Alpha-ketoglutarate-dependent dioxygenase alkB homolog 2 | ALKBH2 | 12q24.11 | Q6NS38 | details | | Prolyl 3-hydroxylase 1 | LEPRE1 | 1p34.1 | Q32P28 | details | | Prolyl 3-hydroxylase 2 | LEPREL1 | 3q28 | Q8IVL5 | details | | Prolyl 3-hydroxylase 3 | LEPREL2 | 12q13 | Q8IVL6 | details | | Putative aspartate aminotransferase, cytoplasmic 2 | GOT1L1 | 8p11.23 | Q8NHS2 | details | | Kynurenine--oxoglutarate transaminase 3 | CCBL2 | 1p22.2 | Q6YP21 | details | | Probable 2-oxoglutarate dehydrogenase E1 component DHKTD1, mitochondrial | DHTKD1 | 10p14 | Q96HY7 | details | | 2-oxoglutarate dehydrogenase-like, mitochondrial | OGDHL | | Q9ULD0 | details | | Prolyl 4-hydroxylase subunit alpha-3 | P4HA3 | 11q13.4 | Q7Z4N8 | details | | Lysine-specific demethylase 2A | KDM2A | 11q13.2 | Q9Y2K7 | details | | Lysine-specific demethylase 2B | KDM2B | 12q24.31 | Q8NHM5 | details | | Egl nine homolog 1 | EGLN1 | 1q42.1 | Q9GZT9 | details | | Egl nine homolog 2 | EGLN2 | 19q13.2 | Q96KS0 | details | | Egl nine homolog 3 | EGLN3 | 14q13.1 | Q9H6Z9 | details | | Transmembrane prolyl 4-hydroxylase | P4HTM | 3p21.3 | Q9NXG6 | details | | Lysine-specific demethylase 8 | KDM8 | 16p12.1 | Q8N371 | details | | Bifunctional lysine-specific demethylase and histidyl-hydroxylase MINA | MINA | 3q11.2 | Q8IUF8 | details | | Omega-amidase NIT2 | NIT2 | 3q12.2 | Q9NQR4 | details | | Bifunctional lysine-specific demethylase and histidyl-hydroxylase NO66 | NO66 | 14q24.3 | Q9H6W3 | details | | Histone lysine demethylase PHF8 | PHF8 | Xp11.22 | Q9UPP1 | details | | Methylcytosine dioxygenase TET1 | TET1 | 10q21 | Q8NFU7 | details | | Methylcytosine dioxygenase TET2 | TET2 | 4q24 | Q6N021 | details | | Methylcytosine dioxygenase TET3 | TET3 | 2p13.1 | O43151 | details |
|
|---|
| Transporters | |
| Mitochondrial 2-oxoglutarate/malate carrier protein | SLC25A11 | 17p13.3 | Q02978 | details |
|
|---|
| Metal Bindings | |
| Phytanoyl-CoA dioxygenase, peroxisomal | PHYH | 10p13 | O14832 | details | | Prolyl 4-hydroxylase subunit alpha-2 | P4HA2 | 5q31 | O15460 | details | | Prolyl 4-hydroxylase subunit alpha-1 | P4HA1 | 10q21.3-q23.1 | P13674 | details | | Gamma-butyrobetaine dioxygenase | BBOX1 | 11p14.2 | O75936 | details | | Procollagen-lysine,2-oxoglutarate 5-dioxygenase 1 | PLOD1 | 1p36.22 | Q02809 | details | | Procollagen-lysine,2-oxoglutarate 5-dioxygenase 2 | PLOD2 | 3q24 | O00469 | details | | Procollagen-lysine,2-oxoglutarate 5-dioxygenase 3 | PLOD3 | 7q22 | O60568 | details | | Isocitrate dehydrogenase [NADP], mitochondrial | IDH2 | 15q26.1 | P48735 | details | | Isocitrate dehydrogenase [NAD] subunit beta, mitochondrial | IDH3B | 20p13 | O43837 | details | | Isocitrate dehydrogenase [NADP] cytoplasmic | IDH1 | 2q33.3 | O75874 | details | | Isocitrate dehydrogenase [NAD] subunit alpha, mitochondrial | IDH3A | 15q25.1-q25.2 | P50213 | details | | Isocitrate dehydrogenase [NAD] subunit gamma, mitochondrial | IDH3G | Xq28 | P51553 | details | | Trimethyllysine dioxygenase, mitochondrial | TMLHE | Xq28 | Q9NVH6 | details | | Hydroxyacid-oxoacid transhydrogenase, mitochondrial | ADHFE1 | 8q13.1 | Q8IWW8 | details | | Prolyl 3-hydroxylase 1 | LEPRE1 | 1p34.1 | Q32P28 | details | | Prolyl 3-hydroxylase 2 | LEPREL1 | 3q28 | Q8IVL5 | details | | Prolyl 3-hydroxylase 3 | LEPREL2 | 12q13 | Q8IVL6 | details | | Prolyl 4-hydroxylase subunit alpha-3 | P4HA3 | 11q13.4 | Q7Z4N8 | details | | Lysine-specific demethylase 2A | KDM2A | 11q13.2 | Q9Y2K7 | details | | Lysine-specific demethylase 2B | KDM2B | 12q24.31 | Q8NHM5 | details | | Egl nine homolog 1 | EGLN1 | 1q42.1 | Q9GZT9 | details | | Egl nine homolog 2 | EGLN2 | 19q13.2 | Q96KS0 | details | | Egl nine homolog 3 | EGLN3 | 14q13.1 | Q9H6Z9 | details | | Transmembrane prolyl 4-hydroxylase | P4HTM | 3p21.3 | Q9NXG6 | details | | Lysine-specific demethylase 8 | KDM8 | 16p12.1 | Q8N371 | details | | Methylcytosine dioxygenase TET2 | TET2 | 4q24 | Q6N021 | details | | Methylcytosine dioxygenase TET3 | TET3 | 2p13.1 | O43151 | details |
|
|---|
| Receptors | |
|---|
| Transcriptional Factors | |
|---|
| Concentrations Data |
|---|
| Not Available |
|---|
| External Links |
|---|
| HMDB ID | HMDB0000208 |
|---|
| DrugBank ID | DB08845 |
|---|
| Phenol Explorer Compound ID | Not Available |
|---|
| FoodDB ID | FDB003361 |
|---|
| KNApSAcK ID | C00000769 |
|---|
| Chemspider ID | 50 |
|---|
| KEGG Compound ID | C00026 |
|---|
| BioCyc ID | 2-KETOGLUTARATE |
|---|
| BiGG ID | 33565 |
|---|
| Wikipedia Link | Alpha-Ketoglutaric_acid |
|---|
| METLIN ID | 5218 |
|---|
| PubChem Compound | 51 |
|---|
| PDB ID | Not Available |
|---|
| ChEBI ID | 30915 |
|---|
| References |
|---|
| General References | Not Available |
|---|



