| Record Information |
|---|
| Version | 1.0 |
|---|
| Created at | 2020-04-17 19:09:55 UTC |
|---|
| Updated at | 2020-11-18 16:39:22 UTC |
|---|
| CannabisDB ID | CDB005106 |
|---|
| Secondary Accession Numbers | Not Available |
|---|
| Cannabis Compound Identification |
|---|
| Common Name | Carbamoyl phosphate |
|---|
| Description | Carbamoyl phosphate, also known as carbamoyl-p or phosphate, carbamyl, belongs to the class of organic compounds known as organic phosphoric acids and derivatives. These are organic compounds containing phosphoric acid or a derivative thereof. Carbamoyl phosphate is a moderately acidic compound (based on its pKa). Carbamoyl phosphate exists in all living species, ranging from bacteria to humans. Within humans, carbamoyl phosphate participates in a number of enzymatic reactions. In particular, carbamoyl phosphate can be biosynthesized from L-glutamine and hydrogen carbonate through the action of the enzyme cad protein. In addition, carbamoyl phosphate can be converted into ureidosuccinic acid through the action of the enzyme cad protein. Its enzymatic counterpart, carbamoyl phosphate synthetase I (CPS I), interacts with a class of molecules called sirtuins, NAD dependent protein deacetylases, and ATP to form carbamoyl phosphate. It is produced from bicarbonate, ammonia (derived from amino acids), and phosphate (from ATP). In humans, carbamoyl phosphate is involved in the metabolic disorder called the beta-ureidopropionase deficiency pathway. Outside of the human body, Carbamoyl phosphate has been detected, but not quantified in, several different foods, such as cucurbita, peanuts, common oregano, figs, and chinese water chestnuts. This could make carbamoyl phosphate a potential biomarker for the consumption of these foods. Carbamoyl phosphate is a potentially toxic compound. CP then enters the urea cycle in which it reacts with ornithine (a process catalyzed by the enzyme ornithine transcarbamylase) to form citrulline. A defect in the CPS I enzyme, and a subsequent deficiency in the production of carbamoyl phosphate has been linked to hyper-ammonemia in humans. In land-dwelling animals, it is an intermediary metabolite in nitrogen disposal through the urea cycle and the synthesis of pyrimidines. Carbamoyl phosphate is an anion of biochemical significance. Carbamoyl phosphate is expected to be in Cannabis as all living plants are known to produce and metabolize it. |
|---|
| Structure | |
|---|
| Synonyms | | Value | Source |
|---|
| Aminocarbonyl dihydrogen phosphate | ChEBI | | Carbamic phosphoric monoanhydride | ChEBI | | Carbamyl phosphate | ChEBI | | Monocarbamoyl phosphate | ChEBI | | Phosphoric acid mono(formamide)ester | ChEBI | | Aminocarbonyl dihydrogen phosphoric acid | Generator | | Carbamoyl phosphoric acid | Generator | | Carbamyl phosphoric acid | Generator | | Monocarbamoyl phosphoric acid | Generator | | Phosphate mono(formamide)ester | Generator | | Carbamic acid monoanhydride with phosphorate | HMDB | | Carbamic acid monoanhydride with phosphoric acid | HMDB | | Carbamoyl-P | HMDB | | Carbamoyl-phosphate | HMDB | | Carbamoylphosphate | HMDB | | Carbamyl-phosphate | HMDB | | Phosphate, carbamyl | MeSH, HMDB | | Phosphate, carbamoyl | MeSH, HMDB | | (Carbamoyloxy)phosphonic acid | HMDB | | Carbamoyl phosphate | HMDB |
|
|---|
| Chemical Formula | CH4NO5P |
|---|
| Average Molecular Weight | 141.02 |
|---|
| Monoisotopic Molecular Weight | 140.9827 |
|---|
| IUPAC Name | (carbamoyloxy)phosphonic acid |
|---|
| Traditional Name | carbamoyl-phosphate |
|---|
| CAS Registry Number | 590-55-6 |
|---|
| SMILES | NC(=O)OP(O)(O)=O |
|---|
| InChI Identifier | InChI=1S/CH4NO5P/c2-1(3)7-8(4,5)6/h(H2,2,3)(H2,4,5,6) |
|---|
| InChI Key | FFQKYPRQEYGKAF-UHFFFAOYSA-N |
|---|
| Chemical Taxonomy |
|---|
| Description | Belongs to the class of organic compounds known as organic phosphoric acids and derivatives. These are organic compounds containing phosphoric acid or a derivative thereof. |
|---|
| Kingdom | Organic compounds |
|---|
| Super Class | Organic acids and derivatives |
|---|
| Class | Organic phosphoric acids and derivatives |
|---|
| Sub Class | Not Available |
|---|
| Direct Parent | Organic phosphoric acids and derivatives |
|---|
| Alternative Parents | |
|---|
| Substituents | - Organic phosphoric acid derivative
- Carboximidic acid derivative
- Organic nitrogen compound
- Organic oxygen compound
- Organopnictogen compound
- Organic oxide
- Hydrocarbon derivative
- Organooxygen compound
- Organonitrogen compound
- Imine
- Aliphatic acyclic compound
|
|---|
| Molecular Framework | Aliphatic acyclic compounds |
|---|
| External Descriptors | |
|---|
| Ontology |
|---|
|
| Disposition | Route of exposure: Source: Biological location: |
|---|
| Physical Properties |
|---|
| State | Solid |
|---|
| Experimental Properties | | Property | Value | Reference |
|---|
| Melting Point | Not Available | Not Available | | Boiling Point | Not Available | Not Available | | Water Solubility | Not Available | Not Available | | logP | Not Available | Not Available |
|
|---|
| Predicted Properties | [] |
|---|
| Spectra |
|---|
| EI-MS/GC-MS | | Type | Description | Splash Key | View |
|---|
| Predicted GC-MS | Carbamoyl phosphate, non-derivatized, Predicted GC-MS Spectrum - 70eV, Positive | splash10-0002-9100000000-6a951539300f7f5b64d6 | Spectrum | | Predicted GC-MS | Carbamoyl phosphate, non-derivatized, Predicted GC-MS Spectrum - 70eV, Positive | Not Available | Spectrum | | Predicted GC-MS | Carbamoyl phosphate, non-derivatized, Predicted GC-MS Spectrum - 70eV, Positive | Not Available | Spectrum |
|
|---|
| MS/MS | | Type | Description | Splash Key | View |
|---|
| Predicted MS/MS | Predicted LC-MS/MS Spectrum - 10V, Positive | splash10-0002-9000000000-1e9b8619eed467aca3cc | 2015-05-26 | View Spectrum | | Predicted MS/MS | Predicted LC-MS/MS Spectrum - 20V, Positive | splash10-0002-9000000000-49d39aaea7b7b239f046 | 2015-05-26 | View Spectrum | | Predicted MS/MS | Predicted LC-MS/MS Spectrum - 40V, Positive | splash10-0005-9000000000-ff8b83b8641d1d23539f | 2015-05-26 | View Spectrum | | Predicted MS/MS | Predicted LC-MS/MS Spectrum - 10V, Negative | splash10-002b-9100000000-9b7601209f4777504c50 | 2015-05-27 | View Spectrum | | Predicted MS/MS | Predicted LC-MS/MS Spectrum - 20V, Negative | splash10-004i-9000000000-068148ee9b7b7acd6692 | 2015-05-27 | View Spectrum | | Predicted MS/MS | Predicted LC-MS/MS Spectrum - 40V, Negative | splash10-004i-9000000000-2eef12354837d65af0c1 | 2015-05-27 | View Spectrum | | Predicted MS/MS | Predicted LC-MS/MS Spectrum - 10V, Positive | splash10-0002-9200000000-df9df6c92477a4e65654 | 2021-09-23 | View Spectrum | | Predicted MS/MS | Predicted LC-MS/MS Spectrum - 20V, Positive | splash10-0002-9000000000-82163424a165525ca974 | 2021-09-23 | View Spectrum | | Predicted MS/MS | Predicted LC-MS/MS Spectrum - 40V, Positive | splash10-0006-9000000000-959c63a896bb7c629164 | 2021-09-23 | View Spectrum | | Predicted MS/MS | Predicted LC-MS/MS Spectrum - 10V, Negative | splash10-004i-9000000000-a5e502a2627af2048a1f | 2021-09-24 | View Spectrum | | Predicted MS/MS | Predicted LC-MS/MS Spectrum - 20V, Negative | splash10-004i-9000000000-a5e502a2627af2048a1f | 2021-09-24 | View Spectrum | | Predicted MS/MS | Predicted LC-MS/MS Spectrum - 40V, Negative | splash10-004i-9000000000-a5e502a2627af2048a1f | 2021-09-24 | View Spectrum |
|
|---|
| NMR | Not Available |
|---|
| Pathways |
|---|
| Pathways | | Name | SMPDB/Pathwhiz | KEGG | | Glutamate Metabolism | 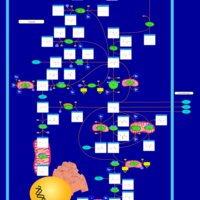 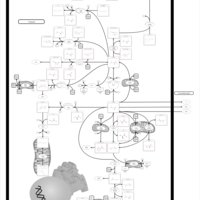 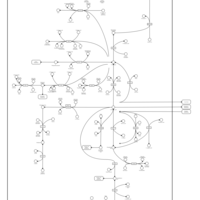 |  | | Arginine and Proline Metabolism | 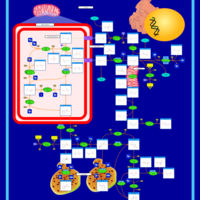 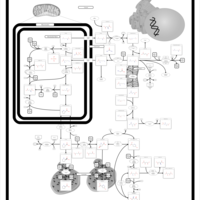 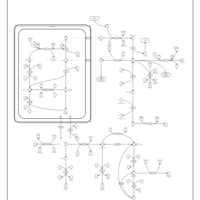 |  | | Urea Cycle | 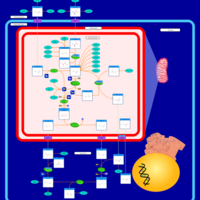 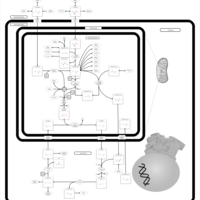 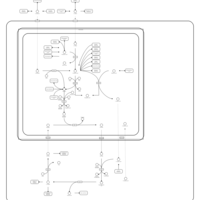 |  | | Ammonia Recycling |  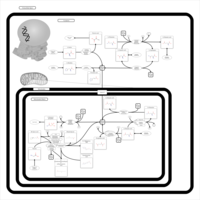 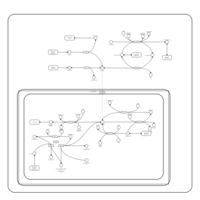 | 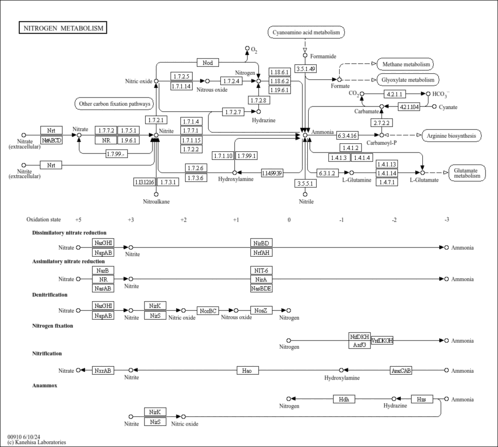 | | Pyrimidine Metabolism |  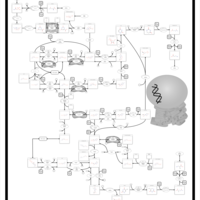  |  |
|
|---|
| Protein Targets |
|---|
| Enzymes | |
| Carbamoyl-phosphate synthase [ammonia], mitochondrial | CPS1 | 2q35 | P31327 | details | | Ornithine carbamoyltransferase, mitochondrial | OTC | Xp21.1 | P00480 | details |
|
|---|
| Transporters | Not Available |
|---|
| Metal Bindings | |
|---|
| Receptors | Not Available |
|---|
| Transcriptional Factors | Not Available |
|---|
| Concentrations Data |
|---|
| Not Available |
|---|
| External Links |
|---|
| HMDB ID | HMDB0001096 |
|---|
| DrugBank ID | Not Available |
|---|
| Phenol Explorer Compound ID | Not Available |
|---|
| FoodDB ID | FDB022424 |
|---|
| KNApSAcK ID | C00007513 |
|---|
| Chemspider ID | 272 |
|---|
| KEGG Compound ID | C00169 |
|---|
| BioCyc ID | CARBAMOYL-P |
|---|
| BiGG ID | 34125 |
|---|
| Wikipedia Link | Carbamoyl phosphate |
|---|
| METLIN ID | 6002 |
|---|
| PubChem Compound | 278 |
|---|
| PDB ID | Not Available |
|---|
| ChEBI ID | 17672 |
|---|
| References |
|---|
| General References | Not Available |
|---|



