| Record Information |
|---|
| Version | 1.0 |
|---|
| Created at | 2020-04-17 19:25:34 UTC |
|---|
| Updated at | 2020-11-18 16:39:37 UTC |
|---|
| CannabisDB ID | CDB005262 |
|---|
| Secondary Accession Numbers | Not Available |
|---|
| Cannabis Compound Identification |
|---|
| Common Name | Fructose 1-phosphate |
|---|
| Description | Fructose 1-phosphate, also known as D-fructose-1-p, belongs to the class of organic compounds known as hexose phosphates. These are carbohydrate derivatives containing a hexose substituted by one or more phosphate groups. Fructose 1-phosphate is an extremely weak basic (essentially neutral) compound (based on its pKa). Fructose 1-phosphate exists in all living organisms, ranging from bacteria to humans. Within humans, fructose 1-phosphate participates in a number of enzymatic reactions. In particular, fructose 1-phosphate can be biosynthesized from D-fructose; which is catalyzed by the enzyme ketohexokinase. In addition, fructose 1-phosphate can be converted into dihydroxyacetone phosphate and glyceraldehyde through its interaction with the enzyme fructose-bisphosphate aldolase a. A D-fructofuranose 1-phosphate in which the anomeric centre has beta-configuration. In humans, fructose 1-phosphate is involved in fructose intolerance, hereditary. Outside of the human body, Fructose 1-phosphate has been detected, but not quantified in, milk (cow). This could make fructose 1-phosphate a potential biomarker for the consumption of these foods. Fructose 1-phosphate is expected to be in Cannabis as all living plants are known to produce and metabolize it. |
|---|
| Structure | |
|---|
| Synonyms | | Value | Source |
|---|
| beta-D-Fructose 1-phosphate | ChEBI | | b-D-Fructose 1-phosphate | Generator | | b-D-Fructose 1-phosphoric acid | Generator | | beta-D-Fructose 1-phosphoric acid | Generator | | Β-D-fructose 1-phosphate | Generator | | Β-D-fructose 1-phosphoric acid | Generator | | Fructose 1-phosphoric acid | Generator | | D-Fructose 1-phosphate | HMDB | | D-Fructose-1-p | HMDB | | D-Fructose-1-phosphate | HMDB | | Fructose-1-p | HMDB | | Fructose-1-phosphate | HMDB | | b-D-Fructofuranose 1-phosphate | Generator | | b-D-Fructofuranose 1-phosphoric acid | Generator | | beta-D-Fructofuranose 1-phosphoric acid | Generator | | Β-D-fructofuranose 1-phosphate | Generator | | Β-D-fructofuranose 1-phosphoric acid | Generator |
|
|---|
| Chemical Formula | C6H13O9P |
|---|
| Average Molecular Weight | 260.14 |
|---|
| Monoisotopic Molecular Weight | 260.0297 |
|---|
| IUPAC Name | {[(2R,3S,4S,5R)-2,3,4-trihydroxy-5-(hydroxymethyl)oxolan-2-yl]methoxy}phosphonic acid |
|---|
| Traditional Name | [(2R,3S,4S,5R)-2,3,4-trihydroxy-5-(hydroxymethyl)oxolan-2-yl]methoxyphosphonic acid |
|---|
| CAS Registry Number | 15978-08-2 |
|---|
| SMILES | OC[C@H]1O[C@](O)(COP(O)(O)=O)[C@@H](O)[C@@H]1O |
|---|
| InChI Identifier | InChI=1S/C6H13O9P/c7-1-3-4(8)5(9)6(10,15-3)2-14-16(11,12)13/h3-5,7-10H,1-2H2,(H2,11,12,13)/t3-,4-,5+,6-/m1/s1 |
|---|
| InChI Key | RHKKZBWRNHGJEZ-ARQDHWQXSA-N |
|---|
| Chemical Taxonomy |
|---|
| Description | Belongs to the class of organic compounds known as hexose phosphates. These are carbohydrate derivatives containing a hexose substituted by one or more phosphate groups. |
|---|
| Kingdom | Organic compounds |
|---|
| Super Class | Organic oxygen compounds |
|---|
| Class | Organooxygen compounds |
|---|
| Sub Class | Carbohydrates and carbohydrate conjugates |
|---|
| Direct Parent | Hexose phosphates |
|---|
| Alternative Parents | |
|---|
| Substituents | - Hexose phosphate
- Pentose phosphate
- Pentose-5-phosphate
- C-glycosyl compound
- Glycosyl compound
- Monosaccharide phosphate
- Pentose monosaccharide
- Monoalkyl phosphate
- Organic phosphoric acid derivative
- Alkyl phosphate
- Phosphoric acid ester
- Tetrahydrofuran
- Secondary alcohol
- Hemiacetal
- Organoheterocyclic compound
- Oxacycle
- Polyol
- Alcohol
- Hydrocarbon derivative
- Organic oxide
- Primary alcohol
- Aliphatic heteromonocyclic compound
|
|---|
| Molecular Framework | Aliphatic heteromonocyclic compounds |
|---|
| External Descriptors | Not Available |
|---|
| Ontology |
|---|
|
| Disposition | Source: Biological location: |
|---|
| Physical Properties |
|---|
| State | Solid |
|---|
| Experimental Properties | | Property | Value | Reference |
|---|
| Melting Point | Not Available | Not Available | | Boiling Point | Not Available | Not Available | | Water Solubility | Not Available | Not Available | | logP | Not Available | Not Available |
|
|---|
| Predicted Properties | [] |
|---|
| Spectra |
|---|
| EI-MS/GC-MS | | Type | Description | Splash Key | View |
|---|
| Predicted GC-MS | Fructose 1-phosphate, non-derivatized, Predicted GC-MS Spectrum - 70eV, Positive | splash10-03di-9510000000-71d828159b61f8342118 | Spectrum | | Predicted GC-MS | Fructose 1-phosphate, 4 TMS, Predicted GC-MS Spectrum - 70eV, Positive | splash10-001i-2912320000-08ffe3d23f5fa5d6162c | Spectrum | | Predicted GC-MS | Fructose 1-phosphate, non-derivatized, Predicted GC-MS Spectrum - 70eV, Positive | Not Available | Spectrum | | Predicted GC-MS | Fructose 1-phosphate, non-derivatized, Predicted GC-MS Spectrum - 70eV, Positive | Not Available | Spectrum |
|
|---|
| MS/MS | | Type | Description | Splash Key | View |
|---|
| Predicted MS/MS | Predicted LC-MS/MS Spectrum - 10V, Positive | splash10-03dm-9470000000-18ac582a370338a3f5f1 | 2015-09-15 | View Spectrum | | Predicted MS/MS | Predicted LC-MS/MS Spectrum - 20V, Positive | splash10-01oy-7390000000-1a5a72af8bb734e50eb8 | 2015-09-15 | View Spectrum | | Predicted MS/MS | Predicted LC-MS/MS Spectrum - 40V, Positive | splash10-009y-9100000000-4f151f373d7730140f38 | 2015-09-15 | View Spectrum | | Predicted MS/MS | Predicted LC-MS/MS Spectrum - 10V, Negative | splash10-0a6r-9270000000-5af91634007091008210 | 2015-09-15 | View Spectrum | | Predicted MS/MS | Predicted LC-MS/MS Spectrum - 20V, Negative | splash10-004i-9000000000-5cf3f457b8c6f6270b68 | 2015-09-15 | View Spectrum | | Predicted MS/MS | Predicted LC-MS/MS Spectrum - 40V, Negative | splash10-004i-9000000000-4e6feccf18943de8c419 | 2015-09-15 | View Spectrum | | Predicted MS/MS | Predicted LC-MS/MS Spectrum - 10V, Negative | splash10-0a4i-1090000000-03030e5df4374fea160c | 2021-09-21 | View Spectrum | | Predicted MS/MS | Predicted LC-MS/MS Spectrum - 20V, Negative | splash10-004j-9140000000-be637d8fd4ff371117c8 | 2021-09-21 | View Spectrum | | Predicted MS/MS | Predicted LC-MS/MS Spectrum - 40V, Negative | splash10-004i-9010000000-26f483248da003aafbdb | 2021-09-21 | View Spectrum | | Predicted MS/MS | Predicted LC-MS/MS Spectrum - 10V, Positive | splash10-03di-0190000000-6beaa046be46044471be | 2021-09-25 | View Spectrum | | Predicted MS/MS | Predicted LC-MS/MS Spectrum - 20V, Positive | splash10-01p2-6920000000-7fd7dcf4a4f1479d91b1 | 2021-09-25 | View Spectrum | | Predicted MS/MS | Predicted LC-MS/MS Spectrum - 40V, Positive | splash10-052b-9100000000-60f4d9de70c1b7a7e1de | 2021-09-25 | View Spectrum |
|
|---|
| NMR | Not Available |
|---|
| Pathways |
|---|
| Pathways | | Name | SMPDB/Pathwhiz | KEGG | | Fructose and Mannose Degradation | 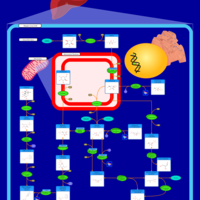 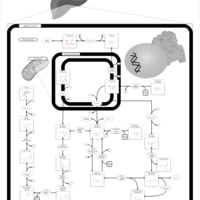 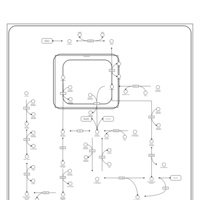 | 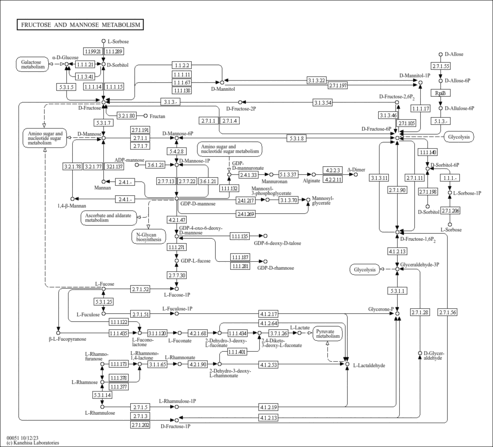 | | Fructosuria | 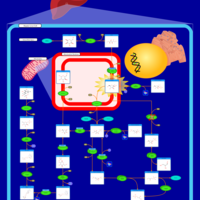 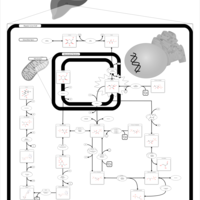 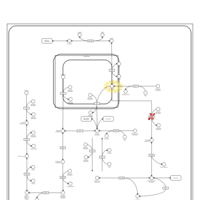 | Not Available | | Fructose intolerance, hereditary | 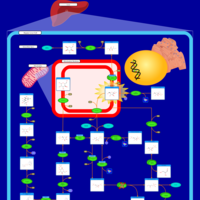 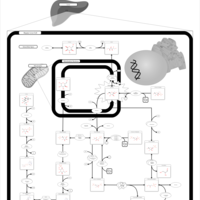 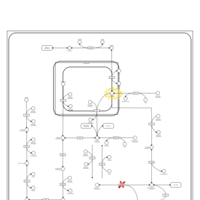 | Not Available |
|
|---|
| Protein Targets |
|---|
| Enzymes | |
|---|
| Transporters | Not Available |
|---|
| Metal Bindings | Not Available |
|---|
| Receptors | Not Available |
|---|
| Transcriptional Factors | Not Available |
|---|
| Concentrations Data |
|---|
| Not Available |
|---|
| External Links |
|---|
| HMDB ID | HMDB0001076 |
|---|
| DrugBank ID | Not Available |
|---|
| Phenol Explorer Compound ID | Not Available |
|---|
| FoodDB ID | FDB022410 |
|---|
| KNApSAcK ID | Not Available |
|---|
| Chemspider ID | 8575807 |
|---|
| KEGG Compound ID | C01094 |
|---|
| BioCyc ID | FRU1P |
|---|
| BiGG ID | 40936 |
|---|
| Wikipedia Link | Fructose 1-phosphate |
|---|
| METLIN ID | 378 |
|---|
| PubChem Compound | 10400369 |
|---|
| PDB ID | Not Available |
|---|
| ChEBI ID | 139419 |
|---|
| References |
|---|
| General References | Not Available |
|---|
