| Record Information |
|---|
| Version | 1.0 |
|---|
| Created at | 2020-04-17 18:37:15 UTC |
|---|
| Updated at | 2020-12-07 19:10:57 UTC |
|---|
| CannabisDB ID | CDB004792 |
|---|
| Secondary Accession Numbers | Not Available |
|---|
| Cannabis Compound Identification |
|---|
| Common Name | beta-Alanine |
|---|
| Description | beta-Alanine, also known as 3-aminopropanoate or bala, belongs to the class of organic compounds known as beta amino acids and derivatives. These are amino acids having a (-NH2) group attached to the beta carbon atom. beta-Alanine is a very strong basic compound (based on its pKa). beta-Alanine exists in all living species, ranging from bacteria to humans. beta-Alanine is a sweet and odorless tasting compound. Outside of the human body, beta-Alanine is found, on average, in the highest concentration within a few different foods, such as beers, blackcurrants, and garden tomato (var.). beta-Alanine has also been detected, but not quantified in, several different foods, such as apples, bitter gourds, carrots, and sour cherries. This could make beta-alanine a potential biomarker for the consumption of these foods. beta-Alanine is a potentially toxic compound. beta-Alanine, with regard to humans, has been found to be associated with several diseases such as methylmalonate semialdehyde dehydrogenase deficiency, lewy body disease, crohn's disease, and perillyl alcohol administration for cancer treatment; beta-alanine has also been linked to the inborn metabolic disorder dihydropyrimidine dehydrogenase deficiency. A naturally-occurring beta-amino acid comprising propionic acid with the amino group in the 3-position. beta-Alanine is expected to be in Cannabis as all living plants are known to produce and metabolize it. |
|---|
| Structure | |
|---|
| Synonyms | | Value | Source |
|---|
| 3-Aminopropanoic acid | ChEBI | | 3-Aminopropionic acid | ChEBI | | BAla | ChEBI | | beta-Aminopropionic acid | ChEBI | | H-beta-Ala-OH | ChEBI | | Omega-aminopropionic acid | ChEBI | | 3-Aminopropanoate | Kegg | | Abufene | Kegg | | 3-Aminopropionate | Generator | | b-Aminopropionate | Generator | | b-Aminopropionic acid | Generator | | beta-Aminopropionate | Generator | | Β-aminopropionate | Generator | | Β-aminopropionic acid | Generator | | H-b-Ala-OH | Generator | | H-Β-ala-OH | Generator | | Omega-aminopropionate | Generator | | b-Alanine | Generator | | Β-alanine | Generator | | 2-Carboxyethylamine | HMDB | | 3-Amino-propanoate | HMDB | | 3-Amino-propanoic acid | HMDB | | b-Aminopropanoate | HMDB | | b-Aminopropanoic acid | HMDB | | beta Alanine | HMDB | | beta-Aminopropanoate | HMDB | | beta-Aminopropanoic acid | HMDB | | 3 Aminopropionic acid | HMDB | | β-Aminopropanoic acid | PhytoBank | | omega-Aminopropanoic acid | PhytoBank | | ω-Aminopropanoic acid | PhytoBank | | ω-Aminopropionic acid | PhytoBank | | beta-Alanine | PhytoBank |
|
|---|
| Chemical Formula | C3H7NO2 |
|---|
| Average Molecular Weight | 89.09 |
|---|
| Monoisotopic Molecular Weight | 89.0477 |
|---|
| IUPAC Name | 3-aminopropanoic acid |
|---|
| Traditional Name | β alanine |
|---|
| CAS Registry Number | 107-95-9 |
|---|
| SMILES | NCCC(O)=O |
|---|
| InChI Identifier | InChI=1S/C3H7NO2/c4-2-1-3(5)6/h1-2,4H2,(H,5,6) |
|---|
| InChI Key | UCMIRNVEIXFBKS-UHFFFAOYSA-N |
|---|
| Chemical Taxonomy |
|---|
| Description | Belongs to the class of organic compounds known as beta amino acids and derivatives. These are amino acids having a (-NH2) group attached to the beta carbon atom. |
|---|
| Kingdom | Organic compounds |
|---|
| Super Class | Organic acids and derivatives |
|---|
| Class | Carboxylic acids and derivatives |
|---|
| Sub Class | Amino acids, peptides, and analogues |
|---|
| Direct Parent | Beta amino acids and derivatives |
|---|
| Alternative Parents | |
|---|
| Substituents | - Beta amino acid or derivatives
- Amino acid
- Carboxylic acid
- Monocarboxylic acid or derivatives
- Amine
- Organic oxide
- Hydrocarbon derivative
- Primary amine
- Organooxygen compound
- Organonitrogen compound
- Organopnictogen compound
- Primary aliphatic amine
- Organic oxygen compound
- Carbonyl group
- Organic nitrogen compound
- Aliphatic acyclic compound
|
|---|
| Molecular Framework | Aliphatic acyclic compounds |
|---|
| External Descriptors | |
|---|
| Ontology |
|---|
|
| Physiological effect | Health effect: |
|---|
| Disposition | Route of exposure: Source: Biological location: |
|---|
| Role | Indirect biological role: Biological role: Environmental role: Industrial application: |
|---|
| Physical Properties |
|---|
| State | Solid |
|---|
| Experimental Properties | | Property | Value | Reference |
|---|
| Melting Point | 200 °C | Not Available | | Boiling Point | Not Available | Not Available | | Water Solubility | 545 mg/mL | Not Available | | logP | -3.05 | TSAI,RS ET AL. (1991) |
|
|---|
| Predicted Properties | [] |
|---|
| Spectra |
|---|
| EI-MS/GC-MS | | Type | Description | Splash Key | View |
|---|
| EI-MS | Mass Spectrum (Electron Ionization) | splash10-001i-9000000000-be815b0a56f225da04d8 | 2014-09-20 | View Spectrum | | GC-MS | beta-Alanine, 3 TMS, GC-MS Spectrum | splash10-007k-1920000000-9389ee061bd6e9ae3b22 | Spectrum | | GC-MS | beta-Alanine, 3 TMS, GC-MS Spectrum | splash10-0fdt-2930000000-5799fddb7fac04d22372 | Spectrum | | GC-MS | beta-Alanine, 3 TMS, GC-MS Spectrum | splash10-006t-1940000000-49f1b7c80f35cbcf102e | Spectrum | | GC-MS | beta-Alanine, non-derivatized, GC-MS Spectrum | splash10-006t-2920000000-c701f2e5652fa4f35e50 | Spectrum | | GC-MS | beta-Alanine, 3 TMS, GC-MS Spectrum | splash10-00di-8930000000-c4f8138d4cb7f1ea851a | Spectrum | | GC-MS | beta-Alanine, 1 TMS, GC-MS Spectrum | splash10-014i-2900000000-c54f1d5878600393dca7 | Spectrum | | GC-MS | beta-Alanine, 2 TMS, GC-MS Spectrum | splash10-0udi-1900000000-2f55a09bba074034dee7 | Spectrum | | GC-MS | beta-Alanine, 3 TMS, GC-MS Spectrum | splash10-00ds-2940000000-4192453eaba2cbecf800 | Spectrum | | GC-MS | beta-Alanine, non-derivatized, GC-MS Spectrum | splash10-006t-1960000000-665868f9adaa275dbdb3 | Spectrum | | GC-MS | beta-Alanine, non-derivatized, GC-MS Spectrum | splash10-007k-1920000000-9389ee061bd6e9ae3b22 | Spectrum | | GC-MS | beta-Alanine, non-derivatized, GC-MS Spectrum | splash10-0fdt-2930000000-5799fddb7fac04d22372 | Spectrum | | GC-MS | beta-Alanine, non-derivatized, GC-MS Spectrum | splash10-006t-1940000000-49f1b7c80f35cbcf102e | Spectrum | | GC-MS | beta-Alanine, non-derivatized, GC-MS Spectrum | splash10-006t-2920000000-c701f2e5652fa4f35e50 | Spectrum | | GC-MS | beta-Alanine, non-derivatized, GC-MS Spectrum | splash10-00di-8930000000-c4f8138d4cb7f1ea851a | Spectrum | | GC-MS | beta-Alanine, non-derivatized, GC-MS Spectrum | splash10-00ds-2940000000-4192453eaba2cbecf800 | Spectrum | | GC-MS | beta-Alanine, non-derivatized, GC-MS Spectrum | splash10-014i-2900000000-c54f1d5878600393dca7 | Spectrum | | GC-MS | beta-Alanine, non-derivatized, GC-MS Spectrum | splash10-0udi-1900000000-2f55a09bba074034dee7 | Spectrum | | GC-MS | beta-Alanine, non-derivatized, GC-MS Spectrum | splash10-0udi-0900000000-efc0822e26bcf842e232 | Spectrum | | GC-MS | beta-Alanine, non-derivatized, GC-MS Spectrum | splash10-0072-2920000000-284cff207c712b6698d6 | Spectrum | | Predicted GC-MS | beta-Alanine, non-derivatized, Predicted GC-MS Spectrum - 70eV, Positive | splash10-001i-9000000000-ad7f96b2c6a1f5183794 | Spectrum | | Predicted GC-MS | beta-Alanine, 1 TMS, Predicted GC-MS Spectrum - 70eV, Positive | splash10-0fk9-9700000000-d73baf312b8af27575b3 | Spectrum | | Predicted GC-MS | beta-Alanine, non-derivatized, Predicted GC-MS Spectrum - 70eV, Positive | Not Available | Spectrum | | Predicted GC-MS | beta-Alanine, TMS_1_2, Predicted GC-MS Spectrum - 70eV, Positive | Not Available | Spectrum | | Predicted GC-MS | beta-Alanine, TBDMS_1_1, Predicted GC-MS Spectrum - 70eV, Positive | Not Available | Spectrum | | Predicted GC-MS | beta-Alanine, TBDMS_1_2, Predicted GC-MS Spectrum - 70eV, Positive | Not Available | Spectrum |
|
|---|
| MS/MS | | Type | Description | Splash Key | View |
|---|
| MS/MS | LC-MS/MS Spectrum - LC-ESI-QQ (API3000, Applied Biosystems) 10V, Negative | splash10-000i-9000000000-61740a52186a51b8543c | 2012-08-31 | View Spectrum | | MS/MS | LC-MS/MS Spectrum - LC-ESI-QQ (API3000, Applied Biosystems) 20V, Negative | splash10-000i-9000000000-1bdf967935160468d550 | 2012-08-31 | View Spectrum | | MS/MS | LC-MS/MS Spectrum - LC-ESI-QQ (API3000, Applied Biosystems) 30V, Negative | splash10-000i-9000000000-0ba5264cfc5ec108718b | 2012-08-31 | View Spectrum | | MS/MS | LC-MS/MS Spectrum - LC-ESI-QQ , negative | splash10-000i-9000000000-61740a52186a51b8543c | 2017-09-14 | View Spectrum | | MS/MS | LC-MS/MS Spectrum - LC-ESI-QQ , negative | splash10-000i-9000000000-1bdf967935160468d550 | 2017-09-14 | View Spectrum | | MS/MS | LC-MS/MS Spectrum - LC-ESI-QQ , negative | splash10-000i-9000000000-0ba5264cfc5ec108718b | 2017-09-14 | View Spectrum | | MS/MS | LC-MS/MS Spectrum - 40V, Negative | splash10-000i-9000000000-1c44f6e35a8ed7d36b09 | 2021-09-20 | View Spectrum | | MS/MS | LC-MS/MS Spectrum - 20V, Negative | splash10-00di-9000000000-799cbaba87e33fa5afde | 2021-09-20 | View Spectrum | | MS/MS | LC-MS/MS Spectrum - 10V, Negative | splash10-000i-9000000000-86d934e6151015ddb810 | 2021-09-20 | View Spectrum | | MS/MS | LC-MS/MS Spectrum - Quattro_QQQ 10V, Positive (Annotated) | splash10-00dl-9000000000-c30cc77c93e4ec09265a | 2012-07-24 | View Spectrum | | MS/MS | LC-MS/MS Spectrum - Quattro_QQQ 25V, Positive (Annotated) | splash10-000j-9000000000-540a52fcec629c0a0647 | 2012-07-24 | View Spectrum | | MS/MS | LC-MS/MS Spectrum - Quattro_QQQ 40V, Positive (Annotated) | splash10-000b-9000000000-531b17f8e16896225186 | 2012-07-24 | View Spectrum | | MS/MS | LC-MS/MS Spectrum - LC-ESI-QQ (API3000, Applied Biosystems) 10V, Positive | splash10-0006-9000000000-c6a2c78d322328400127 | 2012-08-31 | View Spectrum | | MS/MS | LC-MS/MS Spectrum - LC-ESI-QQ (API3000, Applied Biosystems) 20V, Positive | splash10-00di-9000000000-d5f47100c42e7777802e | 2012-08-31 | View Spectrum | | MS/MS | LC-MS/MS Spectrum - LC-ESI-QQ (API3000, Applied Biosystems) 30V, Positive | splash10-001i-9000000000-e8f43be75c153e5be3cc | 2012-08-31 | View Spectrum | | MS/MS | LC-MS/MS Spectrum - LC-ESI-QQ (API3000, Applied Biosystems) 40V, Positive | splash10-1004-9000100000-ccb09493216d97165c8d | 2012-08-31 | View Spectrum | | MS/MS | LC-MS/MS Spectrum - LC-ESI-QQ (API3000, Applied Biosystems) 50V, Positive | splash10-00l7-9230000000-644b1dbe52c5f07e285a | 2012-08-31 | View Spectrum | | MS/MS | LC-MS/MS Spectrum - LC-ESI-QTOF (UPLC Q-Tof Premier, Waters) , Positive | splash10-0006-9000000000-ff4a990aa1fb1b7cc33d | 2012-08-31 | View Spectrum | | MS/MS | LC-MS/MS Spectrum - LC-ESI-QQ , positive | splash10-0006-9000000000-c6a2c78d322328400127 | 2017-09-14 | View Spectrum | | MS/MS | LC-MS/MS Spectrum - LC-ESI-QQ , positive | splash10-00di-9000000000-d5f47100c42e7777802e | 2017-09-14 | View Spectrum | | MS/MS | LC-MS/MS Spectrum - LC-ESI-QQ , positive | splash10-001i-9000000000-e8f43be75c153e5be3cc | 2017-09-14 | View Spectrum | | MS/MS | LC-MS/MS Spectrum - LC-ESI-QQ , positive | splash10-1004-9000100000-ccb09493216d97165c8d | 2017-09-14 | View Spectrum | | MS/MS | LC-MS/MS Spectrum - LC-ESI-QQ , positive | splash10-00l7-9230000000-644b1dbe52c5f07e285a | 2017-09-14 | View Spectrum | | MS/MS | LC-MS/MS Spectrum - LC-ESI-QTOF , positive | splash10-0006-9000000000-ff4a990aa1fb1b7cc33d | 2017-09-14 | View Spectrum | | MS/MS | LC-MS/MS Spectrum - 10V, Positive | splash10-00dl-9000000000-363393d05fd6a4b82318 | 2021-09-20 | View Spectrum |
|
|---|
| NMR | | Type | Description | | View |
|---|
| 1D NMR | 1H NMR Spectrum (1D, 600 MHz, H2O, experimental) | | Spectrum | | 1D NMR | 13C NMR Spectrum (1D, 125 MHz, H2O, experimental) | | Spectrum | | 1D NMR | 1H NMR Spectrum (1D, 400 MHz, D2O, experimental) | | Spectrum | | 1D NMR | 13C NMR Spectrum (1D, 25.16 MHz, D2O, experimental) | | Spectrum | | 1D NMR | 13C NMR Spectrum (1D, 400 MHz, H2O, experimental) | | Spectrum | | 2D NMR | [1H, 1H]-TOCSY. Unexported temporarily by An Chi on Oct 15, 2021 until json or nmrML file is generated. 2D NMR Spectrum (experimental) | | Spectrum | | 2D NMR | [1H, 13C]-HSQC NMR Spectrum (2D, 600 MHz, H2O, experimental) | | Spectrum |
|
|---|
| Pathways |
|---|
| Pathways | | Name | SMPDB/Pathwhiz | KEGG | | Propanoate Metabolism |    |  | | Aspartate Metabolism | 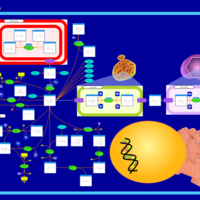 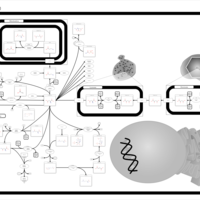 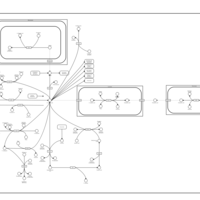 |  | | Beta-Alanine Metabolism | 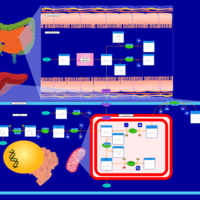 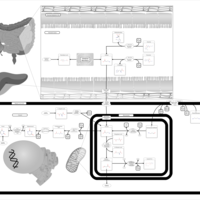 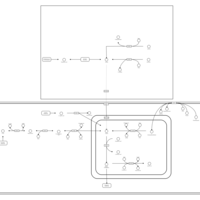 | 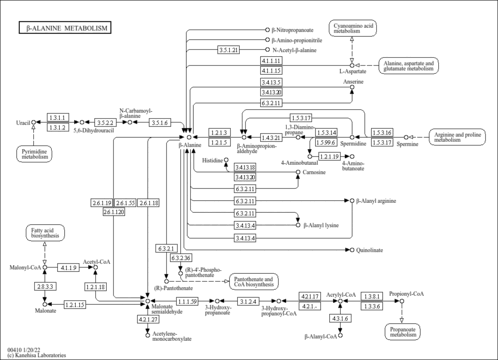 | | Pyrimidine Metabolism |  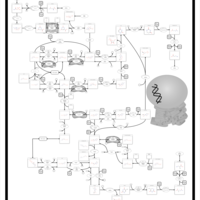  |  | | Canavan Disease | 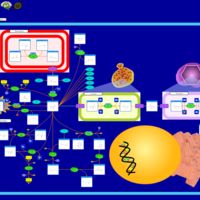 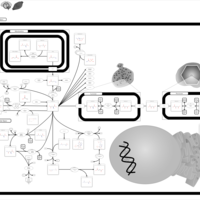 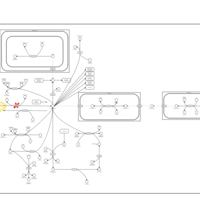 | Not Available |
|
|---|
| Protein Targets |
|---|
| Enzymes | |
|---|
| Transporters | |
| Sodium- and chloride-dependent taurine transporter | SLC6A6 | 3p25.1 | P31641 | details | | Monocarboxylate transporter 10 | SLC16A10 | 6q21-q22 | Q8TF71 | details |
|
|---|
| Metal Bindings | |
|---|
| Receptors | Not Available |
|---|
| Transcriptional Factors | Not Available |
|---|
| Concentrations Data |
|---|
| |
| Alien Dawg | Detected and Quantified | 0.26 mg/g dry wt | | details | | Sensi Star | Detected and Quantified | 0.39 mg/g dry wt | | details | | Tangerine Dream | Detected and Quantified | 0.39 mg/g dry wt | | details |
|
|---|
| External Links |
|---|
| HMDB ID | HMDB0000056 |
|---|
| DrugBank ID | DB03107 |
|---|
| Phenol Explorer Compound ID | Not Available |
|---|
| FoodDB ID | FDB002253 |
|---|
| KNApSAcK ID | C00001333 |
|---|
| Chemspider ID | 234 |
|---|
| KEGG Compound ID | C00099 |
|---|
| BioCyc ID | B-ALANINE |
|---|
| BiGG ID | 33848 |
|---|
| Wikipedia Link | Beta-Alanine |
|---|
| METLIN ID | 5119 |
|---|
| PubChem Compound | 239 |
|---|
| PDB ID | Not Available |
|---|
| ChEBI ID | 16958 |
|---|
| References |
|---|
| General References | Not Available |
|---|



