| Record Information |
|---|
| Version | 1.0 |
|---|
| Created at | 2020-03-19 00:51:01 UTC |
|---|
| Updated at | 2020-11-18 16:35:25 UTC |
|---|
| CannabisDB ID | CDB000683 |
|---|
| Secondary Accession Numbers | Not Available |
|---|
| Cannabis Compound Identification |
|---|
| Common Name | 1-Propanol |
|---|
| Description | Propanol, 1-propanol or propyl alcohol, also known as ethylcarbinol, belongs to the class of organic compounds known as primary alcohols. Primary alcohols are compounds comprising the primary alcohol functional group, with the general structure RCOH (R=alkyl, aryl). Propyl alcohol is a colorless liquid that has a mild, alcohol-like odor. It is miscible in water. 1-propanol is an isomer of 2-propanol. Propyl alcohol exists in all eukaryotes, ranging from yeast to plants to humans. It is formed naturally in small amounts during many fermentation processes. For instance, 1-propanol is a major constituent of fusel oil, a by-product formed from certain amino acids when potatoes or grains are fermented to produce ethanol. Propyl alcohol can be found in small amounts in a number of food items such as cashew nuts, chinese mustard, greenthread tea, and chayote. In humans, propyl alcohol can be found primarily in blood, feces, and saliva, as well as in human fibroblasts tissue. Industrially propyl alcohol is used as a solvent in the pharmaceutical industry, mainly for resins and cellulose esters. 1-propanol exhibits low acute toxicity via dermal, inhalation, and oral routes of exposure; it is not very irritating to the skin and dermal absorption is expected to be slow. 1-propanol is readily metabolized by alcohol dehydrogenase to propionic acid, and has no evidence of carcinogenicity or mutagenicity. 1-propanol has a high octane number and it is suitable for engine fuel usage. 1-propanol has also been reported to be found in Cannabis oils (PMID: 6991645 ). |
|---|
| Structure | |
|---|
| Synonyms | | Value | Source |
|---|
| 1-Hydroxypropane | ChEBI | | 1-Propanol | ChEBI | | Ethyl carbinol | ChEBI | | Ethylcarbinol | ChEBI | | N-Propan-1-ol | ChEBI | | N-Propanol | ChEBI | | N-Propyl alcohol | ChEBI | | N-Propylalkohol | ChEBI | | Optal | ChEBI | | Osmosol extra | ChEBI | | Propane-1-ol | ChEBI | | Propanol | ChEBI | | Propanol-1 | ChEBI | | UN 1274 | ChEBI | | Propan-1-ol | Kegg | | 1-Propyl alcohol | HMDB | | Hydroxypropane | HMDB | | Propanole | HMDB | | Propanolen | HMDB | | Propylan-propyl alcohol | HMDB | | Propylic alcohol | HMDB | | 1 Propanol | HMDB | | Alcohol, propyl | HMDB | | N Propanol | HMDB | | Propyl alcohol | ChEBI |
|
|---|
| Chemical Formula | C3H8O |
|---|
| Average Molecular Weight | 60.1 |
|---|
| Monoisotopic Molecular Weight | 60.0575 |
|---|
| IUPAC Name | propan-1-ol |
|---|
| Traditional Name | propanol |
|---|
| CAS Registry Number | 71-23-8 |
|---|
| SMILES | CCCO |
|---|
| InChI Identifier | InChI=1S/C3H8O/c1-2-3-4/h4H,2-3H2,1H3 |
|---|
| InChI Key | BDERNNFJNOPAEC-UHFFFAOYSA-N |
|---|
| Chemical Taxonomy |
|---|
| Description | Belongs to the class of organic compounds known as primary alcohols. Primary alcohols are compounds comprising the primary alcohol functional group, with the general structure RCOH (R=alkyl, aryl). |
|---|
| Kingdom | Organic compounds |
|---|
| Super Class | Organic oxygen compounds |
|---|
| Class | Organooxygen compounds |
|---|
| Sub Class | Alcohols and polyols |
|---|
| Direct Parent | Primary alcohols |
|---|
| Alternative Parents | |
|---|
| Substituents | - Hydrocarbon derivative
- Primary alcohol
- Aliphatic acyclic compound
|
|---|
| Molecular Framework | Aliphatic acyclic compounds |
|---|
| External Descriptors | |
|---|
| Ontology |
|---|
|
| Disposition | Route of exposure: Source: Biological location: |
|---|
| Role | Industrial application: |
|---|
| Physical Properties |
|---|
| State | Solid |
|---|
| Experimental Properties | | Property | Value | Reference |
|---|
| Melting Point | -126.1 °C | Not Available | | Boiling Point | Not Available | Not Available | | Water Solubility | 1000 mg/mL | Not Available | | logP | 0.25 | HANSCH,C ET AL. (1995) |
|
|---|
| Predicted Properties | [] |
|---|
| Spectra |
|---|
| EI-MS/GC-MS | | Type | Description | Splash Key | View |
|---|
| EI-MS | Mass Spectrum (Electron Ionization) | splash10-001i-9000000000-cdf3b05a292d7d658a6f | 2015-03-01 | View Spectrum | | GC-MS | 1-Propanol , non-derivatized, GC-MS Spectrum | splash10-001i-9000000000-b16d56b3c4fc95e00c56 | Spectrum | | GC-MS | 1-Propanol , non-derivatized, GC-MS Spectrum | splash10-001i-9000000000-425940a0b79c32c50c90 | Spectrum | | GC-MS | 1-Propanol , non-derivatized, GC-MS Spectrum | splash10-001i-9000000000-6013791cdd19a27be85c | Spectrum | | GC-MS | 1-Propanol , non-derivatized, GC-MS Spectrum | splash10-001i-9000000000-b16d56b3c4fc95e00c56 | Spectrum | | GC-MS | 1-Propanol , non-derivatized, GC-MS Spectrum | splash10-001i-9000000000-425940a0b79c32c50c90 | Spectrum | | GC-MS | 1-Propanol , non-derivatized, GC-MS Spectrum | splash10-001i-9000000000-6013791cdd19a27be85c | Spectrum | | Predicted GC-MS | 1-Propanol , non-derivatized, Predicted GC-MS Spectrum - 70eV, Positive | splash10-01u3-9000000000-a356816d910101b714f8 | Spectrum | | Predicted GC-MS | 1-Propanol , 1 TMS, Predicted GC-MS Spectrum - 70eV, Positive | splash10-0100-9300000000-b1bb388b3a8da1a92913 | Spectrum | | Predicted GC-MS | 1-Propanol , non-derivatized, Predicted GC-MS Spectrum - 70eV, Positive | Not Available | Spectrum | | Predicted GC-MS | 1-Propanol , non-derivatized, Predicted GC-MS Spectrum - 70eV, Positive | Not Available | Spectrum | | Predicted GC-MS | 1-Propanol , TBDMS_1_1, Predicted GC-MS Spectrum - 70eV, Positive | Not Available | Spectrum |
|
|---|
| MS/MS | | Type | Description | Splash Key | View |
|---|
| MS/MS | LC-MS/MS Spectrum - Quattro_QQQ 10V, Positive (Annotated) | splash10-03di-9000000000-8e60a750ac016bf5f9f7 | 2012-07-24 | View Spectrum | | MS/MS | LC-MS/MS Spectrum - Quattro_QQQ 25V, Positive (Annotated) | splash10-03dl-9000000000-17658e49fed270452ed4 | 2012-07-24 | View Spectrum | | MS/MS | LC-MS/MS Spectrum - Quattro_QQQ 40V, Positive (Annotated) | splash10-03dl-9000000000-10c2ef6f2c375973e1a9 | 2012-07-24 | View Spectrum | | MS/MS | LC-MS/MS Spectrum - EI-B (HITACHI RMU-6M) , Positive | splash10-001i-9000000000-b16d56b3c4fc95e00c56 | 2012-08-31 | View Spectrum | | MS/MS | LC-MS/MS Spectrum - EI-B (HITACHI M-80B) , Positive | splash10-001i-9000000000-16b5a3548bcbe12e38bb | 2012-08-31 | View Spectrum | | MS/MS | LC-MS/MS Spectrum - EI-B (HITACHI RMU-7M) , Positive | splash10-001i-9000000000-6013791cdd19a27be85c | 2012-08-31 | View Spectrum | | Predicted MS/MS | Predicted LC-MS/MS Spectrum - 10V, Positive | splash10-01ox-9000000000-06ef5e8e7805851d0737 | 2015-05-27 | View Spectrum | | Predicted MS/MS | Predicted LC-MS/MS Spectrum - 20V, Positive | splash10-0006-9000000000-03159cae015c86b655e0 | 2015-05-27 | View Spectrum | | Predicted MS/MS | Predicted LC-MS/MS Spectrum - 40V, Positive | splash10-0006-9000000000-db45d3e7d2ae1653f76e | 2015-05-27 | View Spectrum | | Predicted MS/MS | Predicted LC-MS/MS Spectrum - 10V, Negative | splash10-0a4i-9000000000-9517aa42fced76eff1d2 | 2015-05-27 | View Spectrum | | Predicted MS/MS | Predicted LC-MS/MS Spectrum - 20V, Negative | splash10-0a4i-9000000000-f78c10cac4b60ad41988 | 2015-05-27 | View Spectrum | | Predicted MS/MS | Predicted LC-MS/MS Spectrum - 40V, Negative | splash10-0006-9000000000-fb6c8a2550ff427023d8 | 2015-05-27 | View Spectrum | | Predicted MS/MS | Predicted LC-MS/MS Spectrum - 10V, Negative | splash10-0a4i-9000000000-28b1a4f33a69b25b3b12 | 2021-09-23 | View Spectrum | | Predicted MS/MS | Predicted LC-MS/MS Spectrum - 20V, Negative | splash10-0a4i-9000000000-f9165cc110614db9d933 | 2021-09-23 | View Spectrum | | Predicted MS/MS | Predicted LC-MS/MS Spectrum - 40V, Negative | splash10-052f-9000000000-979f46be2c381869e59a | 2021-09-23 | View Spectrum | | Predicted MS/MS | Predicted LC-MS/MS Spectrum - 10V, Positive | splash10-0006-9000000000-5381c9dfee7dd1735ffd | 2021-09-24 | View Spectrum | | Predicted MS/MS | Predicted LC-MS/MS Spectrum - 20V, Positive | splash10-0006-9000000000-5381c9dfee7dd1735ffd | 2021-09-24 | View Spectrum | | Predicted MS/MS | Predicted LC-MS/MS Spectrum - 40V, Positive | splash10-0006-9000000000-2e1a81e7a0d31ddc562c | 2021-09-24 | View Spectrum |
|
|---|
| NMR | | Type | Description | | View |
|---|
| 1D NMR | 1H NMR Spectrum (1D, 500 MHz, H2O, experimental) | | Spectrum | | 1D NMR | 1H NMR Spectrum (1D, 90 MHz, CDCl3, experimental) | | Spectrum | | 1D NMR | 13C NMR Spectrum (1D, 15.09 MHz, CDCl3, experimental) | | Spectrum | | 1D NMR | 1H NMR Spectrum (1D, D2O, experimental) | | Spectrum | | 1D NMR | 13C NMR Spectrum (1D, D2O, experimental) | | Spectrum | | 2D NMR | [1H, 13C]-HSQC NMR Spectrum (2D, 500 MHz, H2O, experimental) | | Spectrum |
|
|---|
| Pathways |
|---|
| Pathways | | Name | SMPDB/Pathwhiz | KEGG | | Sulfate/Sulfite Metabolism | 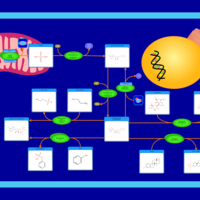   | 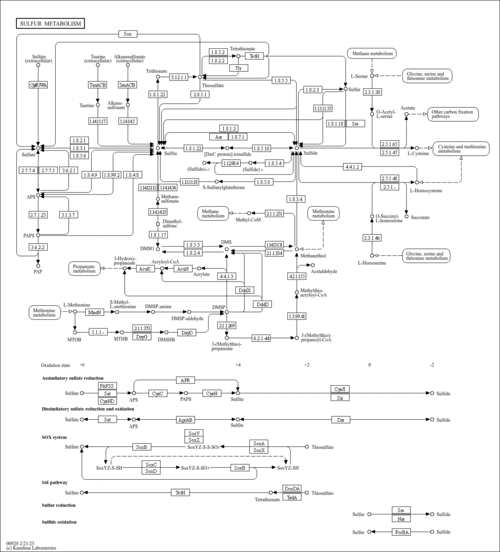 | | Sulfite oxidase deficiency | 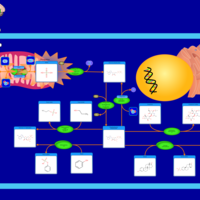   | Not Available |
|
|---|
| Protein Targets |
|---|
| Enzymes | |
|---|
| Transporters | |
|---|
| Metal Bindings | |
|---|
| Receptors | |
|---|
| Transcriptional Factors | |
| Nuclear receptor subfamily 1 group I member 3 | NR1I3 | 1q23.3 | Q14994 | details |
|
|---|
| Concentrations Data |
|---|
| Not Available |
|---|
| External Links |
|---|
| HMDB ID | HMDB0000820 |
|---|
| DrugBank ID | DB03175 |
|---|
| Phenol Explorer Compound ID | Not Available |
|---|
| FoodDB ID | FDB031044 |
|---|
| KNApSAcK ID | Not Available |
|---|
| Chemspider ID | 1004 |
|---|
| KEGG Compound ID | C05979 |
|---|
| BioCyc ID | PROPANOL |
|---|
| BiGG ID | Not Available |
|---|
| Wikipedia Link | Propan-1-ol |
|---|
| METLIN ID | 5783 |
|---|
| PubChem Compound | 1031 |
|---|
| PDB ID | Not Available |
|---|
| ChEBI ID | 28831 |
|---|
| References |
|---|
| General References | - Turner CE, Elsohly MA, Boeren EG: Constituents of Cannabis sativa L. XVII. A review of the natural constituents. J Nat Prod. 1980 Mar-Apr;43(2):169-234. doi: 10.1021/np50008a001. [PubMed:6991645 ]
|
|---|
