| Record Information |
|---|
| Version | 1.0 |
|---|
| Created at | 2020-04-17 19:11:37 UTC |
|---|
| Updated at | 2020-12-07 19:11:38 UTC |
|---|
| CannabisDB ID | CDB005123 |
|---|
| Secondary Accession Numbers | Not Available |
|---|
| Cannabis Compound Identification |
|---|
| Common Name | 3-Methyl-2-oxovaleric acid |
|---|
| Description | 3-Methyl-2-oxovaleric acid, also known as 3-methyl-2-oxopentanoate or α-keto-β-methyl-N-valeric acid, belongs to the class of organic compounds known as short-chain keto acids and derivatives. These are keto acids with an alkyl chain the contains less than 6 carbon atoms. 3-Methyl-2-oxovaleric acid is an extremely weak basic (essentially neutral) compound (based on its pKa). 3-Methyl-2-oxovaleric acid exists in all living organisms, ranging from bacteria to humans. Outside of the human body, 3-Methyl-2-oxovaleric acid has been detected, but not quantified in, several different foods, such as alcoholic beverages, asparagus, cocoa and cocoa products, green vegetables, and milk and milk products. This could make 3-methyl-2-oxovaleric acid a potential biomarker for the consumption of these foods. An alpha-keto acid analogue and metabolite of isoleucine in man, animals and bacteria. 3-Methyl-2-oxovaleric acid is a potentially toxic compound. A 2-oxo monocarboxylic acid that is valeric acid carrying oxo- and methyl substituents at C-2 and C-3, respectively. 3-Methyl-2-oxovaleric acid, with regard to humans, has been found to be associated with several diseases such as colorectal cancer, prosthesis/missing teeth, attachment loss, and missing teeth; 3-methyl-2-oxovaleric acid has also been linked to the inborn metabolic disorder maple syrup urine disease. Used as a clinical marker for maple syrup urine disease (MSUD). 3-Methyl-2-oxovaleric acid is expected to be in Cannabis as all living plants are known to produce and metabolize it. |
|---|
| Structure | |
|---|
| Synonyms | | Value | Source |
|---|
| 2-oxo-3-Methyl-N-valeric acid | ChEBI | | 2-oxo-3-Methylpentanoic acid | ChEBI | | 2-oxo-3-Methylvalerate | ChEBI | | 2-oxo-3-Methylvaleric acid | ChEBI | | 2-Oxoisoleucine | ChEBI | | 2-Oxokolavenic acid | ChEBI | | 3-Ethyl-3-methylpyruvic acid | ChEBI | | 3-Methyl-2-oxopentanoic acid | ChEBI | | alpha-Keto-beta-methyl-N-valeric acid | ChEBI | | alpha-Keto-beta-methylvaleric acid | ChEBI | | alpha-oxo-beta-Methyl-N-valeric acid | ChEBI | | alpha-oxo-beta-Methylvaleric acid | ChEBI | | Ketoisoleucine | ChEBI | | 2-oxo-3-Methyl-N-valerate | Generator | | 2-oxo-3-Methylpentanoate | Generator | | 2-Oxokolavenate | Generator | | 3-Ethyl-3-methylpyruvate | Generator | | 3-Methyl-2-oxopentanoate | Generator | | a-Keto-b-methyl-N-valerate | Generator | | a-Keto-b-methyl-N-valeric acid | Generator | | alpha-Keto-beta-methyl-N-valerate | Generator | | Α-keto-β-methyl-N-valerate | Generator | | Α-keto-β-methyl-N-valeric acid | Generator | | a-Keto-b-methylvalerate | Generator | | a-Keto-b-methylvaleric acid | Generator | | alpha-Keto-beta-methylvalerate | Generator | | Α-keto-β-methylvalerate | Generator | | Α-keto-β-methylvaleric acid | Generator | | a-oxo-b-Methyl-N-valerate | Generator | | a-oxo-b-Methyl-N-valeric acid | Generator | | alpha-oxo-beta-Methyl-N-valerate | Generator | | Α-oxo-β-methyl-N-valerate | Generator | | Α-oxo-β-methyl-N-valeric acid | Generator | | a-oxo-b-Methylvalerate | Generator | | a-oxo-b-Methylvaleric acid | Generator | | alpha-oxo-beta-Methylvalerate | Generator | | Α-oxo-β-methylvalerate | Generator | | Α-oxo-β-methylvaleric acid | Generator | | 3-Methyl-2-oxovalerate | Generator | | (3R)-3-Methyl-2-oxopentanoate | HMDB | | (3R)-3-Methyl-2-oxopentanoic acid | HMDB | | (R)-3-Methyl-2-oxopentanoate | HMDB | | (R)-3-Methyl-2-oxopentanoic acid | HMDB | | (S)-3-Methyl-2-oxopentanoate | HMDB | | (S)-3-Methyl-2-oxopentanoic acid | HMDB | | 2-Keto-3-methylvalerate | HMDB | | 2-Keto-3-methylvaleric acid | HMDB | | 3-Methyl-2-oxo-pentanoate | HMDB | | 3-Methyl-2-oxo-pentanoic acid | HMDB | | 3-Methyl-2-oxo-valerate | HMDB | | 3-Methyl-2-oxo-valeric acid | HMDB | | 3-Methyl-2-oxovaleric | HMDB | | 2-Keto methylvalerate | HMDB | | KMVA | HMDB | | alpha-Keto-beta-methylvaleric acid, calcium salt, (S)-isomer | HMDB | | alpha-Keto-beta-methylvaleric acid, monosodium salt, (S)-isomer | HMDB | | alpha-Keto-beta-methylvaleric acid, (S)-isomer | HMDB | | alpha-Keto-beta-methylvaleric acid, calcium salt | HMDB | | alpha-Keto-beta-methylvaleric acid, sodium salt, (+-)-isomer | HMDB | | alpha-Keto-beta-methylvaleric acid, (+-)-isomer | HMDB | | alpha-Keto-beta-methylvaleric acid, sodium salt | HMDB |
|
|---|
| Chemical Formula | C6H10O3 |
|---|
| Average Molecular Weight | 130.14 |
|---|
| Monoisotopic Molecular Weight | 130.063 |
|---|
| IUPAC Name | 3-methyl-2-oxopentanoic acid |
|---|
| Traditional Name | 3-methyl-2-oxopentanoic acid |
|---|
| CAS Registry Number | 1460-34-0 |
|---|
| SMILES | CCC(C)C(=O)C(O)=O |
|---|
| InChI Identifier | InChI=1S/C6H10O3/c1-3-4(2)5(7)6(8)9/h4H,3H2,1-2H3,(H,8,9) |
|---|
| InChI Key | JVQYSWDUAOAHFM-UHFFFAOYSA-N |
|---|
| Chemical Taxonomy |
|---|
| Description | Belongs to the class of organic compounds known as short-chain keto acids and derivatives. These are keto acids with an alkyl chain the contains less than 6 carbon atoms. |
|---|
| Kingdom | Organic compounds |
|---|
| Super Class | Organic acids and derivatives |
|---|
| Class | Keto acids and derivatives |
|---|
| Sub Class | Short-chain keto acids and derivatives |
|---|
| Direct Parent | Short-chain keto acids and derivatives |
|---|
| Alternative Parents | |
|---|
| Substituents | - Branched fatty acid
- Methyl-branched fatty acid
- Short-chain keto acid
- Alpha-keto acid
- Fatty acyl
- Alpha-hydroxy ketone
- Ketone
- Carboxylic acid derivative
- Carboxylic acid
- Monocarboxylic acid or derivatives
- Carbonyl group
- Hydrocarbon derivative
- Organic oxygen compound
- Organooxygen compound
- Organic oxide
- Aliphatic acyclic compound
|
|---|
| Molecular Framework | Aliphatic acyclic compounds |
|---|
| External Descriptors | |
|---|
| Ontology |
|---|
|
| Physiological effect | Health effect: |
|---|
| Disposition | Route of exposure: Source: Biological location: |
|---|
| Role | Indirect biological role: Industrial application: Biological role: |
|---|
| Physical Properties |
|---|
| State | Solid |
|---|
| Experimental Properties | | Property | Value | Reference |
|---|
| Melting Point | Not Available | Not Available | | Boiling Point | Not Available | Not Available | | Water Solubility | 915.5 mg/mL | Not Available | | logP | Not Available | Not Available |
|
|---|
| Predicted Properties | [] |
|---|
| Spectra |
|---|
| EI-MS/GC-MS | | Type | Description | Splash Key | View |
|---|
| GC-MS | 3-Methyl-2-oxovaleric acid, 3 TMS, GC-MS Spectrum | splash10-000i-9610000000-d2ffd4c2796688c555ee | Spectrum | | GC-MS | 3-Methyl-2-oxovaleric acid, 3 TMS, GC-MS Spectrum | splash10-000i-9720000000-9cbc934e100bedd87f17 | Spectrum | | GC-MS | 3-Methyl-2-oxovaleric acid, non-derivatized, GC-MS Spectrum | splash10-000i-9720000000-5acbdc73aee24c023763 | Spectrum | | GC-MS | 3-Methyl-2-oxovaleric acid, non-derivatized, GC-MS Spectrum | splash10-000i-6930000000-8c2cf1d8df538010611b | Spectrum | | GC-MS | 3-Methyl-2-oxovaleric acid, non-derivatized, GC-MS Spectrum | splash10-000i-6930000000-7bef6751c4c607393e5e | Spectrum | | GC-MS | 3-Methyl-2-oxovaleric acid, non-derivatized, GC-MS Spectrum | splash10-0hg9-4950000000-3e1c700a6f553bf63afb | Spectrum | | GC-MS | 3-Methyl-2-oxovaleric acid, non-derivatized, GC-MS Spectrum | splash10-000i-9610000000-d2ffd4c2796688c555ee | Spectrum | | GC-MS | 3-Methyl-2-oxovaleric acid, non-derivatized, GC-MS Spectrum | splash10-000i-9720000000-9cbc934e100bedd87f17 | Spectrum | | GC-MS | 3-Methyl-2-oxovaleric acid, non-derivatized, GC-MS Spectrum | splash10-000i-9720000000-5acbdc73aee24c023763 | Spectrum | | GC-MS | 3-Methyl-2-oxovaleric acid, non-derivatized, GC-MS Spectrum | splash10-000i-6930000000-8c2cf1d8df538010611b | Spectrum | | Predicted GC-MS | 3-Methyl-2-oxovaleric acid, non-derivatized, Predicted GC-MS Spectrum - 70eV, Positive | splash10-056r-9000000000-d08073312b337cdae9e6 | Spectrum | | Predicted GC-MS | 3-Methyl-2-oxovaleric acid, 1 TMS, Predicted GC-MS Spectrum - 70eV, Positive | splash10-05bo-9400000000-9937bff65302162dc3b9 | Spectrum |
|
|---|
| MS/MS | | Type | Description | Splash Key | View |
|---|
| MS/MS | LC-MS/MS Spectrum - Quattro_QQQ 10V, Positive (Annotated) | splash10-000w-9300000000-0a6ff60455403c38be39 | 2012-07-24 | View Spectrum | | MS/MS | LC-MS/MS Spectrum - Quattro_QQQ 25V, Positive (Annotated) | splash10-0002-9000000000-7ee6d2fb63c6ea4cccfa | 2012-07-24 | View Spectrum | | MS/MS | LC-MS/MS Spectrum - Quattro_QQQ 40V, Positive (Annotated) | splash10-002b-9000000000-47d18dff23b741029581 | 2012-07-24 | View Spectrum | | MS/MS | LC-MS/MS Spectrum - LC-ESI-ITFT , negative | splash10-004i-0900000000-ae9da2a9acfa56fc6091 | 2017-09-14 | View Spectrum | | MS/MS | LC-MS/MS Spectrum - LC-ESI-IT , negative | splash10-004r-5900000000-a3f740a5c859439255be | 2017-09-14 | View Spectrum | | Predicted MS/MS | Predicted LC-MS/MS Spectrum - 10V, Positive | splash10-06sr-7900000000-4920c1aefcca814ec1f6 | 2017-09-01 | View Spectrum | | Predicted MS/MS | Predicted LC-MS/MS Spectrum - 20V, Positive | splash10-0a4r-9100000000-16995fa8eb603d828a2f | 2017-09-01 | View Spectrum | | Predicted MS/MS | Predicted LC-MS/MS Spectrum - 40V, Positive | splash10-0a4i-9000000000-a6ccc899039a438533ae | 2017-09-01 | View Spectrum | | Predicted MS/MS | Predicted LC-MS/MS Spectrum - 10V, Negative | splash10-004r-5900000000-50107933abbe9c175553 | 2017-09-01 | View Spectrum | | Predicted MS/MS | Predicted LC-MS/MS Spectrum - 20V, Negative | splash10-01ri-9700000000-23998218af9ee2898594 | 2017-09-01 | View Spectrum | | Predicted MS/MS | Predicted LC-MS/MS Spectrum - 40V, Negative | splash10-0a4i-9000000000-a30461e2df45eeb8f795 | 2017-09-01 | View Spectrum |
|
|---|
| NMR | | Type | Description | | View |
|---|
| 1D NMR | 1H NMR Spectrum (1D, 500 MHz, H2O, experimental) | | Spectrum | | 1D NMR | 13C NMR Spectrum (1D, 100 MHz, H2O, predicted) | | Spectrum | | 1D NMR | 1H NMR Spectrum (1D, 100 MHz, H2O, predicted) | | Spectrum | | 1D NMR | 13C NMR Spectrum (1D, 1000 MHz, H2O, predicted) | | Spectrum | | 1D NMR | 1H NMR Spectrum (1D, 1000 MHz, H2O, predicted) | | Spectrum | | 1D NMR | 13C NMR Spectrum (1D, 200 MHz, H2O, predicted) | | Spectrum | | 1D NMR | 1H NMR Spectrum (1D, 200 MHz, H2O, predicted) | | Spectrum | | 1D NMR | 13C NMR Spectrum (1D, 300 MHz, H2O, predicted) | | Spectrum | | 1D NMR | 1H NMR Spectrum (1D, 300 MHz, H2O, predicted) | | Spectrum | | 1D NMR | 13C NMR Spectrum (1D, 400 MHz, H2O, predicted) | | Spectrum | | 1D NMR | 1H NMR Spectrum (1D, 400 MHz, H2O, predicted) | | Spectrum | | 1D NMR | 13C NMR Spectrum (1D, 500 MHz, H2O, predicted) | | Spectrum | | 1D NMR | 1H NMR Spectrum (1D, 500 MHz, H2O, predicted) | | Spectrum | | 1D NMR | 13C NMR Spectrum (1D, 600 MHz, H2O, predicted) | | Spectrum | | 1D NMR | 1H NMR Spectrum (1D, 600 MHz, H2O, predicted) | | Spectrum | | 1D NMR | 13C NMR Spectrum (1D, 700 MHz, H2O, predicted) | | Spectrum | | 1D NMR | 1H NMR Spectrum (1D, 700 MHz, H2O, predicted) | | Spectrum | | 1D NMR | 13C NMR Spectrum (1D, 800 MHz, H2O, predicted) | | Spectrum | | 1D NMR | 1H NMR Spectrum (1D, 800 MHz, H2O, predicted) | | Spectrum | | 1D NMR | 13C NMR Spectrum (1D, 900 MHz, H2O, predicted) | | Spectrum | | 1D NMR | 1H NMR Spectrum (1D, 900 MHz, H2O, predicted) | | Spectrum | | 2D NMR | [1H, 13C]-HSQC NMR Spectrum (2D, 600 MHz, H2O, experimental) | | Spectrum |
|
|---|
| Pathways |
|---|
| Pathways | | Name | SMPDB/Pathwhiz | KEGG | | Valine, Leucine and Isoleucine Degradation |  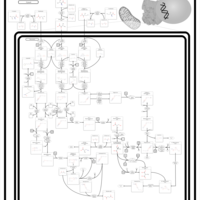 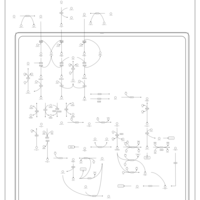 |  | | Beta-Ketothiolase Deficiency | 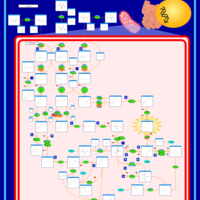 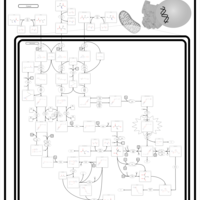 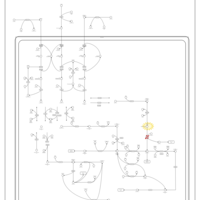 | Not Available | | 2-Methyl-3-Hydroxybutryl CoA Dehydrogenase Deficiency |  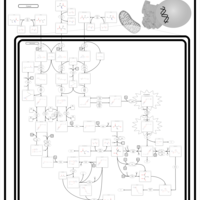 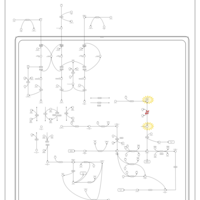 | Not Available | | Propionic Acidemia | 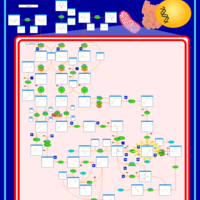 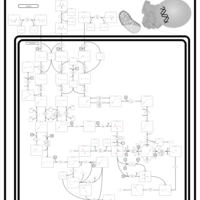 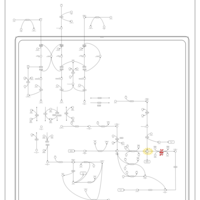 | Not Available | | 3-Hydroxy-3-Methylglutaryl-CoA Lyase Deficiency | 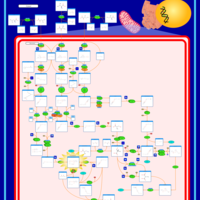 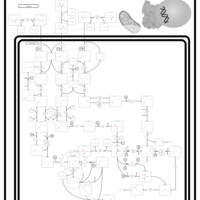 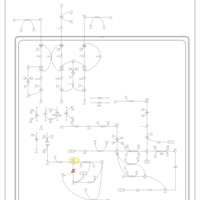 | Not Available |
|
|---|
| Protein Targets |
|---|
| Enzymes | |
| 2-oxoisovalerate dehydrogenase subunit beta, mitochondrial | BCKDHB | 6q14.1 | P21953 | details | | 2-oxoisovalerate dehydrogenase subunit alpha, mitochondrial | BCKDHA | 19q13.1-q13.2 | P12694 | details | | Branched-chain-amino-acid aminotransferase, cytosolic | BCAT1 | 12p12.1 | P54687 | details | | Branched-chain-amino-acid aminotransferase, mitochondrial | BCAT2 | 19q13 | O15382 | details | | L-amino-acid oxidase | IL4I1 | 19q13.3-q13.4 | Q96RQ9 | details |
|
|---|
| Transporters | Not Available |
|---|
| Metal Bindings | |
| 2-oxoisovalerate dehydrogenase subunit alpha, mitochondrial | BCKDHA | 19q13.1-q13.2 | P12694 | details |
|
|---|
| Receptors | Not Available |
|---|
| Transcriptional Factors | Not Available |
|---|
| Concentrations Data |
|---|
| Not Available |
|---|
| External Links |
|---|
| HMDB ID | HMDB0000491 |
|---|
| DrugBank ID | Not Available |
|---|
| Phenol Explorer Compound ID | Not Available |
|---|
| FoodDB ID | FDB021447 |
|---|
| KNApSAcK ID | Not Available |
|---|
| Chemspider ID | 46 |
|---|
| KEGG Compound ID | C03465 |
|---|
| BioCyc ID | Not Available |
|---|
| BiGG ID | 35662 |
|---|
| Wikipedia Link | Not Available |
|---|
| METLIN ID | 5478 |
|---|
| PubChem Compound | 47 |
|---|
| PDB ID | Not Available |
|---|
| ChEBI ID | 35932 |
|---|
| References |
|---|
| General References | Not Available |
|---|
