| Record Information |
|---|
| Version | 1.0 |
|---|
| Created at | 2020-04-17 18:36:50 UTC |
|---|
| Updated at | 2022-12-13 23:36:26 UTC |
|---|
| CannabisDB ID | CDB004788 |
|---|
| Secondary Accession Numbers | Not Available |
|---|
| Cannabis Compound Identification |
|---|
| Common Name | Betaine |
|---|
| Description | Betaine, also known as Bet or trimethylglycine, belongs to the class of organic compounds known as alpha amino acids. These are amino acids in which the amino group is attached to the carbon atom immediately adjacent to the carboxylate group (alpha carbon). Betaine is a moderately acidic compound (based on its pKa). Betaine exists in all eukaryotes, ranging from yeast to humans. In humans, betaine is involved in betaine metabolism. Betaine is a bland tasting compound. Outside of the human body, Betaine is found, on average, in the highest concentration within a few different foods, such as swamp cabbages, quinoa, and lambsquarters and in a lower concentration in common buckwheats, burdocks, and yellow zucchinis. Betaine has also been detected, but not quantified in, several different foods, such as lemon verbena, wasabis, endives, chickpea, and pomegranates. This could make betaine a potential biomarker for the consumption of these foods. Intracellular accumulation of betaines permits water retention in cells, thus protecting from the effects of dehydration (Wikipedia ). Betaine comes from either the diet or by the oxidation of choline. Betaine, with regard to humans, has been found to be associated with several diseases such as chronic renal failure, colorectal cancer, and alzheimer's disease; betaine has also been linked to several inborn metabolic disorders including argininosuccinic aciduria and propionic acidemia. In an acute toxicology study in rats, death frequently occurred at doses equal to or greater than 10,000 mg/kg. Betaine has been shown to have an inhibitory effect on NO release in activated microglial cells and may be an effective therapeutic component to control neurological disorders (PMID: 22801281 ). Betaine insufficiency is associated with metabolic syndrome, lipid disorders, and diabetes, and may have a role in vascular and other diseases (PMID: 20346934 ). Many reports have shown that betaine's therapeutic effectiveness is limited, and does not lower tHcy levels or prevent clinical symptoms . Betaine is expected to be in Cannabis as all living plants are known to produce and metabolize it. |
|---|
| Structure | |
|---|
| Synonyms | | Value | Source |
|---|
| (Trimethylammoniumyl)acetate | ChEBI | | 1-Carboxy-N,N,N-trimethylmethanaminium inner salt | ChEBI | | 2-N,N,N-Trimethylammonio acetate | ChEBI | | Abromine | ChEBI | | Acidol | ChEBI | | Bet | ChEBI | | N,N,N-Trimethylammonioacetate | ChEBI | | N,N,N-Trimethylglycine | ChEBI | | Trimethylaminoacetate | ChEBI | | Trimethylammonioacetate | ChEBI | | Trimethylglycine | ChEBI | | Trimethylglycocoll | ChEBI | | Glycine betaine | Kegg | | Cystadane | Kegg | | (Trimethylammoniumyl)acetic acid | Generator | | 2-N,N,N-Trimethylammonio acetic acid | Generator | | N,N,N-Trimethylammonioacetic acid | Generator | | Trimethylaminoacetic acid | Generator | | Trimethylammonioacetic acid | Generator | | (Carboxymethyl)trimethylammonium hydroxide inner salt | HMDB | | (Trimethylammonio)acetate | HMDB | | 1-Carboxy-N,N,N-trimethyl-methanaminium | HMDB | | 1-Carboxy-N,N,N-trimethyl-methanaminium hydroxide | HMDB | | a-Earleine | HMDB | | alpha-Earleine | HMDB | | Aminocoat | HMDB | | Betafin | HMDB | | Betafin BCR | HMDB | | Betafin BP | HMDB | | Ektasolve ee | HMDB | | FinnStim | HMDB | | Glycocoll betaine | HMDB | | Glycylbetaine | HMDB | | Greenstim | HMDB | | Loramine amb 13 | HMDB | | Loramine amb-13 | HMDB | | Lycine | HMDB | | Oxyneurine | HMDB | | Rubrine C | HMDB | | Trimethylbetaine glycine | HMDB | | Acidin pepsin | HMDB | | Beaufour brand OF betaine citrate | HMDB | | Byk brand OF betaine phosphate | HMDB | | Fournier brand OF betaine ascorbate and hydrate | HMDB | | Hydrochloride, betaine | HMDB | | Logeais brand OF betaine cyclobutyrate | HMDB | | Novobetaine | HMDB | | Betaine hydrochloride | HMDB | | Betaine orphan brand | HMDB | | Boizot brand OF betaine aspartate | HMDB | | C.B.B. | HMDB | | Citrate de bétaïne upsa | HMDB | | Scorbo-bétaïne | HMDB | | Stea-16 | HMDB | | Stea16 | HMDB | | Acidin-pepsin | HMDB | | Citrate de bétaïne beaufour | HMDB | | Orphan brand OF betaine | HMDB | | Scorbo bétaïne | HMDB | | AcidinPepsin | HMDB | | Betaine, glycine | HMDB | | Hepastyl | HMDB | | Scorbobétaïne | HMDB | | UPSA brand OF betaine citrate | HMDB | | Stea 16 | HMDB | | Betaine | ChEBI |
|
|---|
| Chemical Formula | C5H11NO2 |
|---|
| Average Molecular Weight | 117.15 |
|---|
| Monoisotopic Molecular Weight | 117.079 |
|---|
| IUPAC Name | 2-(trimethylazaniumyl)acetate |
|---|
| Traditional Name | (trimethylammonio)acetate |
|---|
| CAS Registry Number | 107-43-7 |
|---|
| SMILES | C[N+](C)(C)CC([O-])=O |
|---|
| InChI Identifier | InChI=1S/C5H11NO2/c1-6(2,3)4-5(7)8/h4H2,1-3H3 |
|---|
| InChI Key | KWIUHFFTVRNATP-UHFFFAOYSA-N |
|---|
| Chemical Taxonomy |
|---|
| Description | Belongs to the class of organic compounds known as alpha amino acids. These are amino acids in which the amino group is attached to the carbon atom immediately adjacent to the carboxylate group (alpha carbon). |
|---|
| Kingdom | Organic compounds |
|---|
| Super Class | Organic acids and derivatives |
|---|
| Class | Carboxylic acids and derivatives |
|---|
| Sub Class | Amino acids, peptides, and analogues |
|---|
| Direct Parent | Alpha amino acids |
|---|
| Alternative Parents | |
|---|
| Substituents | - Alpha-amino acid
- Quaternary ammonium salt
- Tetraalkylammonium salt
- Carboxylic acid salt
- Monocarboxylic acid or derivatives
- Carboxylic acid
- Organopnictogen compound
- Hydrocarbon derivative
- Organic salt
- Organic oxygen compound
- Organooxygen compound
- Organonitrogen compound
- Carbonyl group
- Amine
- Organic nitrogen compound
- Organic oxide
- Aliphatic acyclic compound
|
|---|
| Molecular Framework | Aliphatic acyclic compounds |
|---|
| External Descriptors | |
|---|
| Ontology |
|---|
|
| Physiological effect | Health effect: |
|---|
| Disposition | Route of exposure: Source: Biological location: |
|---|
| Role | Industrial application: Biological role: |
|---|
| Physical Properties |
|---|
| State | Solid |
|---|
| Experimental Properties | | Property | Value | Reference |
|---|
| Melting Point | 293 - 301 °C | Not Available | | Boiling Point | Not Available | Not Available | | Water Solubility | 611 mg/mL at 19 °C | Not Available | | logP | Not Available | Not Available |
|
|---|
| Predicted Properties | [] |
|---|
| Spectra |
|---|
| EI-MS/GC-MS | | Type | Description | Splash Key | View |
|---|
| EI-MS | Mass Spectrum (Electron Ionization) | splash10-0a4l-9000000000-f32989f795eeceb8b692 | 2015-03-01 | View Spectrum | | Predicted GC-MS | Betaine, non-derivatized, Predicted GC-MS Spectrum - 70eV, Positive | splash10-05fu-9200000000-96ab6e2136fe7e03b63a | Spectrum |
|
|---|
| MS/MS | | Type | Description | Splash Key | View |
|---|
| MS/MS | LC-MS/MS Spectrum - Quattro_QQQ 10V, Positive (Annotated) | splash10-014i-0900000000-feacc600820771d2a77b | 2012-07-24 | View Spectrum | | MS/MS | LC-MS/MS Spectrum - Quattro_QQQ 25V, Positive (Annotated) | splash10-0a4i-9000000000-e5bbda9fb66994576062 | 2012-07-24 | View Spectrum | | MS/MS | LC-MS/MS Spectrum - Quattro_QQQ 40V, Positive (Annotated) | splash10-0a4i-9000000000-c382759eab7d0a87ac27 | 2012-07-24 | View Spectrum | | MS/MS | LC-MS/MS Spectrum - LC-ESI-QQ (API3000, Applied Biosystems) 10V, Positive | splash10-014i-0900000000-a7beb42e6944181ae5d9 | 2012-08-31 | View Spectrum | | MS/MS | LC-MS/MS Spectrum - LC-ESI-QQ (API3000, Applied Biosystems) 20V, Positive | splash10-014i-1900000000-77fbc4a2b76030d53644 | 2012-08-31 | View Spectrum | | MS/MS | LC-MS/MS Spectrum - LC-ESI-QQ (API3000, Applied Biosystems) 30V, Positive | splash10-0a4i-9100000000-2941e4997f202293bf71 | 2012-08-31 | View Spectrum | | MS/MS | LC-MS/MS Spectrum - LC-ESI-QQ (API3000, Applied Biosystems) 40V, Positive | splash10-0a4i-9000000000-9b9e3cecb77ed4b40b7c | 2012-08-31 | View Spectrum | | MS/MS | LC-MS/MS Spectrum - LC-ESI-QQ (API3000, Applied Biosystems) 50V, Positive | splash10-0a4i-9000000000-27bbb5f9642306216a0b | 2012-08-31 | View Spectrum | | MS/MS | LC-MS/MS Spectrum - LC-ESI-QTOF (UPLC Q-Tof Premier, Waters) , Positive | splash10-014i-1900000000-82a41cbb0f697910b577 | 2012-08-31 | View Spectrum | | MS/MS | LC-MS/MS Spectrum - LC-ESI-QTOF (UPLC Q-Tof Premier, Waters) 30V, Positive | splash10-0aor-9500000000-9a0f1853188052d798c6 | 2012-08-31 | View Spectrum | | MS/MS | LC-MS/MS Spectrum - LC-ESI-QQ , positive | splash10-066r-5900000000-a6291f86046f32ad3c81 | 2017-09-14 | View Spectrum | | MS/MS | LC-MS/MS Spectrum - LC-ESI-QQ , positive | splash10-0a4i-9200000000-709208d6782f1b588501 | 2017-09-14 | View Spectrum | | MS/MS | LC-MS/MS Spectrum - LC-ESI-QQ , positive | splash10-014i-0900000000-a7beb42e6944181ae5d9 | 2017-09-14 | View Spectrum | | MS/MS | LC-MS/MS Spectrum - LC-ESI-QQ , positive | splash10-014i-1900000000-77fbc4a2b76030d53644 | 2017-09-14 | View Spectrum | | MS/MS | LC-MS/MS Spectrum - LC-ESI-QQ , positive | splash10-0a4i-9100000000-2941e4997f202293bf71 | 2017-09-14 | View Spectrum | | MS/MS | LC-MS/MS Spectrum - LC-ESI-QQ , positive | splash10-0a4i-9000000000-a2560fc2add75f79eadb | 2017-09-14 | View Spectrum | | MS/MS | LC-MS/MS Spectrum - LC-ESI-QQ , positive | splash10-0a4i-9000000000-27bbb5f9642306216a0b | 2017-09-14 | View Spectrum | | MS/MS | LC-MS/MS Spectrum - LC-ESI-ITFT , positive | splash10-014i-0900000000-b45353825d98488d752a | 2017-09-14 | View Spectrum | | MS/MS | LC-MS/MS Spectrum - LC-ESI-IT , positive | splash10-0aor-9500000000-3fdd9d17a169db5c58dd | 2017-09-14 | View Spectrum | | Predicted MS/MS | Predicted LC-MS/MS Spectrum - 10V, Positive | splash10-014i-0900000000-493791dfdd428fc3bd6b | 2017-09-01 | View Spectrum | | Predicted MS/MS | Predicted LC-MS/MS Spectrum - 20V, Positive | splash10-014i-3900000000-b2afc99d27edd18d7f29 | 2017-09-01 | View Spectrum | | Predicted MS/MS | Predicted LC-MS/MS Spectrum - 40V, Positive | splash10-0lk9-9500000000-5c3747866d41ab8df618 | 2017-09-01 | View Spectrum | | Predicted MS/MS | Predicted LC-MS/MS Spectrum - 10V, Negative | splash10-014i-0900000000-5cf478650fcc3a71b790 | 2017-09-01 | View Spectrum | | Predicted MS/MS | Predicted LC-MS/MS Spectrum - 20V, Negative | splash10-014i-0900000000-86832153dde465116f78 | 2017-09-01 | View Spectrum | | Predicted MS/MS | Predicted LC-MS/MS Spectrum - 40V, Negative | splash10-066r-9600000000-2e90f09f2c87dfc45640 | 2017-09-01 | View Spectrum |
|
|---|
| NMR | | Type | Description | | View |
|---|
| 1D NMR | 1H NMR Spectrum (1D, 500 MHz, H2O, experimental) | | Spectrum | | 1D NMR | 13C NMR Spectrum (1D, 400 MHz, H2O, experimental) | | Spectrum | | 1D NMR | 13C NMR Spectrum (1D, 100 MHz, H2O, predicted) | | Spectrum | | 1D NMR | 1H NMR Spectrum (1D, 100 MHz, H2O, predicted) | | Spectrum | | 1D NMR | 13C NMR Spectrum (1D, 1000 MHz, H2O, predicted) | | Spectrum | | 1D NMR | 1H NMR Spectrum (1D, 1000 MHz, H2O, predicted) | | Spectrum | | 1D NMR | 13C NMR Spectrum (1D, 200 MHz, H2O, predicted) | | Spectrum | | 1D NMR | 1H NMR Spectrum (1D, 200 MHz, H2O, predicted) | | Spectrum | | 1D NMR | 13C NMR Spectrum (1D, 300 MHz, H2O, predicted) | | Spectrum | | 1D NMR | 1H NMR Spectrum (1D, 300 MHz, H2O, predicted) | | Spectrum | | 1D NMR | 13C NMR Spectrum (1D, 400 MHz, H2O, predicted) | | Spectrum | | 1D NMR | 1H NMR Spectrum (1D, 400 MHz, H2O, predicted) | | Spectrum | | 1D NMR | 13C NMR Spectrum (1D, 500 MHz, H2O, predicted) | | Spectrum | | 1D NMR | 1H NMR Spectrum (1D, 500 MHz, H2O, predicted) | | Spectrum | | 1D NMR | 13C NMR Spectrum (1D, 600 MHz, H2O, predicted) | | Spectrum | | 1D NMR | 1H NMR Spectrum (1D, 600 MHz, H2O, predicted) | | Spectrum | | 1D NMR | 13C NMR Spectrum (1D, 700 MHz, H2O, predicted) | | Spectrum | | 1D NMR | 1H NMR Spectrum (1D, 700 MHz, H2O, predicted) | | Spectrum | | 1D NMR | 13C NMR Spectrum (1D, 800 MHz, H2O, predicted) | | Spectrum | | 1D NMR | 1H NMR Spectrum (1D, 800 MHz, H2O, predicted) | | Spectrum | | 1D NMR | 13C NMR Spectrum (1D, 900 MHz, H2O, predicted) | | Spectrum | | 1D NMR | 1H NMR Spectrum (1D, 900 MHz, H2O, predicted) | | Spectrum | | 2D NMR | [1H, 1H]-TOCSY 2D NMR Spectrum (experimental) | | Spectrum | | 2D NMR | [1H, 13C]-HSQC NMR Spectrum (2D, 600 MHz, H2O, experimental) | | Spectrum |
|
|---|
| Pathways |
|---|
| Pathways | | Name | SMPDB/Pathwhiz | KEGG | | Glycine and Serine Metabolism | 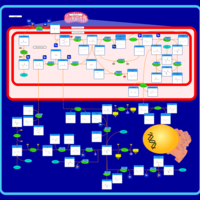 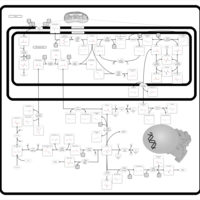 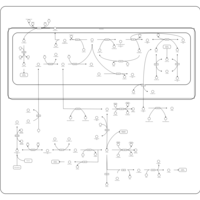 |  | | Betaine Metabolism |  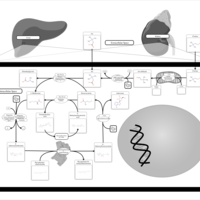  |  | | Methionine Metabolism | 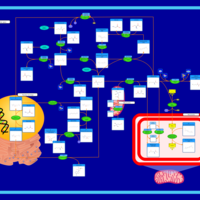 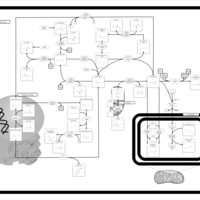 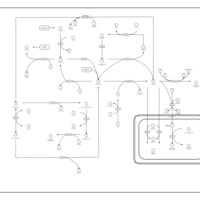 |  | | Cystathionine Beta-Synthase Deficiency | 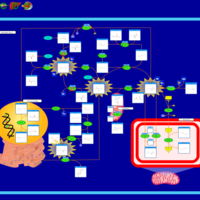 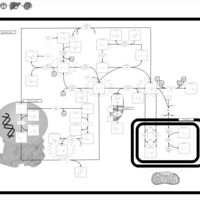 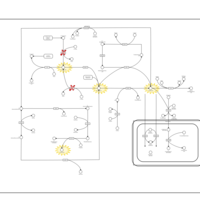 | Not Available | | Hypermethioninemia |  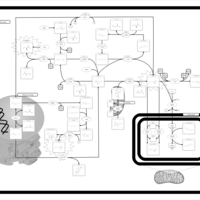  | Not Available |
|
|---|
| Protein Targets |
|---|
| Enzymes | |
| Choline dehydrogenase, mitochondrial | CHDH | 3p21.1 | Q8NE62 | details | | Alpha-aminoadipic semialdehyde dehydrogenase | ALDH7A1 | 5q31 | P49419 | details | | Betaine--homocysteine S-methyltransferase 1 | BHMT | 5q14.1 | Q93088 | details | | S-methylmethionine--homocysteine S-methyltransferase BHMT2 | BHMT2 | 5q13 | Q9H2M3 | details |
|
|---|
| Transporters | |
| Sodium- and chloride-dependent betaine transporter | SLC6A12 | 12p13 | P48065 | details |
|
|---|
| Metal Bindings | |
| Betaine--homocysteine S-methyltransferase 1 | BHMT | 5q14.1 | Q93088 | details | | S-methylmethionine--homocysteine S-methyltransferase BHMT2 | BHMT2 | 5q13 | Q9H2M3 | details |
|
|---|
| Receptors | Not Available |
|---|
| Transcriptional Factors | Not Available |
|---|
| Concentrations Data |
|---|
| |
| Alien Dawg | Detected and Quantified | 0.0168 mg/g dry wt | | details | | Gabriola | Detected and Quantified | 0.00745 mg/g dry wt | | details | | Island Honey | Detected and Quantified | 0.0165 mg/g dry wt | | details | | Quadra | Detected and Quantified | 0.00903 mg/g dry wt | | details | | Sensi Star | Detected and Quantified | 0.00950 mg/g dry wt | | details | | Tangerine Dream | Detected and Quantified | 0.0104 mg/g dry wt | | details |
|
|---|
| External Links |
|---|
| HMDB ID | HMDB0000043 |
|---|
| DrugBank ID | DB06756 |
|---|
| Phenol Explorer Compound ID | Not Available |
|---|
| FoodDB ID | FDB009020 |
|---|
| KNApSAcK ID | C00007291 |
|---|
| Chemspider ID | 242 |
|---|
| KEGG Compound ID | C00719 |
|---|
| BioCyc ID | BETAINE |
|---|
| BiGG ID | 35786 |
|---|
| Wikipedia Link | Trimethylglycine |
|---|
| METLIN ID | 287 |
|---|
| PubChem Compound | 247 |
|---|
| PDB ID | Not Available |
|---|
| ChEBI ID | 17750 |
|---|
| References |
|---|
| General References | - Amiraslani B, Sabouni F, Abbasi S, Nazem H, Sabet M: Recognition of betaine as an inhibitor of lipopolysaccharide-induced nitric oxide production in activated microglial cells. Iran Biomed J. 2012;16(2):84-9. doi: 10.6091/ibj.1012.2012. [PubMed:22801281 ]
- Lever M, Slow S: The clinical significance of betaine, an osmolyte with a key role in methyl group metabolism. Clin Biochem. 2010 Jun;43(9):732-44. doi: 10.1016/j.clinbiochem.2010.03.009. Epub 2010 Mar 25. [PubMed:20346934 ]
|
|---|

