| Record Information |
|---|
| Version | 1.0 |
|---|
| Created at | 2020-04-17 18:43:08 UTC |
|---|
| Updated at | 2020-12-07 19:11:06 UTC |
|---|
| CannabisDB ID | CDB004849 |
|---|
| Secondary Accession Numbers | Not Available |
|---|
| Cannabis Compound Identification |
|---|
| Common Name | D-2-Hydroxyglutaric acid |
|---|
| Description | D-2-Hydroxyglutaric acid, also known as (R)-hydroxyglutarate or (R)-2-hydroxy-pentanedioate, belongs to the class of organic compounds known as short-chain hydroxy acids and derivatives. These are hydroxy acids with an alkyl chain the contains less than 6 carbon atoms. D-2-Hydroxyglutaric acid is an extremely weak basic (essentially neutral) compound (based on its pKa). D-2-Hydroxyglutaric acid exists in all living organisms, ranging from bacteria to humans. D-2-hydroxyglutaric acid can be biosynthesized from oxoglutaric acid through the action of the enzyme D-2-hydroxyglutarate dehydrogenase, mitochondrial. In humans, D-2-hydroxyglutaric acid is involved in the metabolic disorder called the oncogenic action of d-2-hydroxyglutarate in hydroxyglutaric aciduria. Outside of the human body, D-2-Hydroxyglutaric acid has been detected, but not quantified in, milk (cow). This could make D-2-hydroxyglutaric acid a potential biomarker for the consumption of these foods. D-2-Hydroxyglutaric acid is a potentially toxic compound. D-2-Hydroxyglutaric acid, with regard to humans, has been found to be associated with several diseases such as d, l-2-hydroxyglutaric aciduria and spondyloenchondrodysplasia; D-2-hydroxyglutaric acid has also been linked to the inborn metabolic disorder d-2-hydroxyglutaric aciduria. The (R)-enantiomer of 2-hydroxyglutaric acid. D-2-Hydroxyglutaric acid is expected to be in Cannabis as all living plants are known to produce and metabolize it. |
|---|
| Structure | |
|---|
| Synonyms | | Value | Source |
|---|
| (R)-2-Hydroxyglutarate | ChEBI | | (R)-Hydroxyglutarate | ChEBI | | (R)-2-Hydroxyglutaric acid | Generator | | (R)-Hydroxyglutaric acid | Generator | | D-2-Hydroxyglutarate | Generator | | (R)-2-Hydroxy-pentanedioate | HMDB | | (R)-2-Hydroxy-pentanedioic acid | HMDB | | (R)-a-Hydroxyglutarate | HMDB | | (R)-a-Hydroxyglutaric acid | HMDB | | (R)-alpha-Hydroxyglutarate | HMDB | | (R)-alpha-Hydroxyglutaric acid | HMDB | | 2-Hydroxy-D-glutarate | HMDB | | 2-Hydroxy-D-glutaric acid | HMDB | | 2-Hydroxy-delta-glutarate | HMDB | | 2-Hydroxy-delta-glutaric acid | HMDB | | D-a-Hydroxyglutarate | HMDB | | D-a-Hydroxyglutaric acid | HMDB | | delta-2-Hydroxyglutarate | HMDB | | delta-2-Hydroxyglutaric acid | HMDB | | delta-alpha-Hydroxyglutarate | HMDB | | delta-alpha-Hydroxyglutaric acid | HMDB | | 2-Hydroxyglutarate | HMDB | | alpha-Hydroxyglutarate, (D)-isomer | HMDB | | alpha-Hydroxyglutarate, (DL)-isomer | HMDB | | 2-Hydroxyglutaric acid | HMDB | | alpha-Hydroxyglutarate | HMDB | | alpha-Hydroxyglutarate, (L)-isomer | HMDB | | alpha-Hydroxyglutarate, disodium salt | HMDB |
|
|---|
| Chemical Formula | C5H8O5 |
|---|
| Average Molecular Weight | 148.11 |
|---|
| Monoisotopic Molecular Weight | 148.0372 |
|---|
| IUPAC Name | (2R)-2-hydroxypentanedioic acid |
|---|
| Traditional Name | D-2-hydroxyglutaric acid |
|---|
| CAS Registry Number | 13095-47-1 |
|---|
| SMILES | O[C@H](CCC(O)=O)C(O)=O |
|---|
| InChI Identifier | InChI=1S/C5H8O5/c6-3(5(9)10)1-2-4(7)8/h3,6H,1-2H2,(H,7,8)(H,9,10)/t3-/m1/s1 |
|---|
| InChI Key | HWXBTNAVRSUOJR-GSVOUGTGSA-N |
|---|
| Chemical Taxonomy |
|---|
| Description | Belongs to the class of organic compounds known as short-chain hydroxy acids and derivatives. These are hydroxy acids with an alkyl chain the contains less than 6 carbon atoms. |
|---|
| Kingdom | Organic compounds |
|---|
| Super Class | Organic acids and derivatives |
|---|
| Class | Hydroxy acids and derivatives |
|---|
| Sub Class | Short-chain hydroxy acids and derivatives |
|---|
| Direct Parent | Short-chain hydroxy acids and derivatives |
|---|
| Alternative Parents | |
|---|
| Substituents | - Short-chain hydroxy acid
- Fatty acid
- Monosaccharide
- Dicarboxylic acid or derivatives
- Alpha-hydroxy acid
- Secondary alcohol
- Carboxylic acid
- Carboxylic acid derivative
- Organic oxygen compound
- Organic oxide
- Hydrocarbon derivative
- Organooxygen compound
- Carbonyl group
- Alcohol
- Aliphatic acyclic compound
|
|---|
| Molecular Framework | Aliphatic acyclic compounds |
|---|
| External Descriptors | |
|---|
| Ontology |
|---|
|
| Physiological effect | Health effect: |
|---|
| Disposition | Source: Biological location: |
|---|
| Role | Indirect biological role: Biological role: |
|---|
| Physical Properties |
|---|
| State | Solid |
|---|
| Experimental Properties | | Property | Value | Reference |
|---|
| Melting Point | > 300 °C | Not Available | | Boiling Point | Not Available | Not Available | | Water Solubility | Not Available | Not Available | | logP | Not Available | Not Available |
|
|---|
| Predicted Properties | [] |
|---|
| Spectra |
|---|
| EI-MS/GC-MS | | Type | Description | Splash Key | View |
|---|
| Predicted GC-MS | D-2-Hydroxyglutaric acid, non-derivatized, Predicted GC-MS Spectrum - 70eV, Positive | splash10-0zgl-9300000000-805667744aba36281cec | Spectrum | | Predicted GC-MS | D-2-Hydroxyglutaric acid, 3 TMS, Predicted GC-MS Spectrum - 70eV, Positive | splash10-00gv-8191000000-f6fdffd87ffe1acf65b4 | Spectrum | | Predicted GC-MS | D-2-Hydroxyglutaric acid, non-derivatized, Predicted GC-MS Spectrum - 70eV, Positive | Not Available | Spectrum | | Predicted GC-MS | D-2-Hydroxyglutaric acid, non-derivatized, Predicted GC-MS Spectrum - 70eV, Positive | Not Available | Spectrum | | Predicted GC-MS | D-2-Hydroxyglutaric acid, TMS_1_1, Predicted GC-MS Spectrum - 70eV, Positive | Not Available | Spectrum | | Predicted GC-MS | D-2-Hydroxyglutaric acid, TMS_1_2, Predicted GC-MS Spectrum - 70eV, Positive | Not Available | Spectrum | | Predicted GC-MS | D-2-Hydroxyglutaric acid, TMS_1_3, Predicted GC-MS Spectrum - 70eV, Positive | Not Available | Spectrum | | Predicted GC-MS | D-2-Hydroxyglutaric acid, TMS_2_1, Predicted GC-MS Spectrum - 70eV, Positive | Not Available | Spectrum | | Predicted GC-MS | D-2-Hydroxyglutaric acid, TMS_2_2, Predicted GC-MS Spectrum - 70eV, Positive | Not Available | Spectrum | | Predicted GC-MS | D-2-Hydroxyglutaric acid, TMS_2_3, Predicted GC-MS Spectrum - 70eV, Positive | Not Available | Spectrum | | Predicted GC-MS | D-2-Hydroxyglutaric acid, TBDMS_1_1, Predicted GC-MS Spectrum - 70eV, Positive | Not Available | Spectrum | | Predicted GC-MS | D-2-Hydroxyglutaric acid, TBDMS_1_2, Predicted GC-MS Spectrum - 70eV, Positive | Not Available | Spectrum | | Predicted GC-MS | D-2-Hydroxyglutaric acid, TBDMS_1_3, Predicted GC-MS Spectrum - 70eV, Positive | Not Available | Spectrum | | Predicted GC-MS | D-2-Hydroxyglutaric acid, TBDMS_2_1, Predicted GC-MS Spectrum - 70eV, Positive | Not Available | Spectrum | | Predicted GC-MS | D-2-Hydroxyglutaric acid, TBDMS_2_2, Predicted GC-MS Spectrum - 70eV, Positive | Not Available | Spectrum | | Predicted GC-MS | D-2-Hydroxyglutaric acid, TBDMS_2_3, Predicted GC-MS Spectrum - 70eV, Positive | Not Available | Spectrum | | Predicted GC-MS | D-2-Hydroxyglutaric acid, TBDMS_3_1, Predicted GC-MS Spectrum - 70eV, Positive | Not Available | Spectrum |
|
|---|
| MS/MS | | Type | Description | Splash Key | View |
|---|
| Predicted MS/MS | Predicted LC-MS/MS Spectrum - 10V, Positive | splash10-001j-1900000000-dbf80fc2b86ec60d623e | 2016-06-03 | View Spectrum | | Predicted MS/MS | Predicted LC-MS/MS Spectrum - 20V, Positive | splash10-0m5r-9600000000-d1bc7ce2206164b5bc2c | 2016-06-03 | View Spectrum | | Predicted MS/MS | Predicted LC-MS/MS Spectrum - 40V, Positive | splash10-0a4i-9000000000-df3e77cb42d6f7647a64 | 2016-06-03 | View Spectrum | | Predicted MS/MS | Predicted LC-MS/MS Spectrum - 10V, Negative | splash10-0002-1900000000-095c1140772d4cdd149b | 2016-08-03 | View Spectrum | | Predicted MS/MS | Predicted LC-MS/MS Spectrum - 20V, Negative | splash10-0zfr-7900000000-ce158ac5b56eff867735 | 2016-08-03 | View Spectrum | | Predicted MS/MS | Predicted LC-MS/MS Spectrum - 40V, Negative | splash10-0a4i-9000000000-e30760384709aa978986 | 2016-08-03 | View Spectrum | | Predicted MS/MS | Predicted LC-MS/MS Spectrum - 10V, Negative | splash10-0fbi-3900000000-f88fcf3ac0dd06636b16 | 2021-09-22 | View Spectrum | | Predicted MS/MS | Predicted LC-MS/MS Spectrum - 20V, Negative | splash10-0k9i-9400000000-31bf8d3bb7203e7bf200 | 2021-09-22 | View Spectrum | | Predicted MS/MS | Predicted LC-MS/MS Spectrum - 40V, Negative | splash10-0a4u-9000000000-c0ac45c4d66178ac20cf | 2021-09-22 | View Spectrum | | Predicted MS/MS | Predicted LC-MS/MS Spectrum - 10V, Positive | splash10-0w39-4900000000-1385b471f7cdf14ca14d | 2021-09-22 | View Spectrum | | Predicted MS/MS | Predicted LC-MS/MS Spectrum - 20V, Positive | splash10-052r-9500000000-17ece6956644d4cf2069 | 2021-09-22 | View Spectrum | | Predicted MS/MS | Predicted LC-MS/MS Spectrum - 40V, Positive | splash10-0a4r-9000000000-dbb4195080bddb5f6210 | 2021-09-22 | View Spectrum |
|
|---|
| NMR | Not Available |
|---|
| Pathways |
|---|
| Pathways | | Name | SMPDB/Pathwhiz | KEGG | | The oncogenic action of D-2-hydroxyglutarate in Hydroxygluaricaciduria | 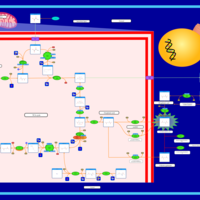 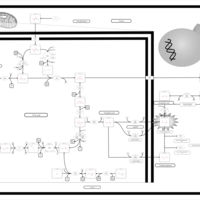 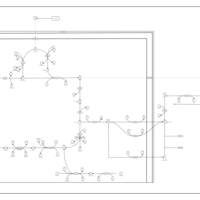 | Not Available | | The oncogenic action of L-2-hydroxyglutarate in Hydroxygluaricaciduria | 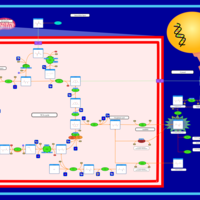 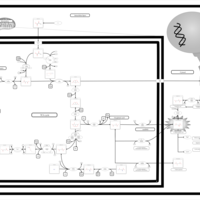 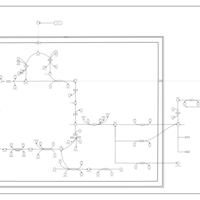 | Not Available |
|
|---|
| Protein Targets |
|---|
| Enzymes | |
| D-3-phosphoglycerate dehydrogenase | PHGDH | 1p12 | O43175 | details | | D-2-hydroxyglutarate dehydrogenase, mitochondrial | D2HGDH | 2q37.3 | Q8N465 | details | | Hydroxyacid-oxoacid transhydrogenase, mitochondrial | ADHFE1 | 8q13.1 | Q8IWW8 | details |
|
|---|
| Transporters | Not Available |
|---|
| Metal Bindings | |
| Hydroxyacid-oxoacid transhydrogenase, mitochondrial | ADHFE1 | 8q13.1 | Q8IWW8 | details |
|
|---|
| Receptors | Not Available |
|---|
| Transcriptional Factors | Not Available |
|---|
| Concentrations Data |
|---|
| Not Available |
|---|
| External Links |
|---|
| HMDB ID | HMDB0000606 |
|---|
| DrugBank ID | Not Available |
|---|
| Phenol Explorer Compound ID | Not Available |
|---|
| FoodDB ID | FDB022139 |
|---|
| KNApSAcK ID | Not Available |
|---|
| Chemspider ID | 388508 |
|---|
| KEGG Compound ID | C01087 |
|---|
| BioCyc ID | R-2-HYDROXYGLUTARATE |
|---|
| BiGG ID | Not Available |
|---|
| Wikipedia Link | Alpha-Hydroxyglutaric acid |
|---|
| METLIN ID | 5581 |
|---|
| PubChem Compound | 439391 |
|---|
| PDB ID | Not Available |
|---|
| ChEBI ID | 32796 |
|---|
| References |
|---|
| General References | Not Available |
|---|