| Record Information |
|---|
| Version | 1.0 |
|---|
| Created at | 2020-04-17 18:36:19 UTC |
|---|
| Updated at | 2020-12-07 19:10:55 UTC |
|---|
| CannabisDB ID | CDB004783 |
|---|
| Secondary Accession Numbers | Not Available |
|---|
| Cannabis Compound Identification |
|---|
| Common Name | alpha-Ketoisovaleric acid |
|---|
| Description | alpha-Ketoisovaleric acid, also known as a-keto-isovalerate or 3-methyl-2-oxobutanoate, belongs to the class of organic compounds known as short-chain keto acids and derivatives. These are keto acids with an alkyl chain the contains less than 6 carbon atoms. alpha-Ketoisovaleric acid is a very hydrophobic molecule, practically insoluble (in water), and relatively neutral. alpha-Ketoisovaleric acid exists in all living species, ranging from bacteria to humans. alpha-Ketoisovaleric acid is a fruity tasting compound. Outside of the human body, alpha-Ketoisovaleric acid has been detected, but not quantified in, several different foods, such as root vegetables, jerusalem artichokes, rapinis, chia, and cowpea. This could make alpha-ketoisovaleric acid a potential biomarker for the consumption of these foods. alpha-Ketoisovaleric acid is a potentially toxic compound. alpha-Ketoisovaleric acid, with regard to humans, has been found to be associated with the diseases such as colorectal cancer; alpha-ketoisovaleric acid has also been linked to the inborn metabolic disorder maple syrup urine disease. A 2-oxo monocarboxylic acid that is the 2-oxo derivative of isovaleric acid. alpha-Ketoisovaleric acid is expected to be in Cannabis as all living plants are known to produce and metabolize it. |
|---|
| Structure | |
|---|
| Synonyms | | Value | Source |
|---|
| 2-Keto-3-methylbutyric acid | ChEBI | | 2-Ketoisovaleric acid | ChEBI | | 2-Ketovaline | ChEBI | | 2-oxo-3-Methylbutanoic acid | ChEBI | | 2-oxo-3-Methylbutyric acid | ChEBI | | 2-Oxoisovaleric acid | ChEBI | | 3-Methyl-2-oxobutanoate | ChEBI | | 3-Methyl-2-oxobutyric acid | ChEBI | | alpha-Keto-isovaleric acid | ChEBI | | alpha-Ketovaline | ChEBI | | alpha-oxo-beta-Methylbutyricacid | ChEBI | | alpha-Oxoisovaleric acid | ChEBI | | Dimethylpyruvic acid | ChEBI | | Isopropylglyoxylic acid | ChEBI | | 2-oxo-3-Methylbutanoate | Kegg | | 2-Oxoisovalerate | Kegg | | 2-Oxoisopentanoate | Kegg | | 2-Keto-3-methylbutyrate | Generator | | 2-Ketoisovalerate | Generator | | 2-oxo-3-Methylbutyrate | Generator | | 3-Methyl-2-oxobutanoic acid | Generator | | 3-Methyl-2-oxobutyrate | Generator | | a-Keto-isovalerate | Generator | | a-Keto-isovaleric acid | Generator | | alpha-Keto-isovalerate | Generator | | Α-keto-isovalerate | Generator | | Α-keto-isovaleric acid | Generator | | a-Ketovaline | Generator | | Α-ketovaline | Generator | | a-oxo-b-Methylbutyricacid | Generator | | Α-oxo-β-methylbutyricacid | Generator | | a-Oxoisovalerate | Generator | | a-Oxoisovaleric acid | Generator | | alpha-Oxoisovalerate | Generator | | Α-oxoisovalerate | Generator | | Α-oxoisovaleric acid | Generator | | Dimethylpyruvate | Generator | | Isopropylglyoxylate | Generator | | 2-Oxoisopentanoic acid | Generator | | a-Ketoisovalerate | Generator | | a-Ketoisovaleric acid | Generator | | alpha-Ketoisovalerate | Generator | | Α-ketoisovalerate | Generator | | Α-ketoisovaleric acid | Generator | | 2-Ketoisvaleric acid | HMDB | | 2-oxo-3-Methyl-butyrate | HMDB | | 3-Methyl-2-oxo-butanoate | HMDB | | 3-Methyl-2-oxo-butanoic acid | HMDB | | 3-Methyl-2-oxo-butyrate | HMDB | | 3-Methyl-2-oxo-butyric acid | HMDB | | 3-Methyl-2-oxobutinoate | HMDB | | 3-Methyl-2-oxobutinoic acid | HMDB | | a-Keto-b-methylbutyrate | HMDB | | a-Keto-b-methylbutyric acid | HMDB | | a-oxo-b-Methylbutyrate | HMDB | | a-oxo-b-Methylbutyric acid | HMDB | | alpha-Keto-beta-methylbutyrate | HMDB | | alpha-Keto-beta-methylbutyric acid | HMDB | | alpha-oxo-beta-Methylbutyrate | HMDB | | alpha-oxo-beta-Methylbutyric acid | HMDB | | Ketovaline | HMDB | | alpha-Ketoisopentanoic acid | HMDB | | alpha-Ketoisovaleric acid | Generator, MeSH |
|
|---|
| Chemical Formula | C5H8O3 |
|---|
| Average Molecular Weight | 116.12 |
|---|
| Monoisotopic Molecular Weight | 116.0473 |
|---|
| IUPAC Name | 3-methyl-2-oxobutanoic acid |
|---|
| Traditional Name | α-ketoisovalerate |
|---|
| CAS Registry Number | 759-05-7 |
|---|
| SMILES | CC(C)C(=O)C(O)=O |
|---|
| InChI Identifier | InChI=1S/C5H8O3/c1-3(2)4(6)5(7)8/h3H,1-2H3,(H,7,8) |
|---|
| InChI Key | QHKABHOOEWYVLI-UHFFFAOYSA-N |
|---|
| Chemical Taxonomy |
|---|
| Description | Belongs to the class of organic compounds known as short-chain keto acids and derivatives. These are keto acids with an alkyl chain the contains less than 6 carbon atoms. |
|---|
| Kingdom | Organic compounds |
|---|
| Super Class | Organic acids and derivatives |
|---|
| Class | Keto acids and derivatives |
|---|
| Sub Class | Short-chain keto acids and derivatives |
|---|
| Direct Parent | Short-chain keto acids and derivatives |
|---|
| Alternative Parents | |
|---|
| Substituents | - Branched fatty acid
- Methyl-branched fatty acid
- Short-chain keto acid
- Alpha-keto acid
- Fatty acyl
- Alpha-hydroxy ketone
- Ketone
- Carboxylic acid derivative
- Carboxylic acid
- Monocarboxylic acid or derivatives
- Carbonyl group
- Hydrocarbon derivative
- Organic oxygen compound
- Organooxygen compound
- Organic oxide
- Aliphatic acyclic compound
|
|---|
| Molecular Framework | Aliphatic acyclic compounds |
|---|
| External Descriptors | |
|---|
| Ontology |
|---|
|
| Disposition | Route of exposure: Source: Biological location: |
|---|
| Role | Indirect biological role: Biological role: Industrial application: |
|---|
| Physical Properties |
|---|
| State | Solid |
|---|
| Experimental Properties | | Property | Value | Reference |
|---|
| Melting Point | 31.5 °C | Not Available | | Boiling Point | Not Available | Not Available | | Water Solubility | Not Available | Not Available | | logP | Not Available | Not Available |
|
|---|
| Predicted Properties | [] |
|---|
| Spectra |
|---|
| EI-MS/GC-MS | | Type | Description | Splash Key | View |
|---|
| GC-MS | alpha-Ketoisovaleric acid, non-derivatized, GC-MS Spectrum | splash10-000i-9500000000-ff936b879a69b5d118f8 | Spectrum | | GC-MS | alpha-Ketoisovaleric acid, non-derivatized, GC-MS Spectrum | splash10-000i-8920000000-e37b37d64d43dcf763f0 | Spectrum | | GC-MS | alpha-Ketoisovaleric acid, 1 MEOX; 1 TMS, GC-MS Spectrum | splash10-000i-9400000000-e3995acc4818a98d0f48 | Spectrum | | GC-MS | alpha-Ketoisovaleric acid, 1 MEOX; 1 TMS, GC-MS Spectrum | splash10-0f79-9720000000-5d89487273e44ea61a68 | Spectrum | | GC-MS | alpha-Ketoisovaleric acid, non-derivatized, GC-MS Spectrum | splash10-000i-9700000000-209c737dcac7df2b198c | Spectrum | | GC-MS | alpha-Ketoisovaleric acid, non-derivatized, GC-MS Spectrum | splash10-0f79-5920000000-759a2f01b4f52767ade2 | Spectrum | | GC-MS | alpha-Ketoisovaleric acid, non-derivatized, GC-MS Spectrum | splash10-000i-9500000000-ff936b879a69b5d118f8 | Spectrum | | GC-MS | alpha-Ketoisovaleric acid, non-derivatized, GC-MS Spectrum | splash10-000i-8920000000-e37b37d64d43dcf763f0 | Spectrum | | GC-MS | alpha-Ketoisovaleric acid, non-derivatized, GC-MS Spectrum | splash10-000i-9400000000-e3995acc4818a98d0f48 | Spectrum | | GC-MS | alpha-Ketoisovaleric acid, non-derivatized, GC-MS Spectrum | splash10-0f79-9720000000-5d89487273e44ea61a68 | Spectrum | | GC-MS | alpha-Ketoisovaleric acid, non-derivatized, GC-MS Spectrum | splash10-000i-7900000000-7e325567183fb56996d5 | Spectrum | | GC-MS | alpha-Ketoisovaleric acid, non-derivatized, GC-MS Spectrum | splash10-0ufr-0930000000-be90b06add7135b4a539 | Spectrum | | Predicted GC-MS | alpha-Ketoisovaleric acid, non-derivatized, Predicted GC-MS Spectrum - 70eV, Positive | splash10-00dl-9000000000-52f9d8fc8386e4c512fe | Spectrum | | Predicted GC-MS | alpha-Ketoisovaleric acid, 1 TMS, Predicted GC-MS Spectrum - 70eV, Positive | splash10-006x-9600000000-52aefdc997dac92c8459 | Spectrum | | Predicted GC-MS | alpha-Ketoisovaleric acid, non-derivatized, Predicted GC-MS Spectrum - 70eV, Positive | Not Available | Spectrum | | Predicted GC-MS | alpha-Ketoisovaleric acid, non-derivatized, Predicted GC-MS Spectrum - 70eV, Positive | Not Available | Spectrum | | Predicted GC-MS | alpha-Ketoisovaleric acid, TMS_1_2, Predicted GC-MS Spectrum - 70eV, Positive | Not Available | Spectrum | | Predicted GC-MS | alpha-Ketoisovaleric acid, TBDMS_1_1, Predicted GC-MS Spectrum - 70eV, Positive | Not Available | Spectrum | | Predicted GC-MS | alpha-Ketoisovaleric acid, TBDMS_1_2, Predicted GC-MS Spectrum - 70eV, Positive | Not Available | Spectrum |
|
|---|
| MS/MS | | Type | Description | Splash Key | View |
|---|
| MS/MS | LC-MS/MS Spectrum - Quattro_QQQ 10V, Positive (Annotated) | splash10-000i-9000000000-10ab58a33e9ca7dbace0 | 2012-07-24 | View Spectrum | | MS/MS | LC-MS/MS Spectrum - Quattro_QQQ 25V, Positive (Annotated) | splash10-000i-9000000000-ad51ff01c94b6046ad64 | 2012-07-24 | View Spectrum | | MS/MS | LC-MS/MS Spectrum - LC-ESI-QQ (API3000, Applied Biosystems) 10V, Negative | splash10-014i-0900000000-9993174a7b1801b90ddb | 2012-08-31 | View Spectrum | | MS/MS | LC-MS/MS Spectrum - LC-ESI-QQ (API3000, Applied Biosystems) 20V, Negative | splash10-00xr-9500000000-293818b81e0879b6feb2 | 2012-08-31 | View Spectrum | | MS/MS | LC-MS/MS Spectrum - LC-ESI-QQ (API3000, Applied Biosystems) 30V, Negative | splash10-00di-9000000000-75058f27a2178b9cf121 | 2012-08-31 | View Spectrum | | MS/MS | LC-MS/MS Spectrum - LC-ESI-QQ (API3000, Applied Biosystems) 40V, Negative | splash10-0006-9000000000-4c20af39e8ee009d5278 | 2012-08-31 | View Spectrum | | MS/MS | LC-MS/MS Spectrum - LC-ESI-QQ , negative | splash10-014i-0900000000-9993174a7b1801b90ddb | 2017-09-14 | View Spectrum | | MS/MS | LC-MS/MS Spectrum - LC-ESI-QQ , negative | splash10-00xr-9500000000-1a58c6a6b4f5477dabdd | 2017-09-14 | View Spectrum | | MS/MS | LC-MS/MS Spectrum - LC-ESI-QQ , negative | splash10-00di-9000000000-75058f27a2178b9cf121 | 2017-09-14 | View Spectrum | | MS/MS | LC-MS/MS Spectrum - LC-ESI-QQ , negative | splash10-0006-9000000000-cc4465a47e663be66df5 | 2017-09-14 | View Spectrum | | MS/MS | LC-MS/MS Spectrum - LC-ESI-IT , negative | splash10-00di-9100000000-19511890852fce513a02 | 2017-09-14 | View Spectrum | | MS/MS | LC-MS/MS Spectrum - 20V, Positive | splash10-00kf-9300000000-30d3bdfb6d38dbb8e101 | 2021-09-20 | View Spectrum | | MS/MS | LC-MS/MS Spectrum - 40V, Positive | splash10-000l-9000000000-879b6502ff380673b776 | 2021-09-20 | View Spectrum | | MS/MS | LC-MS/MS Spectrum - 10V, Positive | splash10-0159-9800000000-0cf4d3a0e7b3356995c6 | 2021-09-20 | View Spectrum | | MS/MS | LC-MS/MS Spectrum - 35V, Positive | splash10-014o-9900000000-f35430a2eaea0d81df32 | 2021-09-20 | View Spectrum | | Predicted MS/MS | Predicted LC-MS/MS Spectrum - 10V, Positive | splash10-00r2-9400000000-21f8c3fae79161c82099 | 2015-04-24 | View Spectrum | | Predicted MS/MS | Predicted LC-MS/MS Spectrum - 20V, Positive | splash10-00dl-9000000000-5f25e41413738bb6b5d7 | 2015-04-24 | View Spectrum | | Predicted MS/MS | Predicted LC-MS/MS Spectrum - 40V, Positive | splash10-0006-9000000000-801af00dea93fcfd637d | 2015-04-24 | View Spectrum | | Predicted MS/MS | Predicted LC-MS/MS Spectrum - 10V, Negative | splash10-01b9-8900000000-5185c7dfc72c25069904 | 2015-04-25 | View Spectrum | | Predicted MS/MS | Predicted LC-MS/MS Spectrum - 20V, Negative | splash10-00xs-9200000000-7e2a275a65197f96f5d7 | 2015-04-25 | View Spectrum | | Predicted MS/MS | Predicted LC-MS/MS Spectrum - 40V, Negative | splash10-0600-9000000000-0013e0ff06f9896a1337 | 2015-04-25 | View Spectrum | | Predicted MS/MS | Predicted LC-MS/MS Spectrum - 10V, Negative | splash10-01b9-6900000000-7f00871b074cbd9a42e6 | 2021-09-25 | View Spectrum | | Predicted MS/MS | Predicted LC-MS/MS Spectrum - 20V, Negative | splash10-06di-9400000000-6d56778068e11e4f8e85 | 2021-09-25 | View Spectrum | | Predicted MS/MS | Predicted LC-MS/MS Spectrum - 40V, Negative | splash10-0a4i-9000000000-b445e7fc67a8ff2bd3ba | 2021-09-25 | View Spectrum | | Predicted MS/MS | Predicted LC-MS/MS Spectrum - 10V, Positive | splash10-006x-9000000000-733fb7227d2d053b112f | 2021-09-25 | View Spectrum |
|
|---|
| NMR | | Type | Description | | View |
|---|
| 1D NMR | 1H NMR Spectrum (1D, 500 MHz, H2O, experimental) | | Spectrum | | 1D NMR | 1H NMR Spectrum (1D, D2O, experimental) | | Spectrum | | 1D NMR | 13C NMR Spectrum (1D, D2O, experimental) | | Spectrum | | 1D NMR | 1H NMR Spectrum (1D, 100 MHz, D2O, predicted) | | Spectrum | | 1D NMR | 13C NMR Spectrum (1D, 100 MHz, D2O, predicted) | | Spectrum | | 1D NMR | 1H NMR Spectrum (1D, 200 MHz, D2O, predicted) | | Spectrum | | 1D NMR | 13C NMR Spectrum (1D, 200 MHz, D2O, predicted) | | Spectrum | | 1D NMR | 1H NMR Spectrum (1D, 300 MHz, D2O, predicted) | | Spectrum | | 1D NMR | 13C NMR Spectrum (1D, 300 MHz, D2O, predicted) | | Spectrum | | 1D NMR | 1H NMR Spectrum (1D, 400 MHz, D2O, predicted) | | Spectrum | | 1D NMR | 13C NMR Spectrum (1D, 400 MHz, D2O, predicted) | | Spectrum | | 1D NMR | 1H NMR Spectrum (1D, 500 MHz, D2O, predicted) | | Spectrum | | 1D NMR | 13C NMR Spectrum (1D, 500 MHz, D2O, predicted) | | Spectrum | | 1D NMR | 1H NMR Spectrum (1D, 600 MHz, D2O, predicted) | | Spectrum | | 1D NMR | 13C NMR Spectrum (1D, 600 MHz, D2O, predicted) | | Spectrum | | 1D NMR | 1H NMR Spectrum (1D, 700 MHz, D2O, predicted) | | Spectrum | | 1D NMR | 13C NMR Spectrum (1D, 700 MHz, D2O, predicted) | | Spectrum | | 1D NMR | 1H NMR Spectrum (1D, 800 MHz, D2O, predicted) | | Spectrum | | 1D NMR | 13C NMR Spectrum (1D, 800 MHz, D2O, predicted) | | Spectrum | | 1D NMR | 1H NMR Spectrum (1D, 900 MHz, D2O, predicted) | | Spectrum | | 1D NMR | 13C NMR Spectrum (1D, 900 MHz, D2O, predicted) | | Spectrum | | 1D NMR | 1H NMR Spectrum (1D, 1000 MHz, D2O, predicted) | | Spectrum | | 1D NMR | 13C NMR Spectrum (1D, 1000 MHz, D2O, predicted) | | Spectrum | | 1D NMR | 1H NMR Spectrum (1D, 500 MHz, H2O, experimental) | | Spectrum | | 2D NMR | [1H, 13C]-HSQC NMR Spectrum (2D, 600 MHz, H2O, experimental) | | Spectrum | | 2D NMR | [1H, 1H]-TOCSY. Unexported temporarily by An Chi on Oct 15, 2021 until json or nmrML file is generated. 2D NMR Spectrum (experimental) | | Spectrum |
|
|---|
| Pathways |
|---|
| Pathways | | Name | SMPDB/Pathwhiz | KEGG | | Valine, Leucine and Isoleucine Degradation |  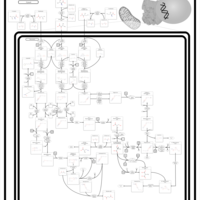 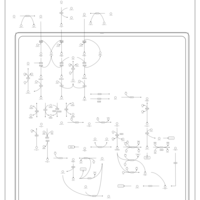 |  | | Beta-Ketothiolase Deficiency | 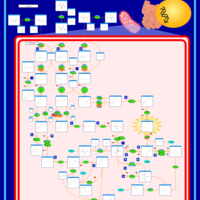 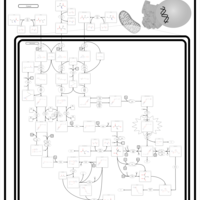 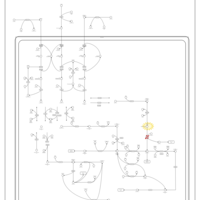 | Not Available | | 2-Methyl-3-Hydroxybutryl CoA Dehydrogenase Deficiency | 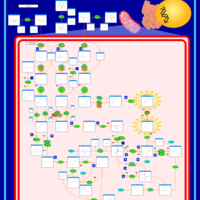 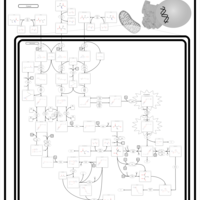 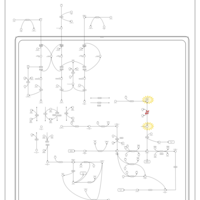 | Not Available | | Propionic Acidemia | 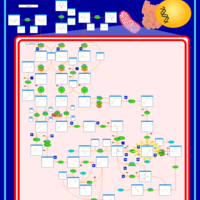 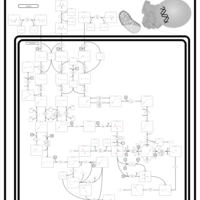 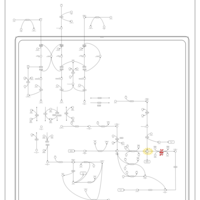 | Not Available | | 3-Hydroxy-3-Methylglutaryl-CoA Lyase Deficiency | 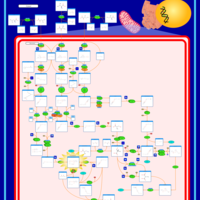 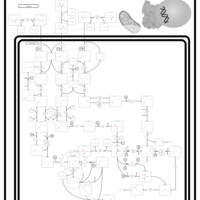 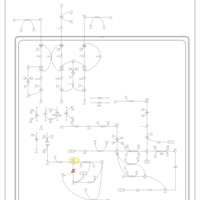 | Not Available |
|
|---|
| Protein Targets |
|---|
| Enzymes | |
| Pyruvate dehydrogenase E1 component subunit beta, mitochondrial | PDHB | 3p21.1-p14.2 | P11177 | details | | Pyruvate dehydrogenase E1 component subunit alpha, somatic form, mitochondrial | PDHA1 | Xp22.1 | P08559 | details | | Pyruvate dehydrogenase E1 component subunit alpha, testis-specific form, mitochondrial | PDHA2 | 4q22-q23 | P29803 | details | | Dihydrolipoyllysine-residue acetyltransferase component of pyruvate dehydrogenase complex, mitochondrial | DLAT | 11q23.1 | P10515 | details | | 2-oxoisovalerate dehydrogenase subunit beta, mitochondrial | BCKDHB | 6q14.1 | P21953 | details | | 2-oxoisovalerate dehydrogenase subunit alpha, mitochondrial | BCKDHA | 19q13.1-q13.2 | P12694 | details | | Branched-chain-amino-acid aminotransferase, cytosolic | BCAT1 | 12p12.1 | P54687 | details | | Branched-chain-amino-acid aminotransferase, mitochondrial | BCAT2 | 19q13 | O15382 | details |
|
|---|
| Transporters | |
|---|
| Metal Bindings | |
| 2-oxoisovalerate dehydrogenase subunit alpha, mitochondrial | BCKDHA | 19q13.1-q13.2 | P12694 | details |
|
|---|
| Receptors | Not Available |
|---|
| Transcriptional Factors | Not Available |
|---|
| Concentrations Data |
|---|
| Not Available |
|---|
| External Links |
|---|
| HMDB ID | HMDB0000019 |
|---|
| DrugBank ID | DB04074 |
|---|
| Phenol Explorer Compound ID | Not Available |
|---|
| FoodDB ID | FDB012250 |
|---|
| KNApSAcK ID | C00007623 |
|---|
| Chemspider ID | 48 |
|---|
| KEGG Compound ID | C00141 |
|---|
| BioCyc ID | 2-KETO-ISOVALERATE |
|---|
| BiGG ID | 34011 |
|---|
| Wikipedia Link | Alpha-Ketoisovaleric acid |
|---|
| METLIN ID | 5091 |
|---|
| PubChem Compound | 49 |
|---|
| PDB ID | Not Available |
|---|
| ChEBI ID | 16530 |
|---|
| References |
|---|
| General References | Not Available |
|---|
