| Record Information |
|---|
| Version | 1.0 |
|---|
| Created at | 2020-04-17 18:37:09 UTC |
|---|
| Updated at | 2020-12-07 19:10:56 UTC |
|---|
| CannabisDB ID | CDB004791 |
|---|
| Secondary Accession Numbers | Not Available |
|---|
| Cannabis Compound Identification |
|---|
| Common Name | Adenosine |
|---|
| Description | Adenosine, also known as adenocard or ade-rib, belongs to the class of organic compounds known as purine nucleosides. Purine nucleosides are compounds comprising a purine base attached to a ribosyl or deoxyribosyl moiety. A ribonucleoside composed of a molecule of adenine attached to a ribofuranose moiety via a beta-N(9)-glycosidic bond. Adenosine is a drug which is used as an initial treatment for the termination of paroxysmal supraventricular tachycardia (pvst), including that associated with accessory bypass tracts, and is a drug of choice for terminating stable, narrow-complex supraventricular tachycardias (svt). also used as an adjunct to thallous chloride ti 201 myocardial perfusion scintigraphy (thallium stress test) in patients who are unable to exercise adequately, as well as an adjunct to vagal maneuvers and clinical assessment to establish a specific diagnosis of undefined, stable, narrow-complex svt. Adenosine is a strong basic compound (based on its pKa). Adenosine exists in all living species, ranging from bacteria to humans. Within humans, adenosine participates in a number of enzymatic reactions. In particular, adenosine can be converted into inosine; which is mediated by the enzyme adenosine deaminase. In addition, adenosine can be converted into adenine and ribose 1-phosphate; which is mediated by the enzyme purine nucleoside phosphorylase. In humans, adenosine is involved in thioguanine action pathway. Outside of the human body, Adenosine is found, on average, in the highest concentration within beers and milk (cow). Adenosine has also been detected, but not quantified in, several different foods, such as capers, new zealand spinachs, welsh onions, hickory nuts, and pepper (spice). This could make adenosine a potential biomarker for the consumption of these foods. Adenosine is a potentially toxic compound. Adenosine is expected to be in Cannabis as all living plants are known to produce and metabolize it. |
|---|
| Structure | |
|---|
| Synonyms | | Value | Source |
|---|
| (2R,3R,4S,5R)-2-(6-Aminopurin-9-yl)-5-(hydroxymethyl)oxolane-3,4-diol | ChEBI | | 6-Amino-9-beta-D-ribofuranosyl-9H-purine | ChEBI | | 9-beta-D-Ribofuranosidoadenine | ChEBI | | 9-beta-D-Ribofuranosyl-9H-purin-6-amine | ChEBI | | Ade-rib | ChEBI | | Adenine deoxyribonucleoside | ChEBI | | Adenocard | ChEBI | | Adenocor | ChEBI | | Adenoscan | ChEBI | | Adenosin | ChEBI | | Adenyldeoxyriboside | ChEBI | | Ado | ChEBI | | beta-D-Adenosine | ChEBI | | Deoxyadenosine | ChEBI | | Desoxyadenosine | ChEBI | | 6-Amino-9-b-D-ribofuranosyl-9H-purine | Generator | | 6-Amino-9-β-D-ribofuranosyl-9H-purine | Generator | | 9-b-D-Ribofuranosidoadenine | Generator | | 9-Β-D-ribofuranosidoadenine | Generator | | 9-b-D-Ribofuranosyl-9H-purin-6-amine | Generator | | 9-Β-D-ribofuranosyl-9H-purin-6-amine | Generator | | b-D-Adenosine | Generator | | Β-D-adenosine | Generator | | 1-(6-Amino-9H-purin-9-yl)-1-deoxy-beta-D-ribofuranose | HMDB | | 1-(6-Amino-9H-purin-9-yl)-1-deoxy-beta-delta-ribofuranose | HMDB | | 6-Amino-9beta-D-ribofuranosyl-9H-purine | HMDB | | 6-Amino-9beta-delta-ribofuranosyl-9H-purine | HMDB | | 9-beta-D-Arabinofuranosyladenine | HMDB | | 9-beta-D-Ribofuranosyladenine | HMDB | | 9-beta-delta-Arabinofuranosyladenine | HMDB | | 9-beta-delta-Ribofuranosidoadenine | HMDB | | 9-beta-delta-Ribofuranosyl-9H-purin-6-amine | HMDB | | 9-beta-delta-Ribofuranosyladenine | HMDB | | 9beta-D-Ribofuranosyl-9H-purin-6-amine | HMDB | | 9beta-D-Ribofuranosyladenine | HMDB | | 9beta-delta-Ribofuranosyl-9H-purin-6-amine | HMDB | | 9beta-delta-Ribofuranosyladenine | HMDB | | Adenine nucleoside | HMDB | | Adenine riboside | HMDB | | Adenine-9beta-D-ribofuranoside | HMDB | | Adenine-9beta-delta-ribofuranoside | HMDB | | beta-Adenosine | HMDB | | beta-delta-Adenosine | HMDB | | Boniton | HMDB | | Myocol | HMDB | | Nucleocardyl | HMDB | | Sandesin | HMDB |
|
|---|
| Chemical Formula | C10H13N5O4 |
|---|
| Average Molecular Weight | 267.24 |
|---|
| Monoisotopic Molecular Weight | 267.0968 |
|---|
| IUPAC Name | (2R,3R,4S,5R)-2-(6-amino-9H-purin-9-yl)-5-(hydroxymethyl)oxolane-3,4-diol |
|---|
| Traditional Name | adenosine |
|---|
| CAS Registry Number | 58-61-7 |
|---|
| SMILES | NC1=C2N=CN([C@@H]3O[C@H](CO)[C@@H](O)[C@H]3O)C2=NC=N1 |
|---|
| InChI Identifier | InChI=1S/C10H13N5O4/c11-8-5-9(13-2-12-8)15(3-14-5)10-7(18)6(17)4(1-16)19-10/h2-4,6-7,10,16-18H,1H2,(H2,11,12,13)/t4-,6-,7-,10-/m1/s1 |
|---|
| InChI Key | OIRDTQYFTABQOQ-KQYNXXCUSA-N |
|---|
| Chemical Taxonomy |
|---|
| Description | Belongs to the class of organic compounds known as purine nucleosides. Purine nucleosides are compounds comprising a purine base attached to a ribosyl or deoxyribosyl moiety. |
|---|
| Kingdom | Organic compounds |
|---|
| Super Class | Nucleosides, nucleotides, and analogues |
|---|
| Class | Purine nucleosides |
|---|
| Sub Class | Not Available |
|---|
| Direct Parent | Purine nucleosides |
|---|
| Alternative Parents | |
|---|
| Substituents | - Purine nucleoside
- Glycosyl compound
- N-glycosyl compound
- 6-aminopurine
- Pentose monosaccharide
- Imidazopyrimidine
- Purine
- Aminopyrimidine
- Monosaccharide
- N-substituted imidazole
- Pyrimidine
- Imidolactam
- Tetrahydrofuran
- Azole
- Imidazole
- Heteroaromatic compound
- Secondary alcohol
- Organoheterocyclic compound
- Azacycle
- Oxacycle
- Organic oxygen compound
- Organic nitrogen compound
- Alcohol
- Organonitrogen compound
- Hydrocarbon derivative
- Organopnictogen compound
- Organooxygen compound
- Amine
- Primary alcohol
- Primary amine
- Aromatic heteropolycyclic compound
|
|---|
| Molecular Framework | Aromatic heteropolycyclic compounds |
|---|
| External Descriptors | |
|---|
| Ontology |
|---|
|
| Physiological effect | Health effect: |
|---|
| Disposition | Route of exposure: Source: Biological location: |
|---|
| Role | Indirect biological role: Biological role: Industrial application: |
|---|
| Physical Properties |
|---|
| State | Solid |
|---|
| Experimental Properties | | Property | Value | Reference |
|---|
| Melting Point | 235.5 °C | Not Available | | Boiling Point | Not Available | Not Available | | Water Solubility | 37 mg/mL | Human Metabolome Project | | logP | -1.05 | HANSCH,C ET AL. (1995) |
|
|---|
| Predicted Properties | [] |
|---|
| Spectra |
|---|
| EI-MS/GC-MS | | Type | Description | Splash Key | View |
|---|
| GC-MS | Adenosine, 4 TMS, GC-MS Spectrum | splash10-0f7w-1890000000-5c70a5423faa5e8eceaf | Spectrum | | GC-MS | Adenosine, 4 TMS, GC-MS Spectrum | splash10-001v-0691000000-8f88854b7d5adec5c558 | Spectrum | | GC-MS | Adenosine, non-derivatized, GC-MS Spectrum | splash10-001s-0790000000-cc3bcd977e11efd4ea25 | Spectrum | | GC-MS | Adenosine, 4 TMS, GC-MS Spectrum | splash10-00di-9460000000-3ae19bfaa5d2df72f9b1 | Spectrum | | GC-MS | Adenosine, 3 TMS, GC-MS Spectrum | splash10-0gc0-0590000000-05874cecded3009c1f4d | Spectrum | | GC-MS | Adenosine, 4 TMS, GC-MS Spectrum | splash10-0fsv-1792000000-b37fd6826f28aaf9e825 | Spectrum | | GC-MS | Adenosine, non-derivatized, GC-MS Spectrum | splash10-0f7w-1890000000-5c70a5423faa5e8eceaf | Spectrum | | GC-MS | Adenosine, non-derivatized, GC-MS Spectrum | splash10-001v-0691000000-8f88854b7d5adec5c558 | Spectrum | | GC-MS | Adenosine, non-derivatized, GC-MS Spectrum | splash10-001s-0790000000-cc3bcd977e11efd4ea25 | Spectrum | | GC-MS | Adenosine, non-derivatized, GC-MS Spectrum | splash10-00di-9460000000-3ae19bfaa5d2df72f9b1 | Spectrum | | GC-MS | Adenosine, non-derivatized, GC-MS Spectrum | splash10-0gc0-0590000000-05874cecded3009c1f4d | Spectrum | | GC-MS | Adenosine, non-derivatized, GC-MS Spectrum | splash10-0fsv-1792000000-b37fd6826f28aaf9e825 | Spectrum | | Predicted GC-MS | Adenosine, non-derivatized, Predicted GC-MS Spectrum - 70eV, Positive | splash10-0a4v-9560000000-986f3b1dfa5d898ed7a2 | Spectrum | | Predicted GC-MS | Adenosine, 3 TMS, Predicted GC-MS Spectrum - 70eV, Positive | splash10-01c4-6912500000-ae103f43432726c1ad22 | Spectrum | | Predicted GC-MS | Adenosine, non-derivatized, Predicted GC-MS Spectrum - 70eV, Positive | Not Available | Spectrum | | Predicted GC-MS | Adenosine, TMS_1_1, Predicted GC-MS Spectrum - 70eV, Positive | Not Available | Spectrum | | Predicted GC-MS | Adenosine, TMS_1_2, Predicted GC-MS Spectrum - 70eV, Positive | Not Available | Spectrum | | Predicted GC-MS | Adenosine, TMS_1_3, Predicted GC-MS Spectrum - 70eV, Positive | Not Available | Spectrum | | Predicted GC-MS | Adenosine, TMS_1_4, Predicted GC-MS Spectrum - 70eV, Positive | Not Available | Spectrum | | Predicted GC-MS | Adenosine, TMS_2_1, Predicted GC-MS Spectrum - 70eV, Positive | Not Available | Spectrum | | Predicted GC-MS | Adenosine, TMS_2_2, Predicted GC-MS Spectrum - 70eV, Positive | Not Available | Spectrum | | Predicted GC-MS | Adenosine, TMS_2_3, Predicted GC-MS Spectrum - 70eV, Positive | Not Available | Spectrum | | Predicted GC-MS | Adenosine, TMS_2_4, Predicted GC-MS Spectrum - 70eV, Positive | Not Available | Spectrum | | Predicted GC-MS | Adenosine, TMS_2_5, Predicted GC-MS Spectrum - 70eV, Positive | Not Available | Spectrum | | Predicted GC-MS | Adenosine, TMS_2_6, Predicted GC-MS Spectrum - 70eV, Positive | Not Available | Spectrum |
|
|---|
| MS/MS | | Type | Description | Splash Key | View |
|---|
| MS/MS | LC-MS/MS Spectrum - Quattro_QQQ 10V, Positive (Annotated) | splash10-014r-0890000000-588861a9494ab4680698 | 2012-07-24 | View Spectrum | | MS/MS | LC-MS/MS Spectrum - Quattro_QQQ 25V, Positive (Annotated) | splash10-000i-1900000000-dd7cbed8e76c6e783831 | 2012-07-24 | View Spectrum | | MS/MS | LC-MS/MS Spectrum - Quattro_QQQ 40V, Positive (Annotated) | splash10-000i-2900000000-c6644520b5943b59403d | 2012-07-24 | View Spectrum | | MS/MS | LC-MS/MS Spectrum - LC-ESI-ITFT (LTQ Orbitrap XL, Thermo Scientfic) , Positive | splash10-014i-0090000000-3d4fd8ea211eaed10531 | 2012-08-31 | View Spectrum | | MS/MS | LC-MS/MS Spectrum - LC-ESI-ITFT (LTQ Orbitrap XL, Thermo Scientfic) , Positive | splash10-000i-0900000000-cb58ec56a5faa09c9a02 | 2012-08-31 | View Spectrum | | MS/MS | LC-MS/MS Spectrum - LC-ESI-ITFT (LTQ Orbitrap XL, Thermo Scientfic) , Positive | splash10-000i-0900000000-9559aa07ec13a688fb16 | 2012-08-31 | View Spectrum | | MS/MS | LC-MS/MS Spectrum - LC-ESI-ITFT (LTQ Orbitrap XL, Thermo Scientfic) , Positive | splash10-000i-0900000000-f7c1371d8e1058df3a32 | 2012-08-31 | View Spectrum | | MS/MS | LC-MS/MS Spectrum - LC-ESI-QQ (API3000, Applied Biosystems) 10V, Negative | splash10-001i-0940000000-da71247788bf1ba222e2 | 2012-08-31 | View Spectrum | | MS/MS | LC-MS/MS Spectrum - LC-ESI-QQ (API3000, Applied Biosystems) 20V, Negative | splash10-001i-0920000000-f2e8ec028abdd674b5e3 | 2012-08-31 | View Spectrum | | MS/MS | LC-MS/MS Spectrum - LC-ESI-QQ (API3000, Applied Biosystems) 30V, Negative | splash10-001i-0910000000-815ed65588c07415cd65 | 2012-08-31 | View Spectrum | | MS/MS | LC-MS/MS Spectrum - LC-ESI-QQ (API3000, Applied Biosystems) 40V, Negative | splash10-001i-1910000000-2780e737da8848411c4f | 2012-08-31 | View Spectrum | | MS/MS | LC-MS/MS Spectrum - LC-ESI-QQ (API3000, Applied Biosystems) 50V, Negative | splash10-053r-1900000000-c6c1128990661334c15b | 2012-08-31 | View Spectrum | | MS/MS | LC-MS/MS Spectrum - LC-ESI-QQ (API3000, Applied Biosystems) 10V, Positive | splash10-014i-0090000000-106e7131507fadd4befb | 2012-08-31 | View Spectrum | | MS/MS | LC-MS/MS Spectrum - LC-ESI-QQ (API3000, Applied Biosystems) 20V, Positive | splash10-000i-0910000000-1c53823486cd2c23f90e | 2012-08-31 | View Spectrum | | MS/MS | LC-MS/MS Spectrum - LC-ESI-QQ (API3000, Applied Biosystems) 30V, Positive | splash10-000i-0900000000-f0313435db4847490fd6 | 2012-08-31 | View Spectrum | | MS/MS | LC-MS/MS Spectrum - LC-ESI-QQ (API3000, Applied Biosystems) 40V, Positive | splash10-000i-1900000000-dcd4c2972882dcd7535a | 2012-08-31 | View Spectrum | | MS/MS | LC-MS/MS Spectrum - LC-ESI-QQ (API3000, Applied Biosystems) 50V, Positive | splash10-000i-2900000000-5ee474864189cef6df64 | 2012-08-31 | View Spectrum | | MS/MS | LC-MS/MS Spectrum - LC-ESI-IT (LC/MSD Trap XCT, Agilent Technologies) , Positive | splash10-000i-0900000000-17edf36faf6eb5750912 | 2012-08-31 | View Spectrum | | MS/MS | LC-MS/MS Spectrum - LC-ESI-IT (LC/MSD Trap XCT, Agilent Technologies) , Positive | splash10-000i-0900000000-0591143763a0bad31c23 | 2012-08-31 | View Spectrum | | MS/MS | LC-MS/MS Spectrum - LC-ESI-QTOF (UPLC Q-Tof Premier, Waters) , Positive | splash10-014i-0290000000-f1d71a97ce39145c8e31 | 2012-08-31 | View Spectrum | | MS/MS | LC-MS/MS Spectrum - LC-ESI-QTOF (UPLC Q-Tof Premier, Waters) 30V, Positive | splash10-00kr-0950000000-9bbdbb14aed287ad5ac8 | 2012-08-31 | View Spectrum | | MS/MS | LC-MS/MS Spectrum - LC-ESI-QTOF (UPLC Q-Tof Premier, Waters) , Negative | splash10-001i-0910000000-35f90965f1f3c2b3236b | 2012-08-31 | View Spectrum | | MS/MS | LC-MS/MS Spectrum - LC-ESI-QTOF (UPLC Q-Tof Premier, Waters) , Positive | splash10-000i-0900000000-f360cca994696d7ae735 | 2012-08-31 | View Spectrum | | Predicted MS/MS | Predicted LC-MS/MS Spectrum - 10V, Positive | splash10-000i-0940000000-42bee9785f9b55b32eea | 2016-09-12 | View Spectrum | | Predicted MS/MS | Predicted LC-MS/MS Spectrum - 20V, Positive | splash10-000i-0900000000-c8cda6a661ace060572c | 2016-09-12 | View Spectrum |
|
|---|
| NMR | | Type | Description | | View |
|---|
| 1D NMR | 1H NMR Spectrum (1D, 500 MHz, H2O, experimental) | | Spectrum | | 1D NMR | 13C NMR Spectrum (1D, 125 MHz, H2O, experimental) | | Spectrum | | 1D NMR | 1H NMR Spectrum (1D, 100 MHz, D2O, predicted) | | Spectrum | | 1D NMR | 13C NMR Spectrum (1D, 100 MHz, D2O, predicted) | | Spectrum | | 1D NMR | 1H NMR Spectrum (1D, 1000 MHz, D2O, predicted) | | Spectrum | | 1D NMR | 13C NMR Spectrum (1D, 1000 MHz, D2O, predicted) | | Spectrum | | 1D NMR | 1H NMR Spectrum (1D, 200 MHz, D2O, predicted) | | Spectrum | | 1D NMR | 13C NMR Spectrum (1D, 200 MHz, D2O, predicted) | | Spectrum | | 1D NMR | 1H NMR Spectrum (1D, 300 MHz, D2O, predicted) | | Spectrum | | 1D NMR | 13C NMR Spectrum (1D, 300 MHz, D2O, predicted) | | Spectrum | | 1D NMR | 1H NMR Spectrum (1D, 400 MHz, D2O, predicted) | | Spectrum | | 1D NMR | 13C NMR Spectrum (1D, 400 MHz, D2O, predicted) | | Spectrum | | 1D NMR | 1H NMR Spectrum (1D, 500 MHz, D2O, predicted) | | Spectrum | | 1D NMR | 13C NMR Spectrum (1D, 500 MHz, D2O, predicted) | | Spectrum | | 1D NMR | 1H NMR Spectrum (1D, 600 MHz, D2O, predicted) | | Spectrum | | 1D NMR | 13C NMR Spectrum (1D, 600 MHz, D2O, predicted) | | Spectrum | | 1D NMR | 1H NMR Spectrum (1D, 700 MHz, D2O, predicted) | | Spectrum | | 1D NMR | 13C NMR Spectrum (1D, 700 MHz, D2O, predicted) | | Spectrum | | 1D NMR | 1H NMR Spectrum (1D, 800 MHz, D2O, predicted) | | Spectrum | | 1D NMR | 13C NMR Spectrum (1D, 800 MHz, D2O, predicted) | | Spectrum | | 1D NMR | 1H NMR Spectrum (1D, 900 MHz, D2O, predicted) | | Spectrum | | 1D NMR | 13C NMR Spectrum (1D, 900 MHz, D2O, predicted) | | Spectrum | | 1D NMR | 13C NMR Spectrum (1D, 400 MHz, H2O, experimental) | | Spectrum | | 2D NMR | [1H, 1H]-TOCSY. Unexported temporarily by An Chi on Oct 15, 2021 until json or nmrML file is generated. 2D NMR Spectrum (experimental) | | Spectrum | | 2D NMR | [1H, 13C]-HSQC NMR Spectrum (2D, 400 MHz, H2O, experimental) | | Spectrum |
|
|---|
| Pathways |
|---|
| Pathways | | Name | SMPDB/Pathwhiz | KEGG | | Purine Metabolism |    |  | | Selenoamino Acid Metabolism |    | 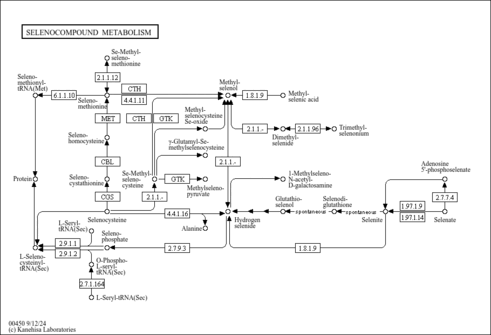 | | Adenosine Deaminase Deficiency |  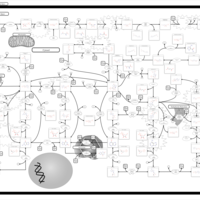 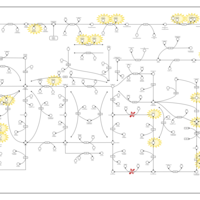 | Not Available | | Adenylosuccinate Lyase Deficiency | 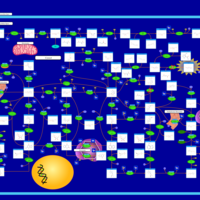 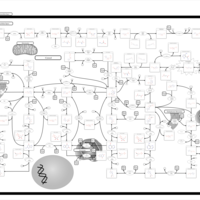 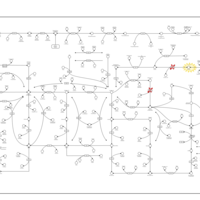 | Not Available | | Gout or Kelley-Seegmiller Syndrome | 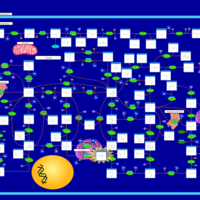 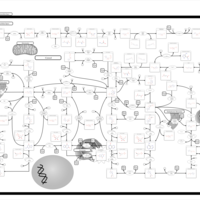 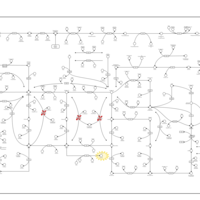 | Not Available |
|
|---|
| Protein Targets |
|---|
| Enzymes | |
|---|
| Transporters | |
|---|
| Metal Bindings | |
|---|
| Receptors | |
|---|
| Transcriptional Factors | Not Available |
|---|
| Concentrations Data |
|---|
| Not Available |
|---|
| External Links |
|---|
| HMDB ID | HMDB0000050 |
|---|
| DrugBank ID | DB00640 |
|---|
| Phenol Explorer Compound ID | Not Available |
|---|
| FoodDB ID | FDB003554 |
|---|
| KNApSAcK ID | C00007444 |
|---|
| Chemspider ID | 54923 |
|---|
| KEGG Compound ID | C00212 |
|---|
| BioCyc ID | ADENOSINE |
|---|
| BiGG ID | 34273 |
|---|
| Wikipedia Link | Adenosine |
|---|
| METLIN ID | 86 |
|---|
| PubChem Compound | 60961 |
|---|
| PDB ID | Not Available |
|---|
| ChEBI ID | 16335 |
|---|
| References |
|---|
| General References | Not Available |
|---|

