| Record Information |
|---|
| Version | 1.0 |
|---|
| Created at | 2020-04-17 19:15:05 UTC |
|---|
| Updated at | 2020-11-18 16:39:27 UTC |
|---|
| CannabisDB ID | CDB005157 |
|---|
| Secondary Accession Numbers | Not Available |
|---|
| Cannabis Compound Identification |
|---|
| Common Name | (S)-3-Hydroxyisobutyric acid |
|---|
| Description | (S)-3-Hydroxyisobutyric acid, also known as (S)-3-hydroxy-2-methylpropanoate or 3-hydroxy-isobutyrate, belongs to the class of organic compounds known as beta hydroxy acids and derivatives. Beta hydroxy acids and derivatives are compounds containing a carboxylic acid substituted with a hydroxyl group on the C3 carbon atom (S)-3-Hydroxyisobutyric acid is an extremely weak basic (essentially neutral) compound (based on its pKa) (S)-3-Hydroxyisobutyric acid exists in all living species, ranging from bacteria to humans. The severity of this disease varies from case to case (S)-3-hydroxyisobutyric acid can be converted into (S)-methylmalonic acid semialdehyde through its interaction with the enzymes 3-hydroxyisobutyrate dehydrogenase, mitochondrial and enoyl-CoA hydratase, mitochondrial. In humans, (S)-3-hydroxyisobutyric acid is involved in the metabolic disorder called the 2-methyl-3-hydroxybutyryl-coa dehydrogenase deficiency pathway (S)-3-Hydroxyisobutyric acid is an intermediate in the metabolic pathways of L-valine and L-thymine amino acids. The deficient enzyme in 3HiB-uria remains unclear (S)-3-Hydroxyisobutyric acid is a potentially toxic compound (S)-3-Hydroxyisobutyric acid, with regard to humans, has been found to be associated with several diseases such as eosinophilic esophagitis, colorectal cancer, and diabetes mellitus type 2; (S)-3-hydroxyisobutyric acid has also been linked to the inborn metabolic disorder 3-hydroxyisobutyric aciduria. Patients with 3-hydroxyisobutyric aciduria excrete a significant amount of (S)-3-Hydroxyisobutyric acid not only during the acute stage but also when stable. (S)-3-Hydroxyisobutyric acid is expected to be in Cannabis as all living plants are known to produce and metabolize it. |
|---|
| Structure | |
|---|
| Synonyms | | Value | Source |
|---|
| (S)-3-Hydroxy-2-methylpropanoic acid | ChEBI | | (S)-3-Hydroxy-2-methylpropionic acid | ChEBI | | (S)-3-Hydroxy-2-methylpropanoate | Generator | | (S)-3-Hydroxy-2-methylpropionate | Generator | | (S)-3-Hydroxyisobutyrate | Generator | | 2-Methyl-L-(+)-hydracrylate | HMDB | | 2-Methyl-L-(+)-hydracrylic acid | HMDB | | 3-Hydroxy(iso)butyric acid | HMDB | | 3-Hydroxy-2-methyl-(S)-propanoate | HMDB | | 3-Hydroxy-2-methyl-(S)-propanoic acid | HMDB | | 3-Hydroxy-2-methylpropanoate | HMDB | | 3-Hydroxy-2-methylpropanoic acid | HMDB | | 3-Hydroxy-isobutyrate | HMDB | | 3-Hydroxyisobutyrate | HMDB | | 3-Hydroxyisobutyric acid | HMDB | | (2S)-3-Hydroxy-2-methylpropanoic acid | HMDB | | (2S)-3-Hydroxy-2-methylpropionic acid | HMDB | | (S)-beta-Hydroxyisobutyric acid | HMDB | | (S)-Β-hydroxyisobutyric acid | HMDB | | (±)-3-hydroxy-2-methylpropanoic acid | HMDB | | (±)-3-hydroxy-2-methylpropionic acid | HMDB | | 2-(Hydroxymethyl)propanoic acid | HMDB | | 2-(Hydroxymethyl)propionic acid | HMDB | | 2-Methyl-3-hydroxypropanoic acid | HMDB | | 2-Methyl-3-hydroxypropionic acid | HMDB | | 3-HIBA | HMDB | | 3-Hydroxy-2-methylpropionic acid | HMDB | | DL-3-Hydroxyisobutyric acid | HMDB | | L-(+)-beta-Hydroxyisobutyric acid | HMDB | | L-(+)-Β-hydroxyisobutyric acid | HMDB | | beta-Hydroxyisobutyric acid | HMDB | | Β-hydroxyisobutyric acid | HMDB | | 3-Hydroxy-2-isobutyrate | HMDB | | 3-Hydroxy-2-isobutyric acid | HMDB | | (S)-3-Hydroxyisobutyric acid | HMDB |
|
|---|
| Chemical Formula | C4H8O3 |
|---|
| Average Molecular Weight | 104.1 |
|---|
| Monoisotopic Molecular Weight | 104.0473 |
|---|
| IUPAC Name | (2S)-3-hydroxy-2-methylpropanoic acid |
|---|
| Traditional Name | (S)-3-hydroxyisobutyric acid |
|---|
| CAS Registry Number | 2068-83-9 |
|---|
| SMILES | C[C@@H](CO)C(O)=O |
|---|
| InChI Identifier | InChI=1S/C4H8O3/c1-3(2-5)4(6)7/h3,5H,2H2,1H3,(H,6,7)/t3-/m0/s1 |
|---|
| InChI Key | DBXBTMSZEOQQDU-VKHMYHEASA-N |
|---|
| Chemical Taxonomy |
|---|
| Description | Belongs to the class of organic compounds known as beta hydroxy acids and derivatives. Beta hydroxy acids and derivatives are compounds containing a carboxylic acid substituted with a hydroxyl group on the C3 carbon atom. |
|---|
| Kingdom | Organic compounds |
|---|
| Super Class | Organic acids and derivatives |
|---|
| Class | Hydroxy acids and derivatives |
|---|
| Sub Class | Beta hydroxy acids and derivatives |
|---|
| Direct Parent | Beta hydroxy acids and derivatives |
|---|
| Alternative Parents | |
|---|
| Substituents | - Beta-hydroxy acid
- Monocarboxylic acid or derivatives
- Carboxylic acid
- Carboxylic acid derivative
- Organic oxygen compound
- Organic oxide
- Hydrocarbon derivative
- Primary alcohol
- Organooxygen compound
- Carbonyl group
- Alcohol
- Aliphatic acyclic compound
|
|---|
| Molecular Framework | Aliphatic acyclic compounds |
|---|
| External Descriptors | |
|---|
| Ontology |
|---|
|
| Physiological effect | Health effect: |
|---|
| Disposition | Source: Biological location: |
|---|
| Physical Properties |
|---|
| State | Solid |
|---|
| Experimental Properties | | Property | Value | Reference |
|---|
| Melting Point | Not Available | Not Available | | Boiling Point | Not Available | Not Available | | Water Solubility | Not Available | Not Available | | logP | Not Available | Not Available |
|
|---|
| Predicted Properties | [] |
|---|
| Spectra |
|---|
| EI-MS/GC-MS | | Type | Description | Splash Key | View |
|---|
| Predicted GC-MS | (S)-3-Hydroxyisobutyric acid, non-derivatized, Predicted GC-MS Spectrum - 70eV, Positive | splash10-0ab9-9000000000-dd1c22f7a337d3289497 | Spectrum | | Predicted GC-MS | (S)-3-Hydroxyisobutyric acid, 2 TMS, Predicted GC-MS Spectrum - 70eV, Positive | splash10-00gr-9720000000-2a61310303ff8f2a7b42 | Spectrum | | Predicted GC-MS | (S)-3-Hydroxyisobutyric acid, non-derivatized, Predicted GC-MS Spectrum - 70eV, Positive | Not Available | Spectrum | | Predicted GC-MS | (S)-3-Hydroxyisobutyric acid, TMS_1_1, Predicted GC-MS Spectrum - 70eV, Positive | Not Available | Spectrum | | Predicted GC-MS | (S)-3-Hydroxyisobutyric acid, TMS_1_2, Predicted GC-MS Spectrum - 70eV, Positive | Not Available | Spectrum | | Predicted GC-MS | (S)-3-Hydroxyisobutyric acid, TBDMS_1_1, Predicted GC-MS Spectrum - 70eV, Positive | Not Available | Spectrum | | Predicted GC-MS | (S)-3-Hydroxyisobutyric acid, TBDMS_1_2, Predicted GC-MS Spectrum - 70eV, Positive | Not Available | Spectrum | | Predicted GC-MS | (S)-3-Hydroxyisobutyric acid, TBDMS_2_1, Predicted GC-MS Spectrum - 70eV, Positive | Not Available | Spectrum |
|
|---|
| MS/MS | | Type | Description | Splash Key | View |
|---|
| MS/MS | LC-MS/MS Spectrum - Quattro_QQQ 10V, Negative (Annotated) | splash10-0zfr-8900000000-b51c12e744025b833f31 | 2012-07-24 | View Spectrum | | MS/MS | LC-MS/MS Spectrum - Quattro_QQQ 25V, Negative (Annotated) | splash10-0a4i-9100000000-0d72fcffff267d7bc980 | 2012-07-24 | View Spectrum | | MS/MS | LC-MS/MS Spectrum - Quattro_QQQ 40V, Negative (Annotated) | splash10-0zfs-9400000000-b3b27a9cc8fd4ebe9eb1 | 2012-07-24 | View Spectrum | | Predicted MS/MS | Predicted LC-MS/MS Spectrum - 10V, Positive | splash10-052r-9500000000-543c65dad21c233fc8a8 | 2016-09-12 | View Spectrum | | Predicted MS/MS | Predicted LC-MS/MS Spectrum - 20V, Positive | splash10-052o-9000000000-7b7dcb34e28cb55ddafb | 2016-09-12 | View Spectrum | | Predicted MS/MS | Predicted LC-MS/MS Spectrum - 40V, Positive | splash10-0006-9000000000-27deee4c6a386a43ebc3 | 2016-09-12 | View Spectrum | | Predicted MS/MS | Predicted LC-MS/MS Spectrum - 10V, Negative | splash10-0udi-3900000000-a7aa709e1985355b4bd9 | 2016-09-12 | View Spectrum | | Predicted MS/MS | Predicted LC-MS/MS Spectrum - 20V, Negative | splash10-0pi0-9200000000-01a40fbe4d19283ef080 | 2016-09-12 | View Spectrum | | Predicted MS/MS | Predicted LC-MS/MS Spectrum - 40V, Negative | splash10-0a4l-9000000000-622de763b5ea31ff4ee6 | 2016-09-12 | View Spectrum | | Predicted MS/MS | Predicted LC-MS/MS Spectrum - 10V, Positive | splash10-0ap3-9000000000-6c76b805d02ad21ab06a | 2021-09-23 | View Spectrum | | Predicted MS/MS | Predicted LC-MS/MS Spectrum - 20V, Positive | splash10-0aou-9000000000-fc5e7dbe1e224cc87d4f | 2021-09-23 | View Spectrum | | Predicted MS/MS | Predicted LC-MS/MS Spectrum - 40V, Positive | splash10-0006-9000000000-021ab3207a43699617f4 | 2021-09-23 | View Spectrum | | Predicted MS/MS | Predicted LC-MS/MS Spectrum - 10V, Negative | splash10-0uki-9700000000-04713faa703ce789efb3 | 2021-09-25 | View Spectrum | | Predicted MS/MS | Predicted LC-MS/MS Spectrum - 20V, Negative | splash10-0a4r-9100000000-b0202d15bd6411bc7d80 | 2021-09-25 | View Spectrum | | Predicted MS/MS | Predicted LC-MS/MS Spectrum - 40V, Negative | splash10-052f-9000000000-d2c32880fd340aac0bad | 2021-09-25 | View Spectrum |
|
|---|
| NMR | | Type | Description | | View |
|---|
| 1D NMR | 1H NMR Spectrum (1D, 500 MHz, H2O, experimental) | | Spectrum | | 2D NMR | [1H, 13C]-HSQC NMR Spectrum (2D, 600 MHz, H2O, experimental) | | Spectrum |
|
|---|
| Pathways |
|---|
| Pathways | | Name | SMPDB/Pathwhiz | KEGG | | Valine, Leucine and Isoleucine Degradation |  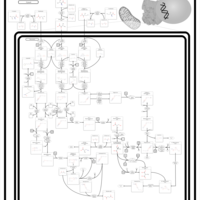 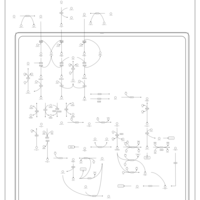 |  | | Beta-Ketothiolase Deficiency | 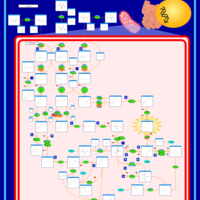 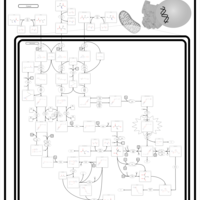 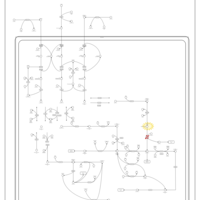 | Not Available | | 2-Methyl-3-Hydroxybutryl CoA Dehydrogenase Deficiency |  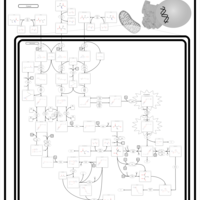 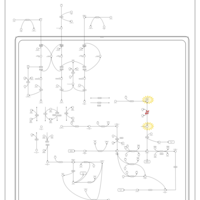 | Not Available | | Propionic Acidemia | 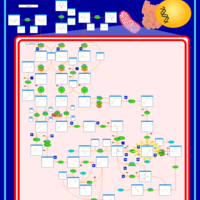 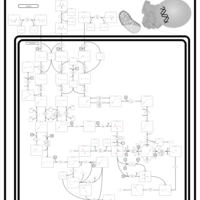 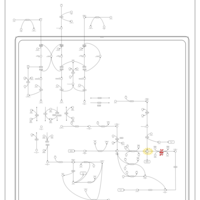 | Not Available | | 3-Hydroxy-3-Methylglutaryl-CoA Lyase Deficiency | 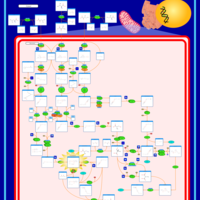 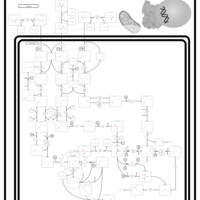 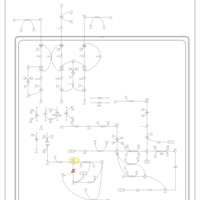 | Not Available |
|
|---|
| Protein Targets |
|---|
| Enzymes | |
| Sulfotransferase family cytosolic 2B member 1 | SULT2B1 | 19q13.3 | O00204 | details | | Hydroxyacyl-coenzyme A dehydrogenase, mitochondrial | HADH | 4q22-q26 | Q16836 | details | | 3-hydroxyisobutyrate dehydrogenase, mitochondrial | HIBADH | 7p15.2 | P31937 | details | | UDP-glucuronosyltransferase 1-1 | UGT1A1 | 2q37 | P22309 | details | | 3-hydroxyisobutyryl-CoA hydrolase, mitochondrial | HIBCH | 2q32.2 | Q6NVY1 | details |
|
|---|
| Transporters | Not Available |
|---|
| Metal Bindings | Not Available |
|---|
| Receptors | Not Available |
|---|
| Transcriptional Factors | Not Available |
|---|
| Concentrations Data |
|---|
| Not Available |
|---|
| External Links |
|---|
| HMDB ID | HMDB0000023 |
|---|
| DrugBank ID | Not Available |
|---|
| Phenol Explorer Compound ID | Not Available |
|---|
| FoodDB ID | FDB021877 |
|---|
| KNApSAcK ID | Not Available |
|---|
| Chemspider ID | 389707 |
|---|
| KEGG Compound ID | C06001 |
|---|
| BioCyc ID | CPD-12175 |
|---|
| BiGG ID | 37034 |
|---|
| Wikipedia Link | Not Available |
|---|
| METLIN ID | 483 |
|---|
| PubChem Compound | 440873 |
|---|
| PDB ID | Not Available |
|---|
| ChEBI ID | 37373 |
|---|
| References |
|---|
| General References | Not Available |
|---|
