| Record Information |
|---|
| Version | 1.0 |
|---|
| Created at | 2020-04-17 19:13:40 UTC |
|---|
| Updated at | 2020-12-07 19:11:40 UTC |
|---|
| CannabisDB ID | CDB005143 |
|---|
| Secondary Accession Numbers | Not Available |
|---|
| Cannabis Compound Identification |
|---|
| Common Name | L-Cystathionine |
|---|
| Description | L-Cystathionine belongs to the class of organic compounds known as l-cysteine-s-conjugates. L-cysteine-S-conjugates are compounds containing L-cysteine where the thio-group is conjugated. L-Cystathionine is a very strong basic compound (based on its pKa). L-Cystathionine exists in all living species, ranging from bacteria to humans. Within humans, L-cystathionine participates in a number of enzymatic reactions. In particular, L-cystathionine can be converted into L-cysteine and 2-ketobutyric acid; which is mediated by the enzyme cystathionine gamma-lyase. In addition, L-cystathionine can be biosynthesized from L-homoserine and L-serine through its interaction with the enzyme cystathionine beta-synthase. In humans, L-cystathionine is involved in methionine metabolism. L-Cystathionine is a potentially toxic compound. L-Cystathionine, with regard to humans, has been found to be associated with several diseases such as eosinophilic esophagitis, alzheimer's disease, and autism; L-cystathionine has also been linked to several inborn metabolic disorders including hypermethioninemia and folate deficiency. A modified amino acid generated by enzymic means from L-homocysteine and L-serine. L-Cystathionine is expected to be in Cannabis as all living plants are known to produce and metabolize it. |
|---|
| Structure | |
|---|
| Synonyms | | Value | Source |
|---|
| (R)-S-(2-Amino-2-carboxyethyl)-L-homocysteine | ChEBI | | L-(+)-Cystathionine | ChEBI | | S-(beta-Amino-beta-carboxyethyl)homocysteine | ChEBI | | S-(b-Amino-b-carboxyethyl)homocysteine | Generator | | S-(Β-amino-β-carboxyethyl)homocysteine | Generator | | Cystathionine | HMDB | | S-[(2R)-2-Amino-2-carboxyethyl]-L-homocysteine | HMDB | | [R-(R*,s*)]-2-amino-4-[(2-amino-2-carboxyethyl)thio]-butanoate | HMDB | | [R-(R*,s*)]-2-amino-4-[(2-amino-2-carboxyethyl)thio]-butanoic acid | HMDB |
|
|---|
| Chemical Formula | C7H14N2O4S |
|---|
| Average Molecular Weight | 222.26 |
|---|
| Monoisotopic Molecular Weight | 222.0674 |
|---|
| IUPAC Name | (2S)-2-amino-4-{[(2R)-2-amino-2-carboxyethyl]sulfanyl}butanoic acid |
|---|
| Traditional Name | L-cystathionine |
|---|
| CAS Registry Number | 56-88-2 |
|---|
| SMILES | N[C@@H](CCSC[C@H](N)C(O)=O)C(O)=O |
|---|
| InChI Identifier | InChI=1S/C7H14N2O4S/c8-4(6(10)11)1-2-14-3-5(9)7(12)13/h4-5H,1-3,8-9H2,(H,10,11)(H,12,13)/t4-,5-/m0/s1 |
|---|
| InChI Key | ILRYLPWNYFXEMH-WHFBIAKZSA-N |
|---|
| Chemical Taxonomy |
|---|
| Description | Belongs to the class of organic compounds known as l-cysteine-s-conjugates. L-cysteine-S-conjugates are compounds containing L-cysteine where the thio-group is conjugated. |
|---|
| Kingdom | Organic compounds |
|---|
| Super Class | Organic acids and derivatives |
|---|
| Class | Carboxylic acids and derivatives |
|---|
| Sub Class | Amino acids, peptides, and analogues |
|---|
| Direct Parent | L-cysteine-S-conjugates |
|---|
| Alternative Parents | |
|---|
| Substituents | - L-cysteine-s-conjugate
- Alpha-amino acid
- L-alpha-amino acid
- Thia fatty acid
- Dicarboxylic acid or derivatives
- Fatty acyl
- Fatty acid
- Amino acid
- Carboxylic acid
- Thioether
- Dialkylthioether
- Sulfenyl compound
- Organic nitrogen compound
- Hydrocarbon derivative
- Organic oxide
- Primary amine
- Organosulfur compound
- Organooxygen compound
- Organonitrogen compound
- Primary aliphatic amine
- Organopnictogen compound
- Organic oxygen compound
- Carbonyl group
- Amine
- Aliphatic acyclic compound
|
|---|
| Molecular Framework | Aliphatic acyclic compounds |
|---|
| External Descriptors | |
|---|
| Ontology |
|---|
|
| Physiological effect | Health effect: |
|---|
| Disposition | Route of exposure: Source: Biological location: |
|---|
| Role | Biological role: |
|---|
| Physical Properties |
|---|
| State | Solid |
|---|
| Experimental Properties | | Property | Value | Reference |
|---|
| Melting Point | 312 °C | Not Available | | Boiling Point | Not Available | Not Available | | Water Solubility | 22 mg/mL | Not Available | | logP | Not Available | Not Available |
|
|---|
| Predicted Properties | [] |
|---|
| Spectra |
|---|
| EI-MS/GC-MS | | Type | Description | Splash Key | View |
|---|
| GC-MS | L-Cystathionine, 4 TMS, GC-MS Spectrum | splash10-00fr-8950000000-640b4294e37e5a01048d | Spectrum | | GC-MS | L-Cystathionine, 2 TMS, GC-MS Spectrum | splash10-0f7c-7910000000-91f9f2b2dd44a3cdb276 | Spectrum | | GC-MS | L-Cystathionine, 3 TMS, GC-MS Spectrum | splash10-00or-1940000000-c0d688e4ea87ff3e03fa | Spectrum | | GC-MS | L-Cystathionine, 4 TMS, GC-MS Spectrum | splash10-00or-0980000000-7d816de782a902900d0e | Spectrum | | GC-MS | L-Cystathionine, non-derivatized, GC-MS Spectrum | splash10-00fr-8950000000-640b4294e37e5a01048d | Spectrum | | GC-MS | L-Cystathionine, non-derivatized, GC-MS Spectrum | splash10-0f7c-7910000000-91f9f2b2dd44a3cdb276 | Spectrum | | GC-MS | L-Cystathionine, non-derivatized, GC-MS Spectrum | splash10-00or-1940000000-c0d688e4ea87ff3e03fa | Spectrum | | GC-MS | L-Cystathionine, non-derivatized, GC-MS Spectrum | splash10-00or-0980000000-7d816de782a902900d0e | Spectrum | | Predicted GC-MS | L-Cystathionine, non-derivatized, Predicted GC-MS Spectrum - 70eV, Positive | splash10-00di-9410000000-9487e9168e86a2297931 | Spectrum | | Predicted GC-MS | L-Cystathionine, 2 TMS, Predicted GC-MS Spectrum - 70eV, Positive | splash10-00dm-9665000000-608a6335880ad1c2519d | Spectrum | | Predicted GC-MS | L-Cystathionine, non-derivatized, Predicted GC-MS Spectrum - 70eV, Positive | Not Available | Spectrum | | Predicted GC-MS | L-Cystathionine, TMS_1_1, Predicted GC-MS Spectrum - 70eV, Positive | Not Available | Spectrum | | Predicted GC-MS | L-Cystathionine, TMS_1_2, Predicted GC-MS Spectrum - 70eV, Positive | Not Available | Spectrum | | Predicted GC-MS | L-Cystathionine, TMS_1_3, Predicted GC-MS Spectrum - 70eV, Positive | Not Available | Spectrum | | Predicted GC-MS | L-Cystathionine, TMS_1_4, Predicted GC-MS Spectrum - 70eV, Positive | Not Available | Spectrum | | Predicted GC-MS | L-Cystathionine, TMS_2_2, Predicted GC-MS Spectrum - 70eV, Positive | Not Available | Spectrum | | Predicted GC-MS | L-Cystathionine, TMS_2_3, Predicted GC-MS Spectrum - 70eV, Positive | Not Available | Spectrum | | Predicted GC-MS | L-Cystathionine, TMS_2_4, Predicted GC-MS Spectrum - 70eV, Positive | Not Available | Spectrum | | Predicted GC-MS | L-Cystathionine, TMS_2_5, Predicted GC-MS Spectrum - 70eV, Positive | Not Available | Spectrum | | Predicted GC-MS | L-Cystathionine, TMS_2_6, Predicted GC-MS Spectrum - 70eV, Positive | Not Available | Spectrum | | Predicted GC-MS | L-Cystathionine, TMS_2_7, Predicted GC-MS Spectrum - 70eV, Positive | Not Available | Spectrum | | Predicted GC-MS | L-Cystathionine, TMS_2_8, Predicted GC-MS Spectrum - 70eV, Positive | Not Available | Spectrum | | Predicted GC-MS | L-Cystathionine, TBDMS_1_1, Predicted GC-MS Spectrum - 70eV, Positive | Not Available | Spectrum | | Predicted GC-MS | L-Cystathionine, TBDMS_1_2, Predicted GC-MS Spectrum - 70eV, Positive | Not Available | Spectrum | | Predicted GC-MS | L-Cystathionine, TBDMS_1_3, Predicted GC-MS Spectrum - 70eV, Positive | Not Available | Spectrum |
|
|---|
| MS/MS | | Type | Description | Splash Key | View |
|---|
| MS/MS | LC-MS/MS Spectrum - Quattro_QQQ 10V, Positive (Annotated) | splash10-0089-1980000000-99cb7d1da067e5436b09 | 2012-07-24 | View Spectrum | | MS/MS | LC-MS/MS Spectrum - Quattro_QQQ 25V, Positive (Annotated) | splash10-000i-9200000000-fb776970b6ef36e72725 | 2012-07-24 | View Spectrum | | MS/MS | LC-MS/MS Spectrum - Quattro_QQQ 40V, Positive (Annotated) | splash10-000i-9200000000-fb776970b6ef36e72725 | 2012-07-24 | View Spectrum | | MS/MS | LC-MS/MS Spectrum - LC-ESI-QQ (API3000, Applied Biosystems) 10V, Positive | splash10-00fr-0690000000-0b572de3fcbc7e2f13cc | 2012-08-31 | View Spectrum | | MS/MS | LC-MS/MS Spectrum - LC-ESI-QQ (API3000, Applied Biosystems) 20V, Positive | splash10-000t-0900000000-5788192b32aadd177e33 | 2012-08-31 | View Spectrum | | MS/MS | LC-MS/MS Spectrum - LC-ESI-QQ (API3000, Applied Biosystems) 30V, Positive | splash10-000b-4900000000-b86ac7de446a762365bd | 2012-08-31 | View Spectrum | | MS/MS | LC-MS/MS Spectrum - LC-ESI-QQ (API3000, Applied Biosystems) 40V, Positive | splash10-000j-9800000000-75ff1bc6e325692a9da9 | 2012-08-31 | View Spectrum | | MS/MS | LC-MS/MS Spectrum - LC-ESI-QQ (API3000, Applied Biosystems) 50V, Positive | splash10-00kr-9100000000-6517562dc0f31a85afc8 | 2012-08-31 | View Spectrum | | MS/MS | LC-MS/MS Spectrum - LC-ESI-IT (LC/MSD Trap XCT, Agilent Technologies) , Positive | splash10-001i-0900000000-d1628d0a880f1b6e6d90 | 2012-08-31 | View Spectrum | | MS/MS | LC-MS/MS Spectrum - LC-ESI-IT (LC/MSD Trap XCT, Agilent Technologies) , Positive | splash10-000i-9000000000-9a2e1e54b77216e2df72 | 2012-08-31 | View Spectrum | | MS/MS | LC-MS/MS Spectrum - LC-ESI-QTOF (UPLC Q-Tof Premier, Waters) , Positive | splash10-00di-0090000000-36678c2f0bf1b01cc9d4 | 2012-08-31 | View Spectrum | | MS/MS | LC-MS/MS Spectrum - LC-ESI-QTOF (UPLC Q-Tof Premier, Waters) 30V, Positive | splash10-05fr-0190000000-e575f57f0d76ef6afa49 | 2012-08-31 | View Spectrum | | MS/MS | LC-MS/MS Spectrum - LC-ESI-QTOF (UPLC Q-Tof Premier, Waters) , Negative | splash10-00e9-0940000000-962ae510f06296b98878 | 2012-08-31 | View Spectrum | | MS/MS | LC-MS/MS Spectrum - LC-ESI-QTOF , negative | splash10-00e9-0940000000-27da1912eac54f485bfa | 2017-09-14 | View Spectrum | | MS/MS | LC-MS/MS Spectrum - LC-ESI-QQ , positive | splash10-00fr-0690000000-82fe2198c98660008c04 | 2017-09-14 | View Spectrum | | MS/MS | LC-MS/MS Spectrum - LC-ESI-QQ , positive | splash10-000t-0900000000-58cceb69cceeff79606c | 2017-09-14 | View Spectrum | | MS/MS | LC-MS/MS Spectrum - LC-ESI-QQ , positive | splash10-000b-4900000000-5877787c54120dc165c9 | 2017-09-14 | View Spectrum | | MS/MS | LC-MS/MS Spectrum - LC-ESI-QQ , positive | splash10-000j-9800000000-fd29b0fcf62b73831829 | 2017-09-14 | View Spectrum | | MS/MS | LC-MS/MS Spectrum - LC-ESI-QQ , positive | splash10-00kr-9100000000-6517562dc0f31a85afc8 | 2017-09-14 | View Spectrum | | Predicted MS/MS | Predicted LC-MS/MS Spectrum - 10V, Positive | splash10-05di-1930000000-887e3ad7da04e4f1aac9 | 2016-09-12 | View Spectrum | | Predicted MS/MS | Predicted LC-MS/MS Spectrum - 20V, Positive | splash10-01uc-6900000000-563444ac8ab717997268 | 2016-09-12 | View Spectrum | | Predicted MS/MS | Predicted LC-MS/MS Spectrum - 40V, Positive | splash10-0uki-9700000000-35397d8e2ab2c6582c52 | 2016-09-12 | View Spectrum | | Predicted MS/MS | Predicted LC-MS/MS Spectrum - 10V, Negative | splash10-00di-0970000000-d0f0a96ca41d1f751c26 | 2016-09-12 | View Spectrum | | Predicted MS/MS | Predicted LC-MS/MS Spectrum - 20V, Negative | splash10-0089-3910000000-22200c974305822456d8 | 2016-09-12 | View Spectrum | | Predicted MS/MS | Predicted LC-MS/MS Spectrum - 40V, Negative | splash10-008l-9300000000-0dfaab5efe3438889c3c | 2016-09-12 | View Spectrum |
|
|---|
| NMR | | Type | Description | | View |
|---|
| 1D NMR | 1H NMR Spectrum (1D, 500 MHz, H2O, experimental) | | Spectrum | | 1D NMR | 1H NMR Spectrum (1D, D2O, experimental) | | Spectrum | | 1D NMR | 13C NMR Spectrum (1D, D2O, experimental) | | Spectrum | | 1D NMR | 1H NMR Spectrum (1D, 100 MHz, D2O, predicted) | | Spectrum | | 1D NMR | 13C NMR Spectrum (1D, 100 MHz, D2O, predicted) | | Spectrum | | 1D NMR | 1H NMR Spectrum (1D, 1000 MHz, D2O, predicted) | | Spectrum | | 1D NMR | 13C NMR Spectrum (1D, 1000 MHz, D2O, predicted) | | Spectrum | | 1D NMR | 1H NMR Spectrum (1D, 200 MHz, D2O, predicted) | | Spectrum | | 1D NMR | 13C NMR Spectrum (1D, 200 MHz, D2O, predicted) | | Spectrum | | 1D NMR | 1H NMR Spectrum (1D, 300 MHz, D2O, predicted) | | Spectrum | | 1D NMR | 13C NMR Spectrum (1D, 300 MHz, D2O, predicted) | | Spectrum | | 1D NMR | 1H NMR Spectrum (1D, 400 MHz, D2O, predicted) | | Spectrum | | 1D NMR | 13C NMR Spectrum (1D, 400 MHz, D2O, predicted) | | Spectrum | | 1D NMR | 1H NMR Spectrum (1D, 500 MHz, D2O, predicted) | | Spectrum | | 1D NMR | 13C NMR Spectrum (1D, 500 MHz, D2O, predicted) | | Spectrum | | 1D NMR | 1H NMR Spectrum (1D, 600 MHz, D2O, predicted) | | Spectrum | | 1D NMR | 13C NMR Spectrum (1D, 600 MHz, D2O, predicted) | | Spectrum | | 1D NMR | 1H NMR Spectrum (1D, 700 MHz, D2O, predicted) | | Spectrum | | 1D NMR | 13C NMR Spectrum (1D, 700 MHz, D2O, predicted) | | Spectrum | | 1D NMR | 1H NMR Spectrum (1D, 800 MHz, D2O, predicted) | | Spectrum | | 1D NMR | 13C NMR Spectrum (1D, 800 MHz, D2O, predicted) | | Spectrum | | 1D NMR | 1H NMR Spectrum (1D, 900 MHz, D2O, predicted) | | Spectrum | | 1D NMR | 13C NMR Spectrum (1D, 900 MHz, D2O, predicted) | | Spectrum | | 2D NMR | [1H, 13C]-HSQC NMR Spectrum (2D, 600 MHz, H2O, experimental) | | Spectrum |
|
|---|
| Pathways |
|---|
| Pathways | | Name | SMPDB/Pathwhiz | KEGG | | Glycine and Serine Metabolism | 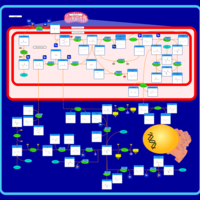 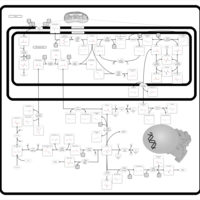 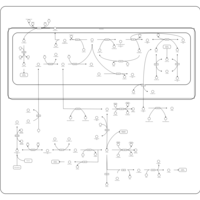 |  | | Methionine Metabolism | 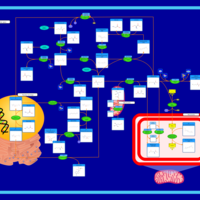 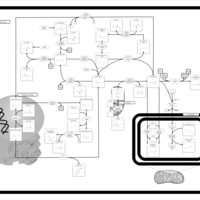 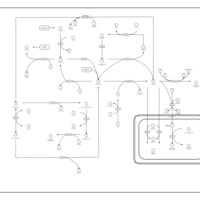 |  | | Homocysteine Degradation |   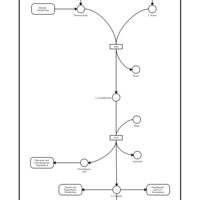 | Not Available | | Gamma-cystathionase deficiency (CTH) | 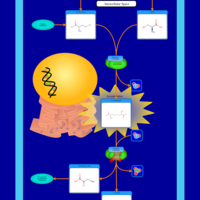 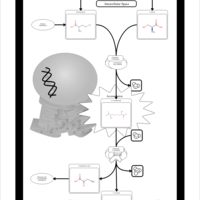 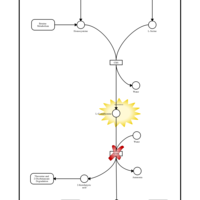 | Not Available | | Homocystinuria, cystathionine beta-synthase deficiency | 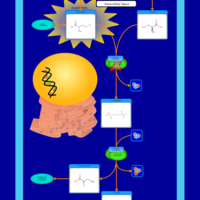   | Not Available |
|
|---|
| Protein Targets |
|---|
| Enzymes | |
|---|
| Transporters | Not Available |
|---|
| Metal Bindings | |
|---|
| Receptors | Not Available |
|---|
| Transcriptional Factors | |
|---|
| Concentrations Data |
|---|
| Not Available |
|---|
| External Links |
|---|
| HMDB ID | HMDB0000099 |
|---|
| DrugBank ID | Not Available |
|---|
| Phenol Explorer Compound ID | Not Available |
|---|
| FoodDB ID | FDB001976 |
|---|
| KNApSAcK ID | C00007498 |
|---|
| Chemspider ID | 388392 |
|---|
| KEGG Compound ID | C02291 |
|---|
| BioCyc ID | L-CYSTATHIONINE |
|---|
| BiGG ID | 39523 |
|---|
| Wikipedia Link | Cystathionine |
|---|
| METLIN ID | 39 |
|---|
| PubChem Compound | 439258 |
|---|
| PDB ID | Not Available |
|---|
| ChEBI ID | 17482 |
|---|
| References |
|---|
| General References | Not Available |
|---|

