| Record Information |
|---|
| Version | 1.0 |
|---|
| Created at | 2020-04-17 18:41:41 UTC |
|---|
| Updated at | 2020-12-07 19:11:04 UTC |
|---|
| CannabisDB ID | CDB004835 |
|---|
| Secondary Accession Numbers | Not Available |
|---|
| Cannabis Compound Identification |
|---|
| Common Name | Saccharopine |
|---|
| Description | Saccharopine belongs to the class of organic compounds known as glutamic acid and derivatives. Glutamic acid and derivatives are compounds containing glutamic acid or a derivative thereof resulting from reaction of glutamic acid at the amino group or the carboxy group, or from the replacement of any hydrogen of glycine by a heteroatom. The N(6)-(1,3-dicarboxypropan-1-yl) derivative of L-lysine. Saccharopine is a very strong basic compound (based on its pKa). Saccharopine exists in all living species, ranging from bacteria to humans. Within humans, saccharopine participates in a number of enzymatic reactions. In particular, saccharopine can be biosynthesized from L-lysine and oxoglutaric acid through the action of the enzyme Alpha-aminoadipic semialdehyde synthase, mitochondrial. In addition, saccharopine can be converted into allysine and L-glutamic acid through its interaction with the enzyme Alpha-aminoadipic semialdehyde synthase, mitochondrial. In humans, saccharopine is involved in the metabolic disorder called 2-aminoadipic 2-oxoadipic aciduria. Saccharopine is a potentially toxic compound. Saccharopine is expected to be in Cannabis as all living plants are known to produce and metabolize it. |
|---|
| Structure | |
|---|
| Synonyms | | Value | Source |
|---|
| epsilon-N-(L-Glutar-2-yl)-L-lysine | ChEBI | | N-[(S)-5-Amino-5-carboxypentyl]-L-glutamic acid | ChEBI | | N6-(L-1,3-Dicarboxypropyl)-L-lysine | ChEBI | | L-Saccharopine | Kegg | | N-[(S)-5-Amino-5-carboxypentyl]-L-glutamate | Generator | | (S)-N-(5-Amino-5-carboxypentyl)-L-glutamic acid | HMDB | | L-N-(5-Amino-5-carboxypentyl)-glutamic acid | HMDB | | L-Saccharopin | HMDB | | N(6)-(L-1,3-Dicarboxypropyl)-L-lysine | HMDB | | N-(5-Amino-5-carboxypentyl)-glutamic acid | HMDB | | N-(5-Amino-5-carboxypentyl)-L-glutamic acid | HMDB | | N-[(5S)-5-Amino-5-carboxypentyl]-L-glutamic acid | HMDB | | Saccharopin | HMDB |
|
|---|
| Chemical Formula | C11H20N2O6 |
|---|
| Average Molecular Weight | 276.29 |
|---|
| Monoisotopic Molecular Weight | 276.1321 |
|---|
| IUPAC Name | (2S)-2-{[(5S)-5-amino-5-carboxypentyl]amino}pentanedioic acid |
|---|
| Traditional Name | saccharopine |
|---|
| CAS Registry Number | 38495-82-8 |
|---|
| SMILES | N[C@@H](CCCCN[C@@H](CCC(O)=O)C(O)=O)C(O)=O |
|---|
| InChI Identifier | InChI=1S/C11H20N2O6/c12-7(10(16)17)3-1-2-6-13-8(11(18)19)4-5-9(14)15/h7-8,13H,1-6,12H2,(H,14,15)(H,16,17)(H,18,19)/t7-,8-/m0/s1 |
|---|
| InChI Key | ZDGJAHTZVHVLOT-YUMQZZPRSA-N |
|---|
| Chemical Taxonomy |
|---|
| Description | Belongs to the class of organic compounds known as glutamic acid and derivatives. Glutamic acid and derivatives are compounds containing glutamic acid or a derivative thereof resulting from reaction of glutamic acid at the amino group or the carboxy group, or from the replacement of any hydrogen of glycine by a heteroatom. |
|---|
| Kingdom | Organic compounds |
|---|
| Super Class | Organic acids and derivatives |
|---|
| Class | Carboxylic acids and derivatives |
|---|
| Sub Class | Amino acids, peptides, and analogues |
|---|
| Direct Parent | Glutamic acid and derivatives |
|---|
| Alternative Parents | |
|---|
| Substituents | - Glutamic acid or derivatives
- Alpha-amino acid
- L-alpha-amino acid
- Tricarboxylic acid or derivatives
- Amino fatty acid
- Fatty acyl
- Amino acid
- Carboxylic acid
- Secondary aliphatic amine
- Secondary amine
- Amine
- Primary amine
- Organooxygen compound
- Organonitrogen compound
- Primary aliphatic amine
- Organic nitrogen compound
- Carbonyl group
- Organic oxygen compound
- Organopnictogen compound
- Organic oxide
- Hydrocarbon derivative
- Aliphatic acyclic compound
|
|---|
| Molecular Framework | Aliphatic acyclic compounds |
|---|
| External Descriptors | |
|---|
| Ontology |
|---|
|
| Disposition | Route of exposure: Source: Biological location: |
|---|
| Role | Indirect biological role: Industrial application: |
|---|
| Physical Properties |
|---|
| State | Not Available |
|---|
| Experimental Properties | | Property | Value | Reference |
|---|
| Melting Point | 247 - 250 °C | Not Available | | Boiling Point | Not Available | Not Available | | Water Solubility | Not Available | Not Available | | logP | Not Available | Not Available |
|
|---|
| Predicted Properties | [] |
|---|
| Spectra |
|---|
| EI-MS/GC-MS | | Type | Description | Splash Key | View |
|---|
| GC-MS | Saccharopine, non-derivatized, GC-MS Spectrum | splash10-0a4i-0923000000-1964addb7be546c2a45a | Spectrum | | GC-MS | Saccharopine, non-derivatized, GC-MS Spectrum | splash10-0a4j-0913000000-49fae417f545c42253dd | Spectrum | | GC-MS | Saccharopine, non-derivatized, GC-MS Spectrum | splash10-0a4i-0923000000-1964addb7be546c2a45a | Spectrum | | GC-MS | Saccharopine, non-derivatized, GC-MS Spectrum | splash10-0a4j-0913000000-49fae417f545c42253dd | Spectrum | | Predicted GC-MS | Saccharopine, non-derivatized, Predicted GC-MS Spectrum - 70eV, Positive | splash10-001u-6950000000-fed481b3c49c1ab6ccd2 | Spectrum | | Predicted GC-MS | Saccharopine, 3 TMS, Predicted GC-MS Spectrum - 70eV, Positive | splash10-006x-9220800000-77f63e444994f915feaa | Spectrum | | Predicted GC-MS | Saccharopine, non-derivatized, Predicted GC-MS Spectrum - 70eV, Positive | Not Available | Spectrum | | Predicted GC-MS | Saccharopine, TMS_1_1, Predicted GC-MS Spectrum - 70eV, Positive | Not Available | Spectrum | | Predicted GC-MS | Saccharopine, TMS_1_2, Predicted GC-MS Spectrum - 70eV, Positive | Not Available | Spectrum | | Predicted GC-MS | Saccharopine, TMS_1_3, Predicted GC-MS Spectrum - 70eV, Positive | Not Available | Spectrum | | Predicted GC-MS | Saccharopine, TMS_1_4, Predicted GC-MS Spectrum - 70eV, Positive | Not Available | Spectrum | | Predicted GC-MS | Saccharopine, TMS_1_5, Predicted GC-MS Spectrum - 70eV, Positive | Not Available | Spectrum | | Predicted GC-MS | Saccharopine, TMS_2_1, Predicted GC-MS Spectrum - 70eV, Positive | Not Available | Spectrum | | Predicted GC-MS | Saccharopine, TMS_2_2, Predicted GC-MS Spectrum - 70eV, Positive | Not Available | Spectrum | | Predicted GC-MS | Saccharopine, TMS_2_3, Predicted GC-MS Spectrum - 70eV, Positive | Not Available | Spectrum | | Predicted GC-MS | Saccharopine, TMS_2_4, Predicted GC-MS Spectrum - 70eV, Positive | Not Available | Spectrum | | Predicted GC-MS | Saccharopine, TMS_2_5, Predicted GC-MS Spectrum - 70eV, Positive | Not Available | Spectrum | | Predicted GC-MS | Saccharopine, TMS_2_6, Predicted GC-MS Spectrum - 70eV, Positive | Not Available | Spectrum | | Predicted GC-MS | Saccharopine, TMS_2_7, Predicted GC-MS Spectrum - 70eV, Positive | Not Available | Spectrum | | Predicted GC-MS | Saccharopine, TMS_2_8, Predicted GC-MS Spectrum - 70eV, Positive | Not Available | Spectrum | | Predicted GC-MS | Saccharopine, TMS_2_9, Predicted GC-MS Spectrum - 70eV, Positive | Not Available | Spectrum | | Predicted GC-MS | Saccharopine, TMS_2_10, Predicted GC-MS Spectrum - 70eV, Positive | Not Available | Spectrum | | Predicted GC-MS | Saccharopine, TMS_2_11, Predicted GC-MS Spectrum - 70eV, Positive | Not Available | Spectrum | | Predicted GC-MS | Saccharopine, TMS_3_2, Predicted GC-MS Spectrum - 70eV, Positive | Not Available | Spectrum | | Predicted GC-MS | Saccharopine, TMS_3_3, Predicted GC-MS Spectrum - 70eV, Positive | Not Available | Spectrum |
|
|---|
| MS/MS | | Type | Description | Splash Key | View |
|---|
| MS/MS | LC-MS/MS Spectrum - LC-ESI-QTOF (UPLC Q-Tof Premier, Waters) , Positive | splash10-001i-9240000000-d841eea4ebc5da1106f7 | 2012-08-31 | View Spectrum | | MS/MS | LC-MS/MS Spectrum - LC-ESI-QTOF (UPLC Q-Tof Premier, Waters) , Negative | splash10-0a4i-0290000000-c0b375ed0505ad2eab6c | 2012-08-31 | View Spectrum | | MS/MS | LC-MS/MS Spectrum - LC-ESI-QTOF , negative | splash10-0a4i-0290000000-c0b375ed0505ad2eab6c | 2017-09-14 | View Spectrum | | MS/MS | LC-MS/MS Spectrum - LC-ESI-QTOF , positive | splash10-001i-9240000000-d841eea4ebc5da1106f7 | 2017-09-14 | View Spectrum | | Predicted MS/MS | Predicted LC-MS/MS Spectrum - 10V, Positive | splash10-06ur-0190000000-34536967c85ac70477b5 | 2016-09-12 | View Spectrum | | Predicted MS/MS | Predicted LC-MS/MS Spectrum - 20V, Positive | splash10-01qi-2790000000-ef0279e3d556894509f0 | 2016-09-12 | View Spectrum | | Predicted MS/MS | Predicted LC-MS/MS Spectrum - 40V, Positive | splash10-0019-6900000000-76748510f1b49f459caf | 2016-09-12 | View Spectrum | | Predicted MS/MS | Predicted LC-MS/MS Spectrum - 10V, Negative | splash10-004i-0190000000-417ff61b4b14d1bfe5fc | 2016-09-12 | View Spectrum | | Predicted MS/MS | Predicted LC-MS/MS Spectrum - 20V, Negative | splash10-01u9-1490000000-b4e2c5921f08ff4ee977 | 2016-09-12 | View Spectrum | | Predicted MS/MS | Predicted LC-MS/MS Spectrum - 40V, Negative | splash10-0kms-7920000000-32382165ff3ba15b14d6 | 2016-09-12 | View Spectrum | | Predicted MS/MS | Predicted LC-MS/MS Spectrum - 10V, Positive | splash10-0059-0190000000-b1accd72c8a2f16e7a71 | 2021-09-22 | View Spectrum | | Predicted MS/MS | Predicted LC-MS/MS Spectrum - 20V, Positive | splash10-001i-9310000000-56c681de611fbd62d1c7 | 2021-09-22 | View Spectrum | | Predicted MS/MS | Predicted LC-MS/MS Spectrum - 40V, Positive | splash10-001i-9000000000-f91927cbf4969536ddce | 2021-09-22 | View Spectrum | | Predicted MS/MS | Predicted LC-MS/MS Spectrum - 10V, Negative | splash10-0a4i-0090000000-dfdb3652043e0e0c5f80 | 2021-09-22 | View Spectrum | | Predicted MS/MS | Predicted LC-MS/MS Spectrum - 20V, Negative | splash10-057i-1390000000-910bea675f309038db9a | 2021-09-22 | View Spectrum | | Predicted MS/MS | Predicted LC-MS/MS Spectrum - 40V, Negative | splash10-0f95-7900000000-1df925bebb97a29a8080 | 2021-09-22 | View Spectrum |
|
|---|
| NMR | | Type | Description | | View |
|---|
| 1D NMR | 1H NMR Spectrum (1D, 500 MHz, H2O, experimental) | | Spectrum | | 1D NMR | 13C NMR Spectrum (1D, 100 MHz, D2O, predicted) | | Spectrum | | 1D NMR | 1H NMR Spectrum (1D, 100 MHz, D2O, predicted) | | Spectrum | | 1D NMR | 13C NMR Spectrum (1D, 1000 MHz, D2O, predicted) | | Spectrum | | 1D NMR | 1H NMR Spectrum (1D, 1000 MHz, D2O, predicted) | | Spectrum | | 1D NMR | 13C NMR Spectrum (1D, 200 MHz, D2O, predicted) | | Spectrum | | 1D NMR | 1H NMR Spectrum (1D, 200 MHz, D2O, predicted) | | Spectrum | | 1D NMR | 13C NMR Spectrum (1D, 300 MHz, D2O, predicted) | | Spectrum | | 1D NMR | 1H NMR Spectrum (1D, 300 MHz, D2O, predicted) | | Spectrum | | 1D NMR | 13C NMR Spectrum (1D, 400 MHz, D2O, predicted) | | Spectrum | | 1D NMR | 1H NMR Spectrum (1D, 400 MHz, D2O, predicted) | | Spectrum | | 1D NMR | 13C NMR Spectrum (1D, 500 MHz, D2O, predicted) | | Spectrum | | 1D NMR | 1H NMR Spectrum (1D, 500 MHz, D2O, predicted) | | Spectrum | | 1D NMR | 13C NMR Spectrum (1D, 600 MHz, D2O, predicted) | | Spectrum | | 1D NMR | 1H NMR Spectrum (1D, 600 MHz, D2O, predicted) | | Spectrum | | 1D NMR | 13C NMR Spectrum (1D, 700 MHz, D2O, predicted) | | Spectrum | | 1D NMR | 1H NMR Spectrum (1D, 700 MHz, D2O, predicted) | | Spectrum | | 1D NMR | 13C NMR Spectrum (1D, 800 MHz, D2O, predicted) | | Spectrum | | 1D NMR | 1H NMR Spectrum (1D, 800 MHz, D2O, predicted) | | Spectrum | | 1D NMR | 13C NMR Spectrum (1D, 900 MHz, D2O, predicted) | | Spectrum | | 1D NMR | 1H NMR Spectrum (1D, 900 MHz, D2O, predicted) | | Spectrum | | 2D NMR | [1H, 13C]-HSQC NMR Spectrum (2D, 600 MHz, H2O, experimental) | | Spectrum |
|
|---|
| Pathways |
|---|
| Pathways | | Name | SMPDB/Pathwhiz | KEGG | | Lysine Degradation | 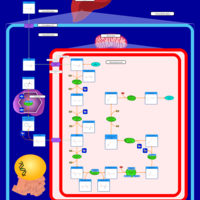 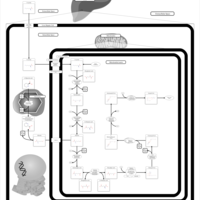  | 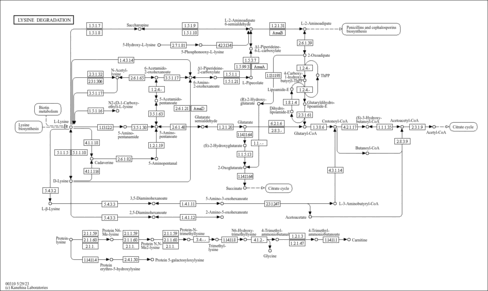 | | Saccharopinuria/Hyperlysinemia II | 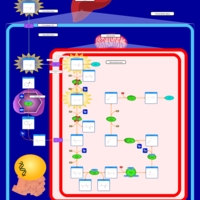 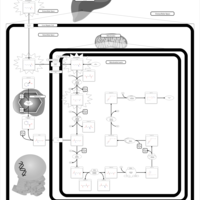 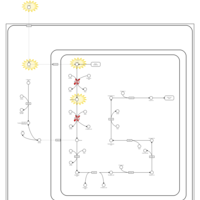 | Not Available | | Hyperlysinemia I, Familial | 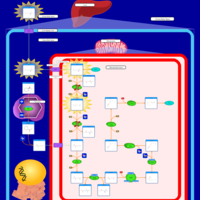 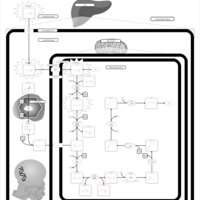 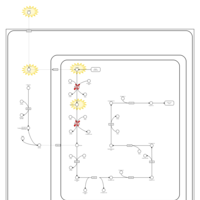 | Not Available | | Hyperlysinemia II or Saccharopinuria | 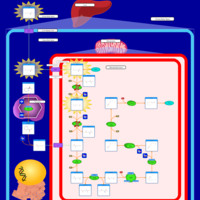 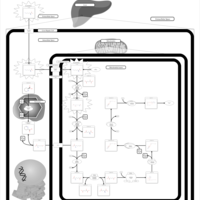  | Not Available | | Pyridoxine dependency with seizures |  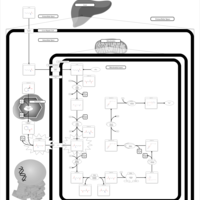 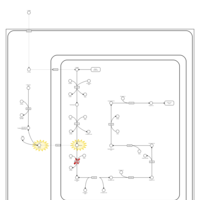 | Not Available |
|
|---|
| Protein Targets |
|---|
| Enzymes | |
| Alpha-aminoadipic semialdehyde synthase, mitochondrial | AASS | 7q31.3 | Q9UDR5 | details | | Probable saccharopine dehydrogenase | SCCPDH | 1q44 | Q8NBX0 | details | | Aminoadipate-semialdehyde synthase | AASS | 7q31.3 | A4D0W4 | details |
|
|---|
| Transporters | Not Available |
|---|
| Metal Bindings | Not Available |
|---|
| Receptors | Not Available |
|---|
| Transcriptional Factors | Not Available |
|---|
| Concentrations Data |
|---|
| Not Available |
|---|
| External Links |
|---|
| HMDB ID | HMDB0000279 |
|---|
| DrugBank ID | DB04207 |
|---|
| Phenol Explorer Compound ID | Not Available |
|---|
| FoodDB ID | FDB000461 |
|---|
| KNApSAcK ID | C00007227 |
|---|
| Chemspider ID | 141086 |
|---|
| KEGG Compound ID | C00449 |
|---|
| BioCyc ID | SACCHAROPINE |
|---|
| BiGG ID | 1484994 |
|---|
| Wikipedia Link | Saccharopine |
|---|
| METLIN ID | 383 |
|---|
| PubChem Compound | 160556 |
|---|
| PDB ID | Not Available |
|---|
| ChEBI ID | 16927 |
|---|
| References |
|---|
| General References | Not Available |
|---|
