| Record Information |
|---|
| Version | 1.0 |
|---|
| Created at | 2020-04-17 19:10:24 UTC |
|---|
| Updated at | 2020-12-07 19:11:36 UTC |
|---|
| CannabisDB ID | CDB005111 |
|---|
| Secondary Accession Numbers | Not Available |
|---|
| Cannabis Compound Identification |
|---|
| Common Name | Succinic acid semialdehyde |
|---|
| Description | Succinic acid semialdehyde, also known as 3-formylpropionic acid or 4-oxobutanoate, belongs to the class of organic compounds known as straight chain fatty acids. These are fatty acids with a straight aliphatic chain. Succinic acid semialdehyde is a very hydrophobic molecule, practically insoluble (in water), and relatively neutral. Succinic acid semialdehyde is a potentially toxic compound. Succinic acid semialdehyde is expected to be in Cannabis as all living plants are known to produce and metabolize it. |
|---|
| Structure | |
|---|
| Synonyms | | Value | Source |
|---|
| 3-Formylpropanoic acid | ChEBI | | 3-Formylpropionic acid | ChEBI | | 4-Oxobutanoate | ChEBI | | beta-Formylpropionic acid | ChEBI | | Semialdehyde succinique | ChEBI | | Succinaldehydic acid | ChEBI | | Succinate semialdehyde | ChEBI | | Succinic semialdehyde | Kegg | | 3-Formylpropanoate | Generator | | 3-Formylpropionate | Generator | | 4-Oxobutanoic acid | Generator | | b-Formylpropionate | Generator | | b-Formylpropionic acid | Generator | | beta-Formylpropionate | Generator | | Β-formylpropionate | Generator | | Β-formylpropionic acid | Generator | | Succinaldehydate | Generator | | 2-Formylpropionic acid ethyl ester | HMDB | | Butryaldehydate | HMDB | | Butryaldehydic acid | HMDB | | gamma-Oxybutyrate | HMDB | | gamma-Oxybutyric acid | HMDB | | Succinic semialdehyde, calcium salt | HMDB | | Succinic acid semialdehyde | ChEBI |
|
|---|
| Chemical Formula | C4H6O3 |
|---|
| Average Molecular Weight | 102.09 |
|---|
| Monoisotopic Molecular Weight | 102.0317 |
|---|
| IUPAC Name | 4-oxobutanoic acid |
|---|
| Traditional Name | succinic semialdehyde |
|---|
| CAS Registry Number | 692-29-5 |
|---|
| SMILES | OC(=O)CCC=O |
|---|
| InChI Identifier | InChI=1S/C4H6O3/c5-3-1-2-4(6)7/h3H,1-2H2,(H,6,7) |
|---|
| InChI Key | UIUJIQZEACWQSV-UHFFFAOYSA-N |
|---|
| Chemical Taxonomy |
|---|
| Description | Belongs to the class of organic compounds known as straight chain fatty acids. These are fatty acids with a straight aliphatic chain. |
|---|
| Kingdom | Organic compounds |
|---|
| Super Class | Lipids and lipid-like molecules |
|---|
| Class | Fatty Acyls |
|---|
| Sub Class | Fatty acids and conjugates |
|---|
| Direct Parent | Straight chain fatty acids |
|---|
| Alternative Parents | |
|---|
| Substituents | - Straight chain fatty acid
- Alpha-hydrogen aldehyde
- Monocarboxylic acid or derivatives
- Carboxylic acid
- Carboxylic acid derivative
- Organic oxygen compound
- Organic oxide
- Hydrocarbon derivative
- Organooxygen compound
- Carbonyl group
- Aldehyde
- Aliphatic acyclic compound
|
|---|
| Molecular Framework | Aliphatic acyclic compounds |
|---|
| External Descriptors | |
|---|
| Ontology |
|---|
|
| Disposition | Route of exposure: Source: Biological location: |
|---|
| Role | Indirect biological role: Industrial application: Biological role: |
|---|
| Physical Properties |
|---|
| State | Solid |
|---|
| Experimental Properties | | Property | Value | Reference |
|---|
| Melting Point | Not Available | Not Available | | Boiling Point | Not Available | Not Available | | Water Solubility | Not Available | Not Available | | logP | Not Available | Not Available |
|
|---|
| Predicted Properties | [] |
|---|
| Spectra |
|---|
| EI-MS/GC-MS | | Type | Description | Splash Key | View |
|---|
| GC-MS | Succinic acid semialdehyde, 1 MEOX; 1 TMS, GC-MS Spectrum | splash10-000i-9400000000-8e1cb554add6ed6c4e35 | Spectrum | | GC-MS | Succinic acid semialdehyde, 1 MEOX; 1 TMS, GC-MS Spectrum | splash10-000i-9300000000-f5403e2e858fded273ac | Spectrum | | GC-MS | Succinic acid semialdehyde, non-derivatized, GC-MS Spectrum | splash10-000i-9400000000-8e1cb554add6ed6c4e35 | Spectrum | | GC-MS | Succinic acid semialdehyde, non-derivatized, GC-MS Spectrum | splash10-000i-9300000000-f5403e2e858fded273ac | Spectrum | | GC-MS | Succinic acid semialdehyde, non-derivatized, GC-MS Spectrum | splash10-000i-9500000000-eb52f4d003b7d5b86b3d | Spectrum | | GC-MS | Succinic acid semialdehyde, non-derivatized, GC-MS Spectrum | splash10-000i-9600000000-463f366ebd0be24a0283 | Spectrum | | Predicted GC-MS | Succinic acid semialdehyde, non-derivatized, Predicted GC-MS Spectrum - 70eV, Positive | splash10-052f-9000000000-d9618eb947af434dcf2f | Spectrum | | Predicted GC-MS | Succinic acid semialdehyde, 1 TMS, Predicted GC-MS Spectrum - 70eV, Positive | splash10-05fr-9800000000-40373963cdda851ebfea | Spectrum | | Predicted GC-MS | Succinic acid semialdehyde, non-derivatized, Predicted GC-MS Spectrum - 70eV, Positive | Not Available | Spectrum |
|
|---|
| MS/MS | | Type | Description | Splash Key | View |
|---|
| MS/MS | LC-MS/MS Spectrum - Quattro_QQQ 10V, Positive (Annotated) | splash10-000i-9000000000-bf9ebea7e4800559c111 | 2012-07-24 | View Spectrum | | MS/MS | LC-MS/MS Spectrum - Orbitrap 1V, negative | splash10-0udi-0900000000-64b3498f7cdd4597da6a | 2020-07-22 | View Spectrum | | MS/MS | LC-MS/MS Spectrum - Orbitrap 1V, negative | splash10-0udi-1900000000-9d47aab2400f1da945bc | 2020-07-22 | View Spectrum | | MS/MS | LC-MS/MS Spectrum - Orbitrap 2V, negative | splash10-0udi-1900000000-94e1ac309574efbe3903 | 2020-07-22 | View Spectrum | | MS/MS | LC-MS/MS Spectrum - Orbitrap 2V, negative | splash10-0udi-3900000000-7ec96b67f820176c39d3 | 2020-07-22 | View Spectrum | | MS/MS | LC-MS/MS Spectrum - Orbitrap 3V, negative | splash10-0udi-5900000000-7708b3a1c05b69363aa9 | 2020-07-22 | View Spectrum | | MS/MS | LC-MS/MS Spectrum - Orbitrap 3V, negative | splash10-0zfr-8900000000-12726b727f621ac971b7 | 2020-07-22 | View Spectrum | | MS/MS | LC-MS/MS Spectrum - n/a 7V, negative | splash10-0089-9000000000-9eb7dd16da8275702c75 | 2020-07-22 | View Spectrum | | MS/MS | LC-MS/MS Spectrum - 35V, Negative | splash10-0udi-1900000000-567d38fac821ab993d19 | 2021-09-20 | View Spectrum | | Predicted MS/MS | Predicted LC-MS/MS Spectrum - 10V, Positive | splash10-000i-9200000000-e31b8066f970e1c178d1 | 2015-05-27 | View Spectrum | | Predicted MS/MS | Predicted LC-MS/MS Spectrum - 20V, Positive | splash10-052u-9000000000-76526f6c67d7a67c1d07 | 2015-05-27 | View Spectrum | | Predicted MS/MS | Predicted LC-MS/MS Spectrum - 40V, Positive | splash10-0a4l-9000000000-70e85383e59b46bc429f | 2015-05-27 | View Spectrum | | Predicted MS/MS | Predicted LC-MS/MS Spectrum - 10V, Negative | splash10-0udi-5900000000-f1c2507387f92b6e1e3f | 2015-05-27 | View Spectrum | | Predicted MS/MS | Predicted LC-MS/MS Spectrum - 20V, Negative | splash10-0zgi-9400000000-ba1b880a90d3ace8beee | 2015-05-27 | View Spectrum | | Predicted MS/MS | Predicted LC-MS/MS Spectrum - 40V, Negative | splash10-052f-9000000000-971836bb1672ad33106e | 2015-05-27 | View Spectrum | | Predicted MS/MS | Predicted LC-MS/MS Spectrum - 10V, Positive | splash10-052f-9000000000-06109e02fc0baa708cdc | 2021-09-21 | View Spectrum | | Predicted MS/MS | Predicted LC-MS/MS Spectrum - 20V, Positive | splash10-052f-9000000000-ce51d68c1c700db8f966 | 2021-09-21 | View Spectrum | | Predicted MS/MS | Predicted LC-MS/MS Spectrum - 40V, Positive | splash10-0006-9000000000-d171f37acc222eeea075 | 2021-09-21 | View Spectrum | | Predicted MS/MS | Predicted LC-MS/MS Spectrum - 10V, Negative | splash10-0a4i-9100000000-b23feac6075bc2594f5e | 2021-09-25 | View Spectrum | | Predicted MS/MS | Predicted LC-MS/MS Spectrum - 20V, Negative | splash10-0a4i-9000000000-aa34fb8bd9988d794431 | 2021-09-25 | View Spectrum | | Predicted MS/MS | Predicted LC-MS/MS Spectrum - 40V, Negative | splash10-052f-9000000000-101491882eb1be877e89 | 2021-09-25 | View Spectrum |
|
|---|
| NMR | | Type | Description | | View |
|---|
| 1D NMR | 1H NMR Spectrum (1D, 600 MHz, H2O, experimental) | | Spectrum | | 1D NMR | 13C NMR Spectrum (1D, 100 MHz, D2O, predicted) | | Spectrum | | 1D NMR | 1H NMR Spectrum (1D, 100 MHz, D2O, predicted) | | Spectrum | | 1D NMR | 13C NMR Spectrum (1D, 1000 MHz, D2O, predicted) | | Spectrum | | 1D NMR | 1H NMR Spectrum (1D, 1000 MHz, D2O, predicted) | | Spectrum | | 1D NMR | 13C NMR Spectrum (1D, 200 MHz, D2O, predicted) | | Spectrum | | 1D NMR | 1H NMR Spectrum (1D, 200 MHz, D2O, predicted) | | Spectrum | | 1D NMR | 13C NMR Spectrum (1D, 300 MHz, D2O, predicted) | | Spectrum | | 1D NMR | 1H NMR Spectrum (1D, 300 MHz, D2O, predicted) | | Spectrum | | 1D NMR | 13C NMR Spectrum (1D, 400 MHz, D2O, predicted) | | Spectrum | | 1D NMR | 1H NMR Spectrum (1D, 400 MHz, D2O, predicted) | | Spectrum | | 1D NMR | 13C NMR Spectrum (1D, 500 MHz, D2O, predicted) | | Spectrum | | 1D NMR | 1H NMR Spectrum (1D, 500 MHz, D2O, predicted) | | Spectrum | | 1D NMR | 13C NMR Spectrum (1D, 600 MHz, D2O, predicted) | | Spectrum | | 1D NMR | 1H NMR Spectrum (1D, 600 MHz, D2O, predicted) | | Spectrum | | 1D NMR | 13C NMR Spectrum (1D, 700 MHz, D2O, predicted) | | Spectrum | | 1D NMR | 1H NMR Spectrum (1D, 700 MHz, D2O, predicted) | | Spectrum | | 1D NMR | 13C NMR Spectrum (1D, 800 MHz, D2O, predicted) | | Spectrum | | 1D NMR | 1H NMR Spectrum (1D, 800 MHz, D2O, predicted) | | Spectrum | | 1D NMR | 13C NMR Spectrum (1D, 900 MHz, D2O, predicted) | | Spectrum | | 1D NMR | 1H NMR Spectrum (1D, 900 MHz, D2O, predicted) | | Spectrum | | 2D NMR | [1H, 13C]-HSQC NMR Spectrum (2D, 600 MHz, H2O, experimental) | | Spectrum |
|
|---|
| Pathways |
|---|
| Pathways | | Name | SMPDB/Pathwhiz | KEGG | | Glutamate Metabolism | 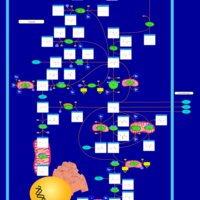 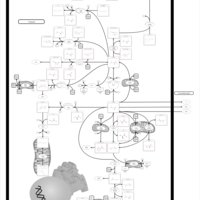 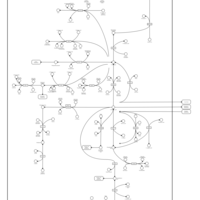 |  | | 4-Hydroxybutyric Aciduria/Succinic Semialdehyde Dehydrogenase Deficiency | 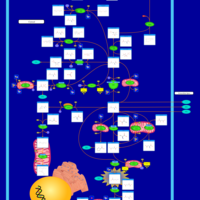 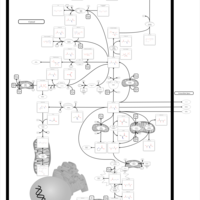 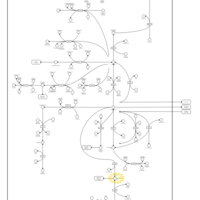 | Not Available | | Homocarnosinosis | 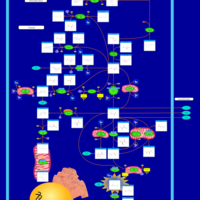 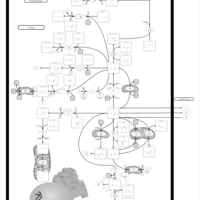 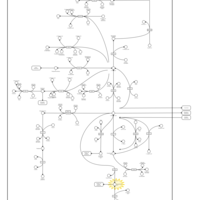 | Not Available | | Hyperinsulinism-Hyperammonemia Syndrome | 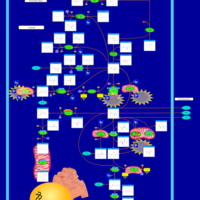 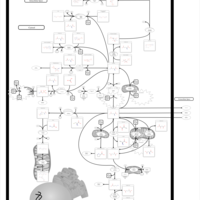 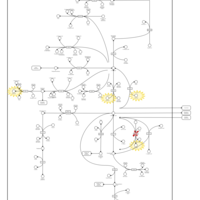 | Not Available | | 2-Hydroxyglutric Aciduria (D And L Form) | 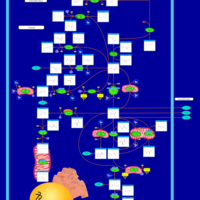 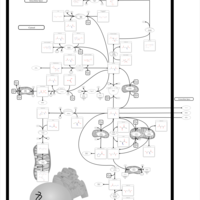 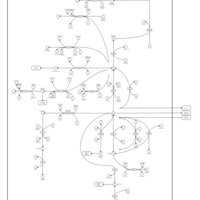 | Not Available |
|
|---|
| Protein Targets |
|---|
| Enzymes | |
| 4-aminobutyrate aminotransferase, mitochondrial | ABAT | 16p13.2 | P80404 | details | | Succinate-semialdehyde dehydrogenase, mitochondrial | ALDH5A1 | 6p22 | P51649 | details | | Aflatoxin B1 aldehyde reductase member 2 | AKR7A2 | 1p36.13 | O43488 | details | | Hydroxyacid-oxoacid transhydrogenase, mitochondrial | ADHFE1 | 8q13.1 | Q8IWW8 | details |
|
|---|
| Transporters | Not Available |
|---|
| Metal Bindings | |
| Hydroxyacid-oxoacid transhydrogenase, mitochondrial | ADHFE1 | 8q13.1 | Q8IWW8 | details |
|
|---|
| Receptors | Not Available |
|---|
| Transcriptional Factors | Not Available |
|---|
| Concentrations Data |
|---|
| Not Available |
|---|
| External Links |
|---|
| HMDB ID | HMDB0001259 |
|---|
| DrugBank ID | DBMET01476 |
|---|
| Phenol Explorer Compound ID | Not Available |
|---|
| FoodDB ID | FDB022516 |
|---|
| KNApSAcK ID | C00019682 |
|---|
| Chemspider ID | 1080 |
|---|
| KEGG Compound ID | C00232 |
|---|
| BioCyc ID | SUCC-S-ALD |
|---|
| BiGG ID | 34331 |
|---|
| Wikipedia Link | Succinic semialdehyde |
|---|
| METLIN ID | 6114 |
|---|
| PubChem Compound | 1112 |
|---|
| PDB ID | Not Available |
|---|
| ChEBI ID | 16265 |
|---|
| References |
|---|
| General References | Not Available |
|---|
