| Record Information |
|---|
| Version | 1.0 |
|---|
| Created at | 2020-04-17 18:39:49 UTC |
|---|
| Updated at | 2022-12-13 23:36:27 UTC |
|---|
| CannabisDB ID | CDB004817 |
|---|
| Secondary Accession Numbers | Not Available |
|---|
| Cannabis Compound Identification |
|---|
| Common Name | L-Lactic acid |
|---|
| Description | L-Lactic acid, also known as L-lactate or L-milchsaeure, belongs to the class of organic compounds known as alpha hydroxy acids and derivatives. These are organic compounds containing a carboxylic acid substituted with a hydroxyl group on the adjacent carbon. L-Lactic acid is an extremely weak basic (essentially neutral) compound (based on its pKa). Within humans, L-lactic acid participates in a number of enzymatic reactions. In particular, L-lactic acid can be biosynthesized from pyruvic acid through the action of the enzyme L-lactate dehydrogenase. In addition, L-lactic acid can be converted into L-lactic acid through the action of the enzyme monocarboxylate transporter. In humans, L-lactic acid is involved in the metabolic disorder called the glutaminolysis and cancer pathway. L-Lactic acid is an acidic and odorless tasting compound. Outside of the human body, L-Lactic acid is found, on average, in the highest concentration within beers and milk (cow). L-Lactic acid has also been detected, but not quantified in, several different foods, such as cow milks, cow milks, cow milks, cow milks, and port wines. This could make L-lactic acid a potential biomarker for the consumption of these foods. L-Lactic acid is a potentially toxic compound. An optically active form of lactic acid having (S)-configuration. L-Lactic acid is expected to be in Cannabis as all living plants are known to produce and metabolize it. |
|---|
| Structure | |
|---|
| Synonyms | | Value | Source |
|---|
| (+)-Lactic acid | ChEBI | | (S)-(+)-Lactic acid | ChEBI | | (S)-2-Hydroxypropanoic acid | ChEBI | | (S)-2-Hydroxypropionic acid | ChEBI | | L-(+)-alpha-Hydroxypropionic acid | ChEBI | | L-(+)-Lactic acid | ChEBI | | L-Milchsaeure | ChEBI | | L-Lactate | Kegg | | (+)-Lactate | Generator | | (S)-(+)-Lactate | Generator | | (S)-2-Hydroxypropanoate | Generator | | (S)-2-Hydroxypropionate | Generator | | L-(+)-a-Hydroxypropionate | Generator | | L-(+)-a-Hydroxypropionic acid | Generator | | L-(+)-alpha-Hydroxypropionate | Generator | | L-(+)-Α-hydroxypropionate | Generator | | L-(+)-Α-hydroxypropionic acid | Generator | | L-(+)-Lactate | Generator | | (alpha)-Lactate | HMDB | | (alpha)-Lactic acid | HMDB | | (S)-(+)-2-Hydroxypropanoate | HMDB | | (S)-(+)-2-Hydroxypropanoic acid | HMDB | | (S)-2-Hydroxy-propanoate | HMDB | | (S)-2-Hydroxy-propanoic acid | HMDB | | (S)-Lactate | HMDB | | (S)-Lactic acid | HMDB | | 1-Hydroxyethane 1-carboxylate | HMDB | | 1-Hydroxyethane 1-carboxylic acid | HMDB | | 1-Hydroxyethanecarboxylate | HMDB | | 1-Hydroxyethanecarboxylic acid | HMDB | | 2-Hydroxypropanoate | HMDB | | 2-Hydroxypropanoic acid | HMDB | | 2-Hydroxypropionate | HMDB | | a-Hydroxypropanoate | HMDB | | a-Hydroxypropanoic acid | HMDB | | a-Hydroxypropionate | HMDB | | a-Hydroxypropionic acid | HMDB | | alpha-Hydroxypropanoate | HMDB | | alpha-Hydroxypropanoic acid | HMDB | | alpha-Hydroxypropionate | HMDB | | alpha-Hydroxypropionic acid | HMDB | | L-(+)- Lactic acid | HMDB | | L-2-Hydroxypropanoate | HMDB | | L-2-Hydroxypropanoic acid | HMDB | | Lactate | HMDB | | Lactic acid | HMDB | | Milk acid | HMDB | | Sarcolactic acid | HMDB | | 2-Hydroxypropionic acid | HMDB | | D-Lactic acid | HMDB | | D Lactic acid | HMDB | | Lactate, ammonium | HMDB | | 2 Hydroxypropanoic acid | HMDB | | 2 Hydroxypropionic acid | HMDB | | Ammonium lactate | HMDB | | L Lactic acid | HMDB | | D-Lactate | HMDB | | L-Lactic acid | ChEBI |
|
|---|
| Chemical Formula | C3H6O3 |
|---|
| Average Molecular Weight | 90.08 |
|---|
| Monoisotopic Molecular Weight | 90.0317 |
|---|
| IUPAC Name | (2S)-2-hydroxypropanoic acid |
|---|
| Traditional Name | (α)-lactate |
|---|
| CAS Registry Number | 79-33-4 |
|---|
| SMILES | C[C@H](O)C(O)=O |
|---|
| InChI Identifier | InChI=1S/C3H6O3/c1-2(4)3(5)6/h2,4H,1H3,(H,5,6)/t2-/m0/s1 |
|---|
| InChI Key | JVTAAEKCZFNVCJ-REOHCLBHSA-N |
|---|
| Chemical Taxonomy |
|---|
| Description | Belongs to the class of organic compounds known as alpha hydroxy acids and derivatives. These are organic compounds containing a carboxylic acid substituted with a hydroxyl group on the adjacent carbon. |
|---|
| Kingdom | Organic compounds |
|---|
| Super Class | Organic acids and derivatives |
|---|
| Class | Hydroxy acids and derivatives |
|---|
| Sub Class | Alpha hydroxy acids and derivatives |
|---|
| Direct Parent | Alpha hydroxy acids and derivatives |
|---|
| Alternative Parents | |
|---|
| Substituents | - Alpha-hydroxy acid
- Secondary alcohol
- Monocarboxylic acid or derivatives
- Carboxylic acid
- Carboxylic acid derivative
- Organic oxygen compound
- Organic oxide
- Hydrocarbon derivative
- Organooxygen compound
- Carbonyl group
- Alcohol
- Aliphatic acyclic compound
|
|---|
| Molecular Framework | Aliphatic acyclic compounds |
|---|
| External Descriptors | |
|---|
| Ontology |
|---|
|
| Physiological effect | Health effect: |
|---|
| Disposition | Route of exposure: Source: Biological location: |
|---|
| Role | Biological role: Indirect biological role: |
|---|
| Physical Properties |
|---|
| State | Liquid |
|---|
| Experimental Properties | | Property | Value | Reference |
|---|
| Melting Point | 16.8 °C | Not Available | | Boiling Point | Not Available | Not Available | | Water Solubility | Not Available | Not Available | | logP | Not Available | Not Available |
|
|---|
| Predicted Properties | [] |
|---|
| Spectra |
|---|
| EI-MS/GC-MS | | Type | Description | Splash Key | View |
|---|
| EI-MS | Mass Spectrum (Electron Ionization) | splash10-002b-9000000000-50213d6b39ef9741c466 | 2014-09-20 | View Spectrum | | GC-MS | L-Lactic acid, non-derivatized, GC-MS Spectrum | splash10-00kb-0900000000-fb59ec16914501aa19ab | Spectrum | | GC-MS | L-Lactic acid, non-derivatized, GC-MS Spectrum | splash10-014j-0900000000-c4d9e12b4b0150eda54b | Spectrum | | GC-MS | L-Lactic acid, non-derivatized, GC-MS Spectrum | splash10-00kb-0900000000-fb59ec16914501aa19ab | Spectrum | | Predicted GC-MS | L-Lactic acid, non-derivatized, Predicted GC-MS Spectrum - 70eV, Positive | splash10-0006-9000000000-a3691f383d440fb00e1f | Spectrum | | Predicted GC-MS | L-Lactic acid, 2 TMS, Predicted GC-MS Spectrum - 70eV, Positive | splash10-01b9-9620000000-f7faa7db9c1be3d9d975 | Spectrum | | Predicted GC-MS | L-Lactic acid, non-derivatized, Predicted GC-MS Spectrum - 70eV, Positive | Not Available | Spectrum | | Predicted GC-MS | L-Lactic acid, TMS_1_1, Predicted GC-MS Spectrum - 70eV, Positive | Not Available | Spectrum | | Predicted GC-MS | L-Lactic acid, TMS_1_2, Predicted GC-MS Spectrum - 70eV, Positive | Not Available | Spectrum | | Predicted GC-MS | L-Lactic acid, TBDMS_1_1, Predicted GC-MS Spectrum - 70eV, Positive | Not Available | Spectrum | | Predicted GC-MS | L-Lactic acid, TBDMS_1_2, Predicted GC-MS Spectrum - 70eV, Positive | Not Available | Spectrum | | Predicted GC-MS | L-Lactic acid, TBDMS_2_1, Predicted GC-MS Spectrum - 70eV, Positive | Not Available | Spectrum |
|
|---|
| MS/MS | | Type | Description | Splash Key | View |
|---|
| MS/MS | LC-MS/MS Spectrum - Quattro_QQQ 10V, Negative (Annotated) | splash10-000i-9000000000-1d5a5f55463acefb6fc7 | 2012-07-24 | View Spectrum | | MS/MS | LC-MS/MS Spectrum - Quattro_QQQ 25V, Negative (Annotated) | splash10-000m-9000000000-c07133799d8f43d4c75a | 2012-07-24 | View Spectrum | | MS/MS | LC-MS/MS Spectrum - Quattro_QQQ 40V, Negative (Annotated) | splash10-000i-9000000000-ddb080250741e69ab137 | 2012-07-24 | View Spectrum | | MS/MS | LC-MS/MS Spectrum - LC-ESI-QQ (API3000, Applied Biosystems) 10V, Negative | splash10-000i-9000000000-8f5d5eddb0b4b2a3b541 | 2012-08-31 | View Spectrum | | MS/MS | LC-MS/MS Spectrum - LC-ESI-QQ (API3000, Applied Biosystems) 20V, Negative | splash10-000i-9000000000-3bcfc4cdc49230f15642 | 2012-08-31 | View Spectrum | | MS/MS | LC-MS/MS Spectrum - LC-ESI-QQ (API3000, Applied Biosystems) 30V, Negative | splash10-0006-9000000000-48b511409f4f60cec04e | 2012-08-31 | View Spectrum | | MS/MS | LC-MS/MS Spectrum - LC-ESI-QQ (API3000, Applied Biosystems) 40V, Negative | splash10-0006-9000000000-365ea3c1bcfff2cab938 | 2012-08-31 | View Spectrum | | MS/MS | LC-MS/MS Spectrum - LC-ESI-QQ (API3000, Applied Biosystems) 50V, Negative | splash10-0006-9000000000-9d0238aeeb837213e81d | 2012-08-31 | View Spectrum | | MS/MS | LC-MS/MS Spectrum - LC-ESI-QQ , negative | splash10-000i-9000000000-8f5d5eddb0b4b2a3b541 | 2017-09-14 | View Spectrum | | MS/MS | LC-MS/MS Spectrum - LC-ESI-QQ , negative | splash10-000i-9000000000-3bcfc4cdc49230f15642 | 2017-09-14 | View Spectrum | | MS/MS | LC-MS/MS Spectrum - LC-ESI-QQ , negative | splash10-0006-9000000000-48b511409f4f60cec04e | 2017-09-14 | View Spectrum | | MS/MS | LC-MS/MS Spectrum - LC-ESI-QQ , negative | splash10-0006-9000000000-365ea3c1bcfff2cab938 | 2017-09-14 | View Spectrum | | MS/MS | LC-MS/MS Spectrum - LC-ESI-QQ , negative | splash10-0006-9000000000-9d0238aeeb837213e81d | 2017-09-14 | View Spectrum | | MS/MS | LC-MS/MS Spectrum - QTOF 30V, positive | splash10-014i-9000000000-046bb7f6cce9744388eb | 2020-07-22 | View Spectrum | | MS/MS | LC-MS/MS Spectrum - 10V, Negative | splash10-000f-9000000000-1c6ea519686b2066052f | 2021-09-20 | View Spectrum | | MS/MS | LC-MS/MS Spectrum - 35V, Negative | splash10-000i-9000000000-013763552dec660fac8c | 2021-09-20 | View Spectrum | | MS/MS | LC-MS/MS Spectrum - 20V, Negative | splash10-0006-9000000000-bc4403a40640361e5a52 | 2021-09-20 | View Spectrum | | Predicted MS/MS | Predicted LC-MS/MS Spectrum - 10V, Positive | splash10-006x-9000000000-5f417f4a6d08f0ab00ed | 2016-09-12 | View Spectrum | | Predicted MS/MS | Predicted LC-MS/MS Spectrum - 20V, Positive | splash10-00dm-9000000000-df7a94bb1a9cf6e78e1a | 2016-09-12 | View Spectrum | | Predicted MS/MS | Predicted LC-MS/MS Spectrum - 40V, Positive | splash10-004j-9000000000-dc2a1b965287b9dfee9c | 2016-09-12 | View Spectrum | | Predicted MS/MS | Predicted LC-MS/MS Spectrum - 10V, Negative | splash10-000i-9000000000-c3686a681cc9bbf039e1 | 2016-09-12 | View Spectrum | | Predicted MS/MS | Predicted LC-MS/MS Spectrum - 20V, Negative | splash10-000i-9000000000-ddad20647c2ac56efd22 | 2016-09-12 | View Spectrum | | Predicted MS/MS | Predicted LC-MS/MS Spectrum - 40V, Negative | splash10-00di-9000000000-b728b45617afcc6b67da | 2016-09-12 | View Spectrum | | Predicted MS/MS | Predicted LC-MS/MS Spectrum - 10V, Negative | splash10-000i-9000000000-b586cb8f053eb4465b4e | 2021-09-25 | View Spectrum | | Predicted MS/MS | Predicted LC-MS/MS Spectrum - 20V, Negative | splash10-000i-9000000000-400a5f1c0dfcc32ef2bb | 2021-09-25 | View Spectrum |
|
|---|
| NMR | | Type | Description | | View |
|---|
| 1D NMR | 1H NMR Spectrum (1D, 500 MHz, H2O, experimental) | | Spectrum | | 1D NMR | 13C NMR Spectrum (1D, 125 MHz, H2O, experimental) | | Spectrum | | 1D NMR | 1H NMR Spectrum (1D, D2O, experimental) | | Spectrum | | 1D NMR | 1H NMR Spectrum (1D, 100 MHz, D2O, predicted) | | Spectrum | | 1D NMR | 13C NMR Spectrum (1D, 100 MHz, D2O, predicted) | | Spectrum | | 1D NMR | 1H NMR Spectrum (1D, 200 MHz, D2O, predicted) | | Spectrum | | 1D NMR | 13C NMR Spectrum (1D, 200 MHz, D2O, predicted) | | Spectrum | | 1D NMR | 1H NMR Spectrum (1D, 300 MHz, D2O, predicted) | | Spectrum | | 1D NMR | 13C NMR Spectrum (1D, 300 MHz, D2O, predicted) | | Spectrum | | 1D NMR | 1H NMR Spectrum (1D, 400 MHz, D2O, predicted) | | Spectrum | | 1D NMR | 13C NMR Spectrum (1D, 400 MHz, D2O, predicted) | | Spectrum | | 1D NMR | 1H NMR Spectrum (1D, 500 MHz, D2O, predicted) | | Spectrum | | 1D NMR | 13C NMR Spectrum (1D, 500 MHz, D2O, predicted) | | Spectrum | | 1D NMR | 1H NMR Spectrum (1D, 600 MHz, D2O, predicted) | | Spectrum | | 1D NMR | 13C NMR Spectrum (1D, 600 MHz, D2O, predicted) | | Spectrum | | 1D NMR | 1H NMR Spectrum (1D, 700 MHz, D2O, predicted) | | Spectrum | | 1D NMR | 13C NMR Spectrum (1D, 700 MHz, D2O, predicted) | | Spectrum | | 1D NMR | 1H NMR Spectrum (1D, 800 MHz, D2O, predicted) | | Spectrum | | 1D NMR | 13C NMR Spectrum (1D, 800 MHz, D2O, predicted) | | Spectrum | | 1D NMR | 1H NMR Spectrum (1D, 900 MHz, D2O, predicted) | | Spectrum | | 1D NMR | 13C NMR Spectrum (1D, 900 MHz, D2O, predicted) | | Spectrum | | 1D NMR | 1H NMR Spectrum (1D, 1000 MHz, D2O, predicted) | | Spectrum | | 1D NMR | 13C NMR Spectrum (1D, 1000 MHz, D2O, predicted) | | Spectrum | | 1D NMR | 13C NMR Spectrum (1D, 400 MHz, H2O, experimental) | | Spectrum | | 2D NMR | [1H, 1H]-TOCSY. Unexported temporarily by An Chi on Oct 15, 2021 until json or nmrML file is generated. 2D NMR Spectrum (experimental) | | Spectrum | | 2D NMR | [1H, 13C]-HSQC NMR Spectrum (2D, 600 MHz, H2O, experimental) | | Spectrum |
|
|---|
| Pathways |
|---|
| Pathways | | Name | SMPDB/Pathwhiz | KEGG | | Gluconeogenesis |    | 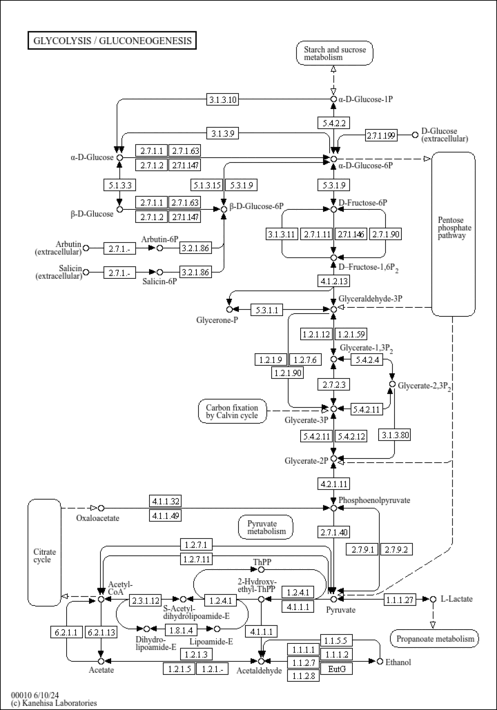 | | Pyruvate Metabolism |    | 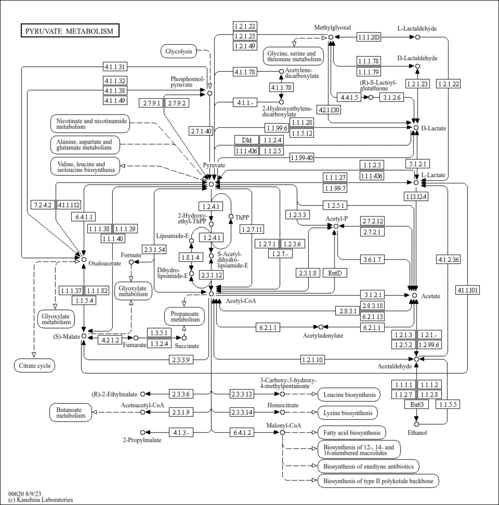 | | Leigh Syndrome |   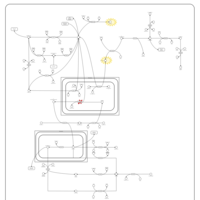 | Not Available | | Pyruvate Decarboxylase E1 Component Deficiency (PDHE1 Deficiency) | 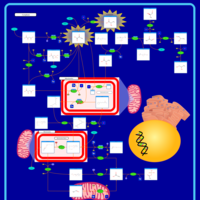 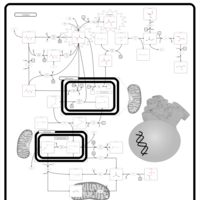 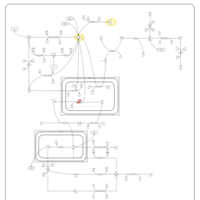 | Not Available | | Pyruvate Dehydrogenase Complex Deficiency | 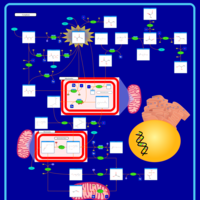 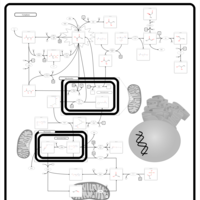 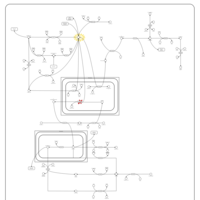 | Not Available |
|
|---|
| Protein Targets |
|---|
| Enzymes | |
|---|
| Transporters | Not Available |
|---|
| Metal Bindings | Not Available |
|---|
| Receptors | Not Available |
|---|
| Transcriptional Factors | Not Available |
|---|
| Concentrations Data |
|---|
| |
| Alien Dawg | Detected and Quantified | 0.0221 mg/g dry wt | | details | | Gabriola | Detected and Quantified | 0.00989 mg/g dry wt | | details | | Island Honey | Detected and Quantified | 0.0400 mg/g dry wt | | details | | Quadra | Detected and Quantified | 0.0182 mg/g dry wt | | details | | Sensi Star | Detected and Quantified | 0.0132 mg/g dry wt | | details | | Tangerine Dream | Detected and Quantified | 0.0443 mg/g dry wt | | details |
|
|---|
| External Links |
|---|
| HMDB ID | HMDB0000190 |
|---|
| DrugBank ID | Not Available |
|---|
| Phenol Explorer Compound ID | Not Available |
|---|
| FoodDB ID | FDB003294 |
|---|
| KNApSAcK ID | C00001191 |
|---|
| Chemspider ID | 96860 |
|---|
| KEGG Compound ID | C00186 |
|---|
| BioCyc ID | L-LACTATE |
|---|
| BiGG ID | 34179 |
|---|
| Wikipedia Link | Lactic_Acid |
|---|
| METLIN ID | 5205 |
|---|
| PubChem Compound | 107689 |
|---|
| PDB ID | Not Available |
|---|
| ChEBI ID | 422 |
|---|
| References |
|---|
| General References | Not Available |
|---|

