| Record Information |
|---|
| Version | 1.0 |
|---|
| Created at | 2020-04-17 19:11:01 UTC |
|---|
| Updated at | 2020-11-18 16:39:23 UTC |
|---|
| CannabisDB ID | CDB005117 |
|---|
| Secondary Accession Numbers | Not Available |
|---|
| Cannabis Compound Identification |
|---|
| Common Name | 5,10-Methenyltetrahydrofolic acid |
|---|
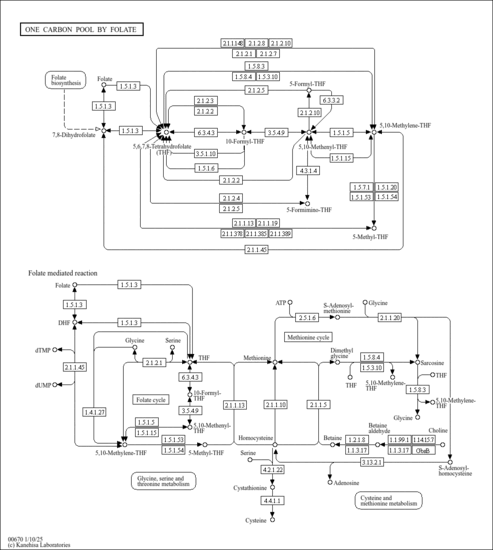
| Description | 5,10-Methenyltetrahydrofolic acid, also known as anhydro-leucovorin or 5,10-methenyl-THF, belongs to the class of organic compounds known as tetrahydrofolic acids. These are heterocyclic compounds based on the 5,6,7,8-tetrahydropteroic acid skeleton conjugated with at least one L-glutamic acid unit. It can also be produced as an intermediate in histidine catabolism, by formiminotransferase cyclodeaminase, from 5-formiminotetrahydrofolate.5,10-CHTHF is a substrate for methenyltetrahydrofolate cyclohydrolase, which converts it into 10-formyltetrahydrofolate. 5,10-Methenyltetrahydrofolic acid is a moderately basic compound (based on its pKa). 5,10-Methenyltetrahydrofolic acid exists in all living species, ranging from bacteria to humans. Click on genes, proteins and metabolites below to link to respective articles. Within humans, 5,10-methenyltetrahydrofolic acid participates in a number of enzymatic reactions. In particular, 5,10-methenyltetrahydrofolic acid can be biosynthesized from 10-formyltetrahydrofolate through the action of the enzyme C-1-tetrahydrofolate synthase, cytoplasmic. In addition, 5,10-methenyltetrahydrofolic acid can be biosynthesized from N5-formyl-THF; which is mediated by the enzyme 5-formyltetrahydrofolate cyclo-ligase. In humans, 5,10-methenyltetrahydrofolic acid is involved in the metabolic disorder called the methylenetetrahydrofolate reductase deficiency (mthfrd) pathway. It is produced from 5,10-methylenetetrahydrofolate by either a NAD+ dependent methylenetetrahydrofolate dehydrogenase, or a NADP+ dependent dehydrogenase. 5,10-Methenyltetrahydrofolate (5,10-CHTHF) is a form of tetrahydrofolate that is an intermediate in metabolism. 5,10-CHTHF is a coenzyme that accepts and donates methenyl (CH) groups. 5,10-Methenyltetrahydrofolic acid is expected to be in Cannabis as all living plants are known to produce and metabolize it. |
|---|
| Structure | |
|---|
| Synonyms | | Value | Source |
|---|
| 5,10-Methenyltetrahydrofolate | Generator | | 5,10-Methenyl-THF | HMDB | | Anhydro-leucovorin | HMDB | | Anhydro-leucovorin a | HMDB | | Anhydroleucovorin | HMDB | | Anhydroleucovorin a | HMDB | | CH-THF | HMDB | | Methenyl-tetrahydrofolate | HMDB | | Methenyl-THF | HMDB | | Methenyltetrahydrofolate | HMDB | | Methenyltetrahydrofolic acid | HMDB | | N5-N10-CH-THF | HMDB | | N5-N10-Methenyltetrahydrofolate | HMDB | | 5,10-Methenyltetrahydropteroylglutamate | HMDB | | N5,N10-Methenyl tetrahydrofolate | HMDB |
|
|---|
| Chemical Formula | C20H21N7O6 |
|---|
| Average Molecular Weight | 455.42 |
|---|
| Monoisotopic Molecular Weight | 455.1553 |
|---|
| IUPAC Name | (6aR)-8-(4-{[(1S)-1,3-dicarboxypropyl]-C-hydroxycarbonimidoyl}phenyl)-1-hydroxy-3-imino-3H,4H,5H,6H,6aH,7H-8lambda5-imidazo[1,5-f]pteridin-8-ylium |
|---|
| Traditional Name | (6aR)-8-(4-{[(1S)-1,3-dicarboxypropyl]-C-hydroxycarbonimidoyl}phenyl)-1-hydroxy-3-imino-4H,5H,6H,6aH,7H-8lambda5-imidazo[1,5-f]pteridin-8-ylium |
|---|
| CAS Registry Number | 7444-29-3 |
|---|
| SMILES | [H][C@@]12CN(C=[N+]1C1=C(NC2)NC(N)=NC1=O)C1=CC=C(C=C1)C(=O)N[C@@H](CCC(O)=O)C([O-])=O |
|---|
| InChI Identifier | InChI=1S/C20H21N7O6/c21-20-24-16-15(18(31)25-20)27-9-26(8-12(27)7-22-16)11-3-1-10(2-4-11)17(30)23-13(19(32)33)5-6-14(28)29/h1-4,9,12-13H,5-8H2,(H6-,21,22,23,24,25,28,29,30,31,32,33)/t12-,13+/m1/s1 |
|---|
| InChI Key | MEANFMOQMXYMCT-OLZOCXBDSA-N |
|---|
| Chemical Taxonomy |
|---|
| Description | Belongs to the class of organic compounds known as tetrahydrofolic acids. These are heterocyclic compounds based on the 5,6,7,8-tetrahydropteroic acid skeleton conjugated with at least one L-glutamic acid unit. |
|---|
| Kingdom | Organic compounds |
|---|
| Super Class | Organoheterocyclic compounds |
|---|
| Class | Pteridines and derivatives |
|---|
| Sub Class | Pterins and derivatives |
|---|
| Direct Parent | Tetrahydrofolic acids |
|---|
| Alternative Parents | |
|---|
| Substituents | - Tetrahydrofolic acid
- Glutamic acid or derivatives
- N-acyl-alpha amino acid or derivatives
- N-acyl-alpha-amino acid
- Hippuric acid
- Hippuric acid or derivatives
- Alpha-amino acid or derivatives
- Benzamide
- Benzoic acid or derivatives
- Aniline or substituted anilines
- Benzoyl
- Aminopyrimidine
- Pyrimidone
- Secondary aliphatic/aromatic amine
- Monocyclic benzene moiety
- Pyrimidine
- Dicarboxylic acid or derivatives
- Benzenoid
- Vinylogous amide
- Heteroaromatic compound
- 2-imidazoline
- Carboxamide group
- Amino acid
- Secondary carboxylic acid amide
- Carboxylic acid salt
- Amino acid or derivatives
- Amidine
- Azacycle
- Carboxylic acid amidine
- Organic 1,3-dipolar compound
- Carboxylic acid derivative
- Propargyl-type 1,3-dipolar organic compound
- Carboxylic acid
- Secondary amine
- Organic oxygen compound
- Amine
- Carbonyl group
- Organic salt
- Organic nitrogen compound
- Organopnictogen compound
- Organic oxide
- Organic zwitterion
- Hydrocarbon derivative
- Primary amine
- Organooxygen compound
- Organonitrogen compound
- Aromatic heteropolycyclic compound
|
|---|
| Molecular Framework | Aromatic heteropolycyclic compounds |
|---|
| External Descriptors | Not Available |
|---|
| Ontology |
|---|
|
| Disposition | Source: Biological location: |
|---|
| Physical Properties |
|---|
| State | Solid |
|---|
| Experimental Properties | | Property | Value | Reference |
|---|
| Melting Point | Not Available | Not Available | | Boiling Point | Not Available | Not Available | | Water Solubility | Not Available | Not Available | | logP | Not Available | Not Available |
|
|---|
| Predicted Properties | [] |
|---|
| EI-MS/GC-MS | | Type | Description | Splash Key | View |
|---|
| Predicted GC-MS | 5,10-Methenyltetrahydrofolic acid, non-derivatized, Predicted GC-MS Spectrum - 70eV, Positive | splash10-03di-2408900000-90c26f197ed01948843b | Spectrum | | Predicted GC-MS | 5,10-Methenyltetrahydrofolic acid, 1 TMS, Predicted GC-MS Spectrum - 70eV, Positive | splash10-0230-9506630000-d9b025a3f36832b17d97 | Spectrum | | Predicted GC-MS | 5,10-Methenyltetrahydrofolic acid, non-derivatized, Predicted GC-MS Spectrum - 70eV, Positive | Not Available | Spectrum | | Predicted GC-MS | 5,10-Methenyltetrahydrofolic acid, non-derivatized, Predicted GC-MS Spectrum - 70eV, Positive | Not Available | Spectrum |
|
|---|
| MS/MS | | Type | Description | Splash Key | View |
|---|
| Predicted MS/MS | Predicted LC-MS/MS Spectrum - 10V, Positive | splash10-0a4i-0000900000-9a74d30be444557240b0 | 2015-09-15 | View Spectrum | | Predicted MS/MS | Predicted LC-MS/MS Spectrum - 20V, Positive | splash10-0a4i-0000900000-87ffec278a0d552070d3 | 2015-09-15 | View Spectrum | | Predicted MS/MS | Predicted LC-MS/MS Spectrum - 40V, Positive | splash10-056r-9210100000-8e44a81962b4227dbc32 | 2015-09-15 | View Spectrum | | Predicted MS/MS | Predicted LC-MS/MS Spectrum - 10V, Negative | splash10-0udi-0000900000-6e21d81553cf048dcee3 | 2015-09-15 | View Spectrum | | Predicted MS/MS | Predicted LC-MS/MS Spectrum - 20V, Negative | splash10-0udi-2000900000-e3e2170131566f2c34e9 | 2015-09-15 | View Spectrum | | Predicted MS/MS | Predicted LC-MS/MS Spectrum - 40V, Negative | splash10-0006-9300100000-8e0fa01b9b99e8b1fea8 | 2015-09-15 | View Spectrum | | Predicted MS/MS | Predicted LC-MS/MS Spectrum - 10V, Positive | splash10-0a4i-0003900000-a08e1d1cb1452492cc40 | 2021-09-22 | View Spectrum | | Predicted MS/MS | Predicted LC-MS/MS Spectrum - 20V, Positive | splash10-0bt9-2109100000-a099f262b4e99916ab71 | 2021-09-22 | View Spectrum | | Predicted MS/MS | Predicted LC-MS/MS Spectrum - 40V, Positive | splash10-053r-0396000000-4a34a1ca0c2780791f62 | 2021-09-22 | View Spectrum | | Predicted MS/MS | Predicted LC-MS/MS Spectrum - 10V, Negative | splash10-0f79-0004900000-b7d25e9a9f2b33373222 | 2021-09-22 | View Spectrum | | Predicted MS/MS | Predicted LC-MS/MS Spectrum - 20V, Negative | splash10-001u-4129500000-674f21af43f4a24fc9aa | 2021-09-22 | View Spectrum | | Predicted MS/MS | Predicted LC-MS/MS Spectrum - 40V, Negative | splash10-0f9y-9643100000-a3b87dfc44080a520c03 | 2021-09-22 | View Spectrum |
|
|---|