| Record Information |
|---|
| Version | 1.0 |
|---|
| Created at | 2020-04-17 18:39:18 UTC |
|---|
| Updated at | 2020-12-07 19:11:00 UTC |
|---|
| CannabisDB ID | CDB004812 |
|---|
| Secondary Accession Numbers | Not Available |
|---|
| Cannabis Compound Identification |
|---|
| Common Name | L-Tyrosine |
|---|
| Description | Tyrosine or L-tyrosine abbreviated Tyr or Y, is a non-essential, hydrophobic amino acid synthesized from phenylalanine, which is derived from food. In humans, the enzyme phenylalanine hydroxylase catalyzes the conversion of phenylalanine to tyrosine. Tyrosine is strong basic compound with an odorless taste. Tyrosine is found in highest concentrations in protein rich foods such as caseins (cheese, yogurt), milk (cow), chicken, turkey, peanuts, almonds, sesame seeds, lima beans and soy products and in lower concentrations in avocado, bananas, semolina, crayfish, and crabs. Tyrosine has also been detected in annual wild rices, lovages, corianders, lumpsuckers, and sugars making tyrosine a potential biomarker for the consumption of these foods. Tyrosine is also found in cannabis plants (PMID: 6991645 ). It is not found in high concentrations in the body as it is rapidly metabolized. Valine and other branched amino acids, and possibly tryptophan and phenylalanine may reduce tyrosine absorption. Tyrosine readily passes the blood-brain barrier where it is converted to L-dopamine (L-dopa) by tyrosine hydroxylase. L-dopa is a precursor of the catecholamines, norepinephrine and epinephrine. L-dopa is used to treat Parkinson's (PMID: 29503325 ) and tyrosine, in large doses, can be used as an adjunct in the treatment of Parkinson's. Tyrosine is also the precursor for the thyroid hormones (PMID: 29892819 ) and for the human pigment, melanin. Tyrosine is an important amino acid in many proteins, peptides and even enkephalins, the body's natural pain reliever. The hydroxyl of tyrosine can be phosphorylated by protein kinases (PMID: 10966463 ), creating phosphotyrosine. This phosphorylation is an important part of signal transduction within the cell and also alters the enzymatic activity of target proteins. There are several genetic errors of tyrosine metabolism such as hawkinsinuria and tyrosinemia I (PMID: 30740402 ) where there is an increased amount of tyrosine in the blood of premature infants. This causes decreased motor activity, lethargy, poor feeding, infection and intellectual deficits. Vitamin C supplements may reverse the disease as increasing amounts of vitamin C improved performance in chicks supplemented with excess tyrosine (PMID: 7104784 ). Some adults also develop elevated tyrosine in their blood signaling a need for more vitamin C. More tyrosine is needed under stress, and tyrosine supplements may prevent the stress-induced depletion of norepinephrine and can cure biochemical depression. However, excess, tyrosine may lead to psychosis. Indeed, many antipsychotic medications apparently function by inhibiting tyrosine metabolism. L-Tyrosine is expected to be in Cannabis as all living plants are known to produce and metabolize it. |
|---|
| Structure | |
|---|
| Synonyms | | Value | Source |
|---|
| (-)-alpha-Amino-p-hydroxyhydrocinnamic acid | ChEBI | | (2S)-2-Amino-3-(4-hydroxyphenyl)propanoic acid | ChEBI | | (S)-(-)-Tyrosine | ChEBI | | (S)-2-Amino-3-(p-hydroxyphenyl)propionic acid | ChEBI | | (S)-3-(p-Hydroxyphenyl)alanine | ChEBI | | (S)-alpha-Amino-4-hydroxybenzenepropanoic acid | ChEBI | | (S)-Tyrosine | ChEBI | | 4-Hydroxy-L-phenylalanine | ChEBI | | L-Tyrosin | ChEBI | | Tyr | ChEBI | | TYROSINE | ChEBI | | Y | ChEBI | | (-)-a-Amino-p-hydroxyhydrocinnamate | Generator | | (-)-a-Amino-p-hydroxyhydrocinnamic acid | Generator | | (-)-alpha-Amino-p-hydroxyhydrocinnamate | Generator | | (-)-Α-amino-p-hydroxyhydrocinnamate | Generator | | (-)-Α-amino-p-hydroxyhydrocinnamic acid | Generator | | (2S)-2-Amino-3-(4-hydroxyphenyl)propanoate | Generator | | (S)-2-Amino-3-(p-hydroxyphenyl)propionate | Generator | | (S)-a-Amino-4-hydroxybenzenepropanoate | Generator | | (S)-a-Amino-4-hydroxybenzenepropanoic acid | Generator | | (S)-alpha-Amino-4-hydroxybenzenepropanoate | Generator | | (S)-Α-amino-4-hydroxybenzenepropanoate | Generator | | (S)-Α-amino-4-hydroxybenzenepropanoic acid | Generator | | (S)-a-Amino-4-hydroxy-benzenepropanoate | HMDB | | (S)-a-Amino-4-hydroxy-benzenepropanoic acid | HMDB | | (S)-alpha-Amino-4-hydroxy-benzenepropanoate | HMDB | | (S)-alpha-Amino-4-hydroxy-benzenepropanoic acid | HMDB | | 2-Amino-3-(4-hydroxyphen yl)-2-amino-3-(4-hydroxyphenyl)-propanoate | HMDB | | 2-Amino-3-(4-hydroxyphen yl)-2-amino-3-(4-hydroxyphenyl)-propanoic acid | HMDB | | 3-(4-Hydroxyphenyl)-L-alanine | HMDB | | Benzenepropanoate | HMDB | | Benzenepropanoic acid | HMDB | | L-p-Tyrosine | HMDB | | p-Tyrosine | HMDB | | L Tyrosine | HMDB | | Tyrosine, L-isomer | HMDB | | Tyrosine, L isomer | HMDB | | Para tyrosine | HMDB | | Para-tyrosine | HMDB |
|
|---|
| Chemical Formula | C9H11NO3 |
|---|
| Average Molecular Weight | 181.19 |
|---|
| Monoisotopic Molecular Weight | 181.0739 |
|---|
| IUPAC Name | (2S)-2-amino-3-(4-hydroxyphenyl)propanoic acid |
|---|
| Traditional Name | L-tyrosine |
|---|
| CAS Registry Number | 140-43-2 |
|---|
| SMILES | N[C@@H](CC1=CC=C(O)C=C1)C(O)=O |
|---|
| InChI Identifier | InChI=1S/C9H11NO3/c10-8(9(12)13)5-6-1-3-7(11)4-2-6/h1-4,8,11H,5,10H2,(H,12,13)/t8-/m0/s1 |
|---|
| InChI Key | OUYCCCASQSFEME-QMMMGPOBSA-N |
|---|
| Chemical Taxonomy |
|---|
| Description | Belongs to the class of organic compounds known as tyrosine and derivatives. Tyrosine and derivatives are compounds containing tyrosine or a derivative thereof resulting from reaction of tyrosine at the amino group or the carboxy group, or from the replacement of any hydrogen of glycine by a heteroatom. |
|---|
| Kingdom | Organic compounds |
|---|
| Super Class | Organic acids and derivatives |
|---|
| Class | Carboxylic acids and derivatives |
|---|
| Sub Class | Amino acids, peptides, and analogues |
|---|
| Direct Parent | Tyrosine and derivatives |
|---|
| Alternative Parents | |
|---|
| Substituents | - Tyrosine or derivatives
- Phenylalanine or derivatives
- 3-phenylpropanoic-acid
- Alpha-amino acid
- Amphetamine or derivatives
- L-alpha-amino acid
- 1-hydroxy-2-unsubstituted benzenoid
- Phenol
- Aralkylamine
- Monocyclic benzene moiety
- Benzenoid
- Amino acid
- Carboxylic acid
- Monocarboxylic acid or derivatives
- Organic oxide
- Organooxygen compound
- Organonitrogen compound
- Amine
- Primary aliphatic amine
- Organic nitrogen compound
- Carbonyl group
- Organopnictogen compound
- Organic oxygen compound
- Hydrocarbon derivative
- Primary amine
- Aromatic homomonocyclic compound
|
|---|
| Molecular Framework | Aromatic homomonocyclic compounds |
|---|
| External Descriptors | |
|---|
| Ontology |
|---|
|
| Physiological effect | Health effect: |
|---|
| Disposition | Route of exposure: Source: Biological location: |
|---|
| Role | Biological role: Industrial application: |
|---|
| Physical Properties |
|---|
| State | Not Available |
|---|
| Experimental Properties | | Property | Value | Reference |
|---|
| Melting Point | 343 °C | Not Available | | Boiling Point | Not Available | Not Available | | Water Solubility | 0.48 mg/mL | Not Available | | logP | -2.26 | HANSCH,C ET AL. (1995) |
|
|---|
| Predicted Properties | [] |
|---|
| Spectra |
|---|
| EI-MS/GC-MS | | Type | Description | Splash Key | View |
|---|
| EI-MS | Mass Spectrum (Electron Ionization) | splash10-0a4i-3900000000-7a26097fda66f2f445b5 | 2018-05-25 | View Spectrum | | GC-MS | L-Tyrosine, 3 TMS, GC-MS Spectrum | splash10-014i-0690000000-cbbf40bb26fc84f2aead | Spectrum | | GC-MS | L-Tyrosine, 3 TMS, GC-MS Spectrum | splash10-014i-0890000000-ca45f993f95c8b0cee44 | Spectrum | | GC-MS | L-Tyrosine, 3 TMS, GC-MS Spectrum | splash10-014i-0890000000-5749069211ba15d713ef | Spectrum | | GC-MS | L-Tyrosine, 3 TMS, GC-MS Spectrum | splash10-00xr-9240000000-2c87373c0d964e0edef5 | Spectrum | | GC-MS | L-Tyrosine, non-derivatized, GC-MS Spectrum | splash10-014i-0890000000-848b2a4f247a0b3f14e8 | Spectrum | | GC-MS | L-Tyrosine, 3 TMS, GC-MS Spectrum | splash10-00xr-9450000000-6d4550940f4dde6f18ff | Spectrum | | GC-MS | L-Tyrosine, 2 TMS, GC-MS Spectrum | splash10-004i-1910000000-5cc19cad5dc24b3b9b11 | Spectrum | | GC-MS | L-Tyrosine, 3 TMS, GC-MS Spectrum | splash10-014i-1790000000-de22041357aadf60a06b | Spectrum | | GC-MS | L-Tyrosine, non-derivatized, GC-MS Spectrum | splash10-014i-0690000000-cbbf40bb26fc84f2aead | Spectrum | | GC-MS | L-Tyrosine, non-derivatized, GC-MS Spectrum | splash10-014i-0890000000-ca45f993f95c8b0cee44 | Spectrum | | GC-MS | L-Tyrosine, non-derivatized, GC-MS Spectrum | splash10-014i-0890000000-5749069211ba15d713ef | Spectrum | | GC-MS | L-Tyrosine, non-derivatized, GC-MS Spectrum | splash10-00xr-9240000000-2c87373c0d964e0edef5 | Spectrum | | GC-MS | L-Tyrosine, non-derivatized, GC-MS Spectrum | splash10-014i-0890000000-848b2a4f247a0b3f14e8 | Spectrum | | GC-MS | L-Tyrosine, non-derivatized, GC-MS Spectrum | splash10-0udi-3319000000-1d3d28a67f82366fff22 | Spectrum | | GC-MS | L-Tyrosine, non-derivatized, GC-MS Spectrum | splash10-00xr-9450000000-6d4550940f4dde6f18ff | Spectrum | | GC-MS | L-Tyrosine, non-derivatized, GC-MS Spectrum | splash10-004i-1910000000-5cc19cad5dc24b3b9b11 | Spectrum | | GC-MS | L-Tyrosine, non-derivatized, GC-MS Spectrum | splash10-014i-1790000000-de22041357aadf60a06b | Spectrum | | Predicted GC-MS | L-Tyrosine, non-derivatized, Predicted GC-MS Spectrum - 70eV, Positive | splash10-052r-4900000000-9be1412408207db5df4e | Spectrum | | Predicted GC-MS | L-Tyrosine, 2 TMS, Predicted GC-MS Spectrum - 70eV, Positive | splash10-05fr-9750000000-937b6ee7a745865ee7ee | Spectrum | | Predicted GC-MS | L-Tyrosine, non-derivatized, Predicted GC-MS Spectrum - 70eV, Positive | Not Available | Spectrum | | Predicted GC-MS | L-Tyrosine, TMS_1_1, Predicted GC-MS Spectrum - 70eV, Positive | Not Available | Spectrum | | Predicted GC-MS | L-Tyrosine, TMS_1_2, Predicted GC-MS Spectrum - 70eV, Positive | Not Available | Spectrum | | Predicted GC-MS | L-Tyrosine, TMS_1_3, Predicted GC-MS Spectrum - 70eV, Positive | Not Available | Spectrum | | Predicted GC-MS | L-Tyrosine, TMS_2_2, Predicted GC-MS Spectrum - 70eV, Positive | Not Available | Spectrum | | Predicted GC-MS | L-Tyrosine, TMS_2_3, Predicted GC-MS Spectrum - 70eV, Positive | Not Available | Spectrum |
|
|---|
| MS/MS | | Type | Description | Splash Key | View |
|---|
| MS/MS | LC-MS/MS Spectrum - Quattro_QQQ 10V, Positive (Annotated) | splash10-01p9-0900000000-580de2c16cd24559cd5c | 2012-07-24 | View Spectrum | | MS/MS | LC-MS/MS Spectrum - Quattro_QQQ 25V, Positive (Annotated) | splash10-0006-9400000000-f233fbc6c58236ee4aeb | 2012-07-24 | View Spectrum | | MS/MS | LC-MS/MS Spectrum - Quattro_QQQ 40V, Positive (Annotated) | splash10-002f-9100000000-8b5e12eba034bcfdf8a1 | 2012-07-24 | View Spectrum | | MS/MS | LC-MS/MS Spectrum - LC-ESI-ITFT (LTQ Orbitrap XL, Thermo Scientfic) , Positive | splash10-001i-0920000000-b43356ad3da227b488cb | 2012-08-31 | View Spectrum | | MS/MS | LC-MS/MS Spectrum - LC-ESI-ITFT (LTQ Orbitrap XL, Thermo Scientfic) , Positive | splash10-014i-0900000000-c04f0be6515621dda5ac | 2012-08-31 | View Spectrum | | MS/MS | LC-MS/MS Spectrum - LC-ESI-ITFT (LTQ Orbitrap XL, Thermo Scientfic) , Positive | splash10-000i-0900000000-3ed8b68bfade9194763b | 2012-08-31 | View Spectrum | | MS/MS | LC-MS/MS Spectrum - LC-ESI-ITFT (LTQ Orbitrap XL, Thermo Scientfic) , Positive | splash10-0udi-0900000000-fe6a1ce69851a8c5db00 | 2012-08-31 | View Spectrum | | MS/MS | LC-MS/MS Spectrum - LC-ESI-ITFT (LTQ Orbitrap XL, Thermo Scientfic) , Positive | splash10-001i-0900000000-aafdcea07be221817fd4 | 2012-08-31 | View Spectrum | | MS/MS | LC-MS/MS Spectrum - LC-ESI-ITFT (LTQ Orbitrap XL, Thermo Scientfic) , Positive | splash10-0002-0900000000-66a5b9a0a48bdc0a2b47 | 2012-08-31 | View Spectrum | | MS/MS | LC-MS/MS Spectrum - LC-ESI-ITFT (LTQ Orbitrap XL, Thermo Scientfic) , Positive | splash10-014i-0900000000-13eb4252ca23455a58da | 2012-08-31 | View Spectrum | | MS/MS | LC-MS/MS Spectrum - LC-ESI-ITFT (LTQ Orbitrap XL, Thermo Scientfic) , Positive | splash10-014i-0900000000-fb85798746829bac3f3d | 2012-08-31 | View Spectrum | | MS/MS | LC-MS/MS Spectrum - LC-ESI-ITFT (LTQ Orbitrap XL, Thermo Scientfic) , Negative | splash10-03e9-0839226000-5504e667281c746669ec | 2012-08-31 | View Spectrum | | MS/MS | LC-MS/MS Spectrum - LC-ESI-ITFT (LTQ Orbitrap XL, Thermo Scientfic) , Negative | splash10-03di-0900000000-f65cb3ad2fa730c922f7 | 2012-08-31 | View Spectrum | | MS/MS | LC-MS/MS Spectrum - LC-ESI-ITFT (LTQ Orbitrap XL, Thermo Scientfic) , Negative | splash10-001i-0900000000-a9276fe43ef61b4693e6 | 2012-08-31 | View Spectrum | | MS/MS | LC-MS/MS Spectrum - LC-ESI-ITFT (LTQ Orbitrap XL, Thermo Scientfic) , Negative | splash10-0a59-0039210000-21b4bd9870bf4965a6d1 | 2012-08-31 | View Spectrum | | MS/MS | LC-MS/MS Spectrum - LC-ESI-ITFT (LTQ Orbitrap XL, Thermo Scientfic) , Negative | splash10-001i-0848491200-aae99eb0b66dd1c7036d | 2012-08-31 | View Spectrum | | MS/MS | LC-MS/MS Spectrum - LC-ESI-ITFT (LTQ Orbitrap XL, Thermo Scientfic) , Negative | splash10-03di-0900000000-4104a2ca5d5ef5f22f6d | 2012-08-31 | View Spectrum | | MS/MS | LC-MS/MS Spectrum - LC-ESI-ITFT (LTQ Orbitrap XL, Thermo Scientfic) , Negative | splash10-001i-0900000000-f309996d57a95c719deb | 2012-08-31 | View Spectrum | | MS/MS | LC-MS/MS Spectrum - LC-ESI-ITFT (LTQ Orbitrap XL, Thermo Scientfic) , Negative | splash10-03di-0013090000-112cd9c2eea42dbc079b | 2012-08-31 | View Spectrum | | MS/MS | LC-MS/MS Spectrum - LC-ESI-QQ (API3000, Applied Biosystems) 10V, Negative | splash10-001i-0900000000-c7f95918d936586f633d | 2012-08-31 | View Spectrum | | MS/MS | LC-MS/MS Spectrum - LC-ESI-QQ (API3000, Applied Biosystems) 20V, Negative | splash10-03yi-1900000000-d1682546c1e0893c71e4 | 2012-08-31 | View Spectrum | | MS/MS | LC-MS/MS Spectrum - LC-ESI-QQ (API3000, Applied Biosystems) 30V, Negative | splash10-014i-2900000000-a0cc78ed35e56dd812a5 | 2012-08-31 | View Spectrum | | MS/MS | LC-MS/MS Spectrum - LC-ESI-QQ (API3000, Applied Biosystems) 40V, Negative | splash10-00kf-9500000000-d3f399f5dd10e338e25a | 2012-08-31 | View Spectrum | | MS/MS | LC-MS/MS Spectrum - LC-ESI-QQ (API3000, Applied Biosystems) 50V, Negative | splash10-0006-9200000000-8acd8d370f194bfe28ed | 2012-08-31 | View Spectrum | | MS/MS | LC-MS/MS Spectrum - LC-ESI-QQ (API3000, Applied Biosystems) 10V, Positive | splash10-001i-0900000000-6b26ce2f5b326ace12a4 | 2012-08-31 | View Spectrum |
|
|---|
| NMR | | Type | Description | | View |
|---|
| 1D NMR | 1H NMR Spectrum (1D, 600 MHz, H2O, experimental) | | Spectrum | | 1D NMR | 13C NMR Spectrum (1D, 125 MHz, H2O, experimental) | | Spectrum | | 1D NMR | 1H NMR Spectrum (1D, D2O, experimental) | | Spectrum | | 1D NMR | 1H NMR Spectrum (1D, D2O, experimental) | | Spectrum | | 1D NMR | 1H NMR Spectrum (1D, D2O, experimental) | | Spectrum | | 1D NMR | 13C NMR Spectrum (1D, D2O, experimental) | | Spectrum | | 1D NMR | 13C NMR Spectrum (1D, 100 MHz, D2O, predicted) | | Spectrum | | 1D NMR | 1H NMR Spectrum (1D, 100 MHz, D2O, predicted) | | Spectrum | | 1D NMR | 13C NMR Spectrum (1D, 200 MHz, D2O, predicted) | | Spectrum | | 1D NMR | 1H NMR Spectrum (1D, 200 MHz, D2O, predicted) | | Spectrum | | 1D NMR | 13C NMR Spectrum (1D, 300 MHz, D2O, predicted) | | Spectrum | | 1D NMR | 1H NMR Spectrum (1D, 300 MHz, D2O, predicted) | | Spectrum | | 1D NMR | 13C NMR Spectrum (1D, 400 MHz, D2O, predicted) | | Spectrum | | 1D NMR | 1H NMR Spectrum (1D, 400 MHz, D2O, predicted) | | Spectrum | | 1D NMR | 13C NMR Spectrum (1D, 500 MHz, D2O, predicted) | | Spectrum | | 1D NMR | 1H NMR Spectrum (1D, 500 MHz, D2O, predicted) | | Spectrum | | 1D NMR | 13C NMR Spectrum (1D, 600 MHz, D2O, predicted) | | Spectrum | | 1D NMR | 1H NMR Spectrum (1D, 600 MHz, D2O, predicted) | | Spectrum | | 1D NMR | 13C NMR Spectrum (1D, 700 MHz, D2O, predicted) | | Spectrum | | 1D NMR | 1H NMR Spectrum (1D, 700 MHz, D2O, predicted) | | Spectrum | | 1D NMR | 13C NMR Spectrum (1D, 800 MHz, D2O, predicted) | | Spectrum | | 1D NMR | 1H NMR Spectrum (1D, 800 MHz, D2O, predicted) | | Spectrum | | 1D NMR | 13C NMR Spectrum (1D, 900 MHz, D2O, predicted) | | Spectrum | | 1D NMR | 1H NMR Spectrum (1D, 900 MHz, D2O, predicted) | | Spectrum | | 1D NMR | 13C NMR Spectrum (1D, 1000 MHz, D2O, predicted) | | Spectrum | | 2D NMR | [1H, 1H]-TOCSY. Unexported temporarily by An Chi on Oct 15, 2021 until json or nmrML file is generated. 2D NMR Spectrum (experimental) | | Spectrum | | 2D NMR | [1H, 13C]-HSQC NMR Spectrum (2D, 400 MHz, H2O, experimental) | | Spectrum |
|
|---|
| Pathways |
|---|
| Pathways | | Name | SMPDB/Pathwhiz | KEGG | | Tyrosine Metabolism | 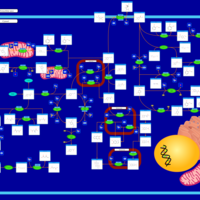 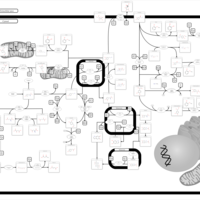 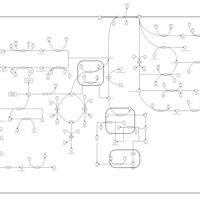 |  | | Transcription/Translation | Not Available | Not Available | | Phenylalanine and Tyrosine Metabolism | 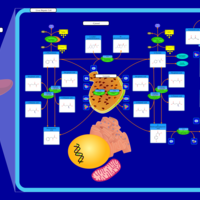 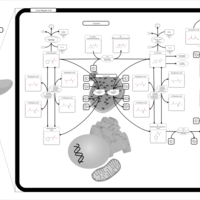 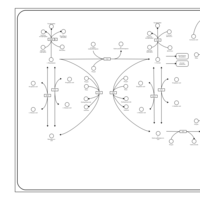 | 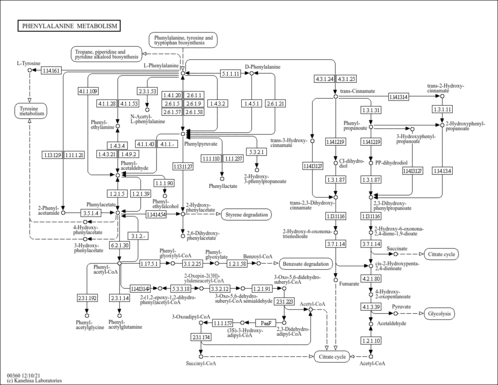 | | Catecholamine Biosynthesis | 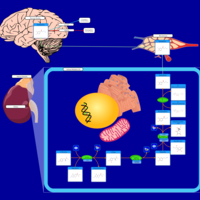   |  | | Aromatic L-Aminoacid Decarboxylase Deficiency |    | Not Available |
|
|---|
| Protein Targets |
|---|
| Enzymes | |
|---|
| Transporters | |
|---|
| Metal Bindings | |
|---|
| Receptors | |
|---|
| Transcriptional Factors | Not Available |
|---|
| Concentrations Data |
|---|
| Not Available |
|---|
| External Links |
|---|
| HMDB ID | HMDB0000158 |
|---|
| DrugBank ID | DB00135 |
|---|
| Phenol Explorer Compound ID | Not Available |
|---|
| FoodDB ID | FDB000446 |
|---|
| KNApSAcK ID | C00001397 |
|---|
| Chemspider ID | 5833 |
|---|
| KEGG Compound ID | C00082 |
|---|
| BioCyc ID | TYR |
|---|
| BiGG ID | 33785 |
|---|
| Wikipedia Link | Tyrosine |
|---|
| METLIN ID | 34 |
|---|
| PubChem Compound | 6057 |
|---|
| PDB ID | Not Available |
|---|
| ChEBI ID | 17895 |
|---|
| References |
|---|
| General References | - Turner CE, Elsohly MA, Boeren EG: Constituents of Cannabis sativa L. XVII. A review of the natural constituents. J Nat Prod. 1980 Mar-Apr;43(2):169-234. doi: 10.1021/np50008a001. [PubMed:6991645 ]
- Radhakrishnan DM, Goyal V: Parkinson's disease: A review. Neurol India. 2018 Mar-Apr;66(Supplement):S26-S35. doi: 10.4103/0028-3886.226451. [PubMed:29503325 ]
- Kohrle J: Thyroid Hormones and Derivatives: Endogenous Thyroid Hormones and Their Targets. Methods Mol Biol. 2018;1801:85-104. doi: 10.1007/978-1-4939-7902-8_9. [PubMed:29892819 ]
- Hubbard SR, Till JH: Protein tyrosine kinase structure and function. Annu Rev Biochem. 2000;69:373-98. doi: 10.1146/annurev.biochem.69.1.373. [PubMed:10966463 ]
- Aliu E, Kanungo S, Arnold GL: Amino acid disorders. Ann Transl Med. 2018 Dec;6(24):471. doi: 10.21037/atm.2018.12.12. [PubMed:30740402 ]
- Yanaka M, Okumura J: Influence of ascorbic acid on the adverse effect of feeding a diet containing excess tyrosine to chicks. Br Poult Sci. 1982 May;23(3):239-45. doi: 10.1080/00071688208447952. [PubMed:7104784 ]
|
|---|

