| Record Information |
|---|
| Version | 1.0 |
|---|
| Created at | 2020-03-19 00:27:52 UTC |
|---|
| Updated at | 2020-12-07 19:07:18 UTC |
|---|
| CannabisDB ID | CDB000267 |
|---|
| Secondary Accession Numbers | Not Available |
|---|
| Cannabis Compound Identification |
|---|
| Common Name | hexadecanol-1 |
|---|
| Description | Cetyl alcohol, also known as 1-hexadecanol and palmityl alcohol, is a solid organic compound and a member of the alcohol class of compounds. Its chemical formula is CH3(CH2)15OH. At room temperature, Cetyl alcohol takes the form of a waxy white solid or flakes. It belongs to the group of fatty alcohols. With the demise of commercial whaling, cetyl alcohol is no longer primarily produced from whale oil, but instead either as an end-product of the petroleum industry or produced from vegetable oils such as palm oil and coconut oil. Production of cetyl alcohol from palm oil gives rise to one of its alternative names, palmityl alcohol. 1-Hexadecanol is a very hydrophobic molecule, practically insoluble in water, and relatively neutral. 1-Hexadecanol is one of the simple alcohols found in cannabis (PMID: 6991645 ). It has a waxy or oily aroma and a “clean cloth” waxy or oily taste. It is used in the food and cosmetic industry as an emollient, an emulsifying agent, a foam boosting agent, a masking agent, a surfactant, a viscosity controlling agent and a pacifying agent. Hexadecanol is found in, or is added to, shampoos, conditioners, toothpaste, inks, paints, pharmaceuticals, skin creams and lotions. It is also added to certain hard candies and frozen treats. People who suffer from eczema can be sensitive to hexadecanol as it is sometimes included in medications used for the treatment of eczema (PMID: 15059111 ). Cetyl alcohol is also found in cannabis smoke and is volatilized during the combustion of cannabis ( Ref:DOI ). |
|---|
| Structure | |
|---|
| Synonyms | | Value | Source |
|---|
| 1-Cetanol | ChEBI | | 1-Hexadecyl alcohol | ChEBI | | 16-Hexadecanol | ChEBI | | Cetanol | ChEBI | | Cetyl alcohol | ChEBI | | Cetylalkohol | ChEBI | | Hexadecanol | ChEBI | | N-1-Hexadecanol | ChEBI | | N-Hexadecyl alcohol | ChEBI | | Palmityl alcohol | ChEBI | | 1-Hexadecyl alc | HMDB | | 1-Hexanedecanol | HMDB | | 1-Hydroxyhexadecane | HMDB | | Adol | HMDB | | Adol 52 | HMDB | | Adol 52 NF | HMDB | | Adol 52nf | HMDB | | Adol 54 | HMDB | | Alcohol C-16 | HMDB | | Aldol 54 | HMDB | | Alfol 16 | HMDB | | Atalco C | HMDB | | C16 Alcohol | HMDB | | Cachalot C-50 | HMDB | | Cachalot C-50 NF | HMDB | | Cachalot C-51 | HMDB | | Cachalot C-52 | HMDB | | Ceraphyl ica | HMDB | | Cetaffine | HMDB | | Cetal | HMDB | | Cetalol ca | HMDB | | Cetostearyl alcohol | HMDB | | Cetyl alcohol NF | HMDB | | Cetylic alcohol | HMDB | | Cetylol | HMDB | | Crodacol C | HMDB | | Crodacol C70 | HMDB | | Crodacol C95NF | HMDB | | Crodacol-cas | HMDB | | Crodacol-cat | HMDB | | Cyclal cetyl alcohol | HMDB | | Dehydag wax 16 | HMDB | | Dytol F-11 | HMDB | | Elfacos C | HMDB | | Epal 16nf | HMDB | | Ethal | HMDB | | Ethol | HMDB | | Eutanol g16 | HMDB | | Exxal 16 | HMDB | | Fancol ca | HMDB | | Fatty alcohol | HMDB | | Hexadecan-1-ol | HMDB | | Hexadecanol NF | HMDB | | Hexadecyl alcohol | HMDB | | Hyfatol | HMDB | | Hyfatol 16 | HMDB | | Isocetyl alcohol | HMDB | | Isohexadecanol | HMDB | | Isohexadecyl alcohol | HMDB | | Lanette 16 | HMDB | | Lanol C | HMDB | | Lipocol C | HMDB | | Lorol 24 | HMDB | | Lorol C16 | HMDB | | LorolL 24 | HMDB | | Loxanol K | HMDB | | Loxanol K extra | HMDB | | Loxanwachs SK | HMDB | | Loxiol VPG 1743 | HMDB | | Michel xo-150-16 | HMDB | | Myristyl alcohol | HMDB | | N-Cetyl alcohol | HMDB | | N-Hexadecan-1-ol | HMDB | | N-Hexadecanol | HMDB | | Normal primary hexadecyl alcohol | HMDB | | Philcohol 1600 | HMDB | | Product 308 | HMDB | | Rita ca | HMDB | | Siponol CC | HMDB | | Siponol wax-a | HMDB | | SSD | HMDB | | SSD RP | HMDB | | Cetyl alcohol, 14C-labeled | HMDB | | Cetyl alcohol, aluminum salt | HMDB | | 1-Hexadecanol | ChEBI |
|
|---|
| Chemical Formula | C16H34O |
|---|
| Average Molecular Weight | 242.44 |
|---|
| Monoisotopic Molecular Weight | 242.261 |
|---|
| IUPAC Name | hexadecan-1-ol |
|---|
| Traditional Name | cetyl alcohol |
|---|
| CAS Registry Number | 36311-34-9 |
|---|
| SMILES | CCCCCCCCCCCCCCCCO |
|---|
| InChI Identifier | InChI=1S/C16H34O/c1-2-3-4-5-6-7-8-9-10-11-12-13-14-15-16-17/h17H,2-16H2,1H3 |
|---|
| InChI Key | BXWNKGSJHAJOGX-UHFFFAOYSA-N |
|---|
| Chemical Taxonomy |
|---|
| Description | Belongs to the class of organic compounds known as long-chain fatty alcohols. These are fatty alcohols that have an aliphatic tail of 13 to 21 carbon atoms. |
|---|
| Kingdom | Organic compounds |
|---|
| Super Class | Lipids and lipid-like molecules |
|---|
| Class | Fatty Acyls |
|---|
| Sub Class | Fatty alcohols |
|---|
| Direct Parent | Long-chain fatty alcohols |
|---|
| Alternative Parents | |
|---|
| Substituents | - Long chain fatty alcohol
- Organic oxygen compound
- Hydrocarbon derivative
- Primary alcohol
- Organooxygen compound
- Alcohol
- Aliphatic acyclic compound
|
|---|
| Molecular Framework | Aliphatic acyclic compounds |
|---|
| External Descriptors | |
|---|
| Ontology |
|---|
|
| Disposition | Route of exposure: Source: Biological location: |
|---|
| Role | Biological role: Industrial application: |
|---|
| Physical Properties |
|---|
| State | Solid |
|---|
| Experimental Properties | | Property | Value | Reference |
|---|
| Melting Point | 49.3 °C | Not Available | | Boiling Point | Not Available | Not Available | | Water Solubility | 1.3e-05 mg/mL at 25 °C | Not Available | | logP | Not Available | Not Available |
|
|---|
| Predicted Properties | [] |
|---|
| Spectra |
|---|
| EI-MS/GC-MS | | Type | Description | Splash Key | View |
|---|
| EI-MS | Mass Spectrum (Electron Ionization) | splash10-0a5c-9000000000-f3299e52ea0593788e37 | 2014-09-20 | View Spectrum | | GC-MS | hexadecanol-1, non-derivatized, GC-MS Spectrum | splash10-0f72-7931000000-00a8faa3fd4f0c38a405 | Spectrum | | GC-MS | hexadecanol-1, 1 TMS, GC-MS Spectrum | splash10-0f6t-9531000000-0dc2948d10d835886750 | Spectrum | | GC-MS | hexadecanol-1, non-derivatized, GC-MS Spectrum | splash10-0a4l-9000000000-1be6f87af09f0bc4339f | Spectrum | | GC-MS | hexadecanol-1, non-derivatized, GC-MS Spectrum | splash10-0a4l-9000000000-83f0c3f9a7d1a2d70b83 | Spectrum | | GC-MS | hexadecanol-1, non-derivatized, GC-MS Spectrum | splash10-0a59-9100000000-51d2315e018d6ebbe25d | Spectrum | | GC-MS | hexadecanol-1, non-derivatized, GC-MS Spectrum | splash10-0f72-7931000000-00a8faa3fd4f0c38a405 | Spectrum | | GC-MS | hexadecanol-1, non-derivatized, GC-MS Spectrum | splash10-0f6t-9531000000-0dc2948d10d835886750 | Spectrum | | GC-MS | hexadecanol-1, non-derivatized, GC-MS Spectrum | splash10-0f72-9821000000-ee82a405e4a854526b64 | Spectrum | | Predicted GC-MS | hexadecanol-1, non-derivatized, Predicted GC-MS Spectrum - 70eV, Positive | splash10-0bvl-7910000000-c4f032970214d082f3cf | Spectrum | | Predicted GC-MS | hexadecanol-1, 1 TMS, Predicted GC-MS Spectrum - 70eV, Positive | splash10-00di-9640000000-6df3de611756e2218c31 | Spectrum | | Predicted GC-MS | hexadecanol-1, non-derivatized, Predicted GC-MS Spectrum - 70eV, Positive | Not Available | Spectrum |
|
|---|
| MS/MS | | Type | Description | Splash Key | View |
|---|
| MS/MS | LC-MS/MS Spectrum - Quattro_QQQ 10V, Negative (Annotated) | splash10-0udi-0910000000-b74545433556690d2c46 | 2012-07-25 | View Spectrum | | MS/MS | LC-MS/MS Spectrum - Quattro_QQQ 25V, Negative (Annotated) | splash10-0udi-0900000000-7aab213ad736e774476c | 2012-07-25 | View Spectrum | | MS/MS | LC-MS/MS Spectrum - Quattro_QQQ 40V, Negative (Annotated) | splash10-0udi-0900000000-55a8272e13510c85c06a | 2012-07-25 | View Spectrum | | MS/MS | LC-MS/MS Spectrum - EI-B (HITACHI M-80B) , Positive | splash10-0a4l-9000000000-a4776db6cf6d30dfb50e | 2012-08-31 | View Spectrum | | MS/MS | LC-MS/MS Spectrum - EI-B (SHIMADZU QP-1000) , Positive | splash10-0a4l-9000000000-83f0c3f9a7d1a2d70b83 | 2012-08-31 | View Spectrum | | MS/MS | LC-MS/MS Spectrum - EI-B (JEOL JMS-HX-100) , Positive | splash10-0a59-9100000000-7ca85869bfef10e335f3 | 2012-08-31 | View Spectrum | | Predicted MS/MS | Predicted LC-MS/MS Spectrum - 10V, Positive | splash10-004l-0190000000-b08f970abae4433e0d8b | 2017-07-26 | View Spectrum | | Predicted MS/MS | Predicted LC-MS/MS Spectrum - 20V, Positive | splash10-004l-5690000000-d0c4cde89f48e5218c95 | 2017-07-26 | View Spectrum | | Predicted MS/MS | Predicted LC-MS/MS Spectrum - 40V, Positive | splash10-052f-9500000000-455427a3ee7620a41403 | 2017-07-26 | View Spectrum | | Predicted MS/MS | Predicted LC-MS/MS Spectrum - 10V, Negative | splash10-0006-0090000000-3fdb1b4b343c7ad295bf | 2017-07-26 | View Spectrum | | Predicted MS/MS | Predicted LC-MS/MS Spectrum - 20V, Negative | splash10-0006-0090000000-a848ffe1f9f31e21601c | 2017-07-26 | View Spectrum | | Predicted MS/MS | Predicted LC-MS/MS Spectrum - 40V, Negative | splash10-01vp-9740000000-15fb3da54ceed633096b | 2017-07-26 | View Spectrum | | Predicted MS/MS | Predicted LC-MS/MS Spectrum - 10V, Positive | splash10-0006-4190000000-44af6a0edc2288305d9c | 2021-09-22 | View Spectrum | | Predicted MS/MS | Predicted LC-MS/MS Spectrum - 20V, Positive | splash10-0abc-9110000000-a07d84c82c62e530754e | 2021-09-22 | View Spectrum | | Predicted MS/MS | Predicted LC-MS/MS Spectrum - 40V, Positive | splash10-0a4l-9000000000-dc9968082483b614a723 | 2021-09-22 | View Spectrum | | Predicted MS/MS | Predicted LC-MS/MS Spectrum - 10V, Negative | splash10-0006-0090000000-3ce1eb2d48c904ebe347 | 2021-09-23 | View Spectrum | | Predicted MS/MS | Predicted LC-MS/MS Spectrum - 20V, Negative | splash10-0006-0090000000-e0b832e3e0af19f4f9ae | 2021-09-23 | View Spectrum | | Predicted MS/MS | Predicted LC-MS/MS Spectrum - 40V, Negative | splash10-0006-9880000000-0db321e9b8178e2cdc8f | 2021-09-23 | View Spectrum |
|
|---|
| NMR | | Type | Description | | View |
|---|
| 1D NMR | 1H NMR Spectrum (1D, 600 MHz, H2O, experimental) | | Spectrum | | 1D NMR | 13C NMR Spectrum (1D, 25.16 MHz, CDCl3, experimental) | | Spectrum | | 2D NMR | [1H, 13C]-HSQC NMR Spectrum (2D, 600 MHz, CD3OD, experimental) | | Spectrum |
|
|---|
| Pathways |
|---|
| Pathways | | Name | SMPDB/Pathwhiz | KEGG | | Plasmalogen Synthesis | 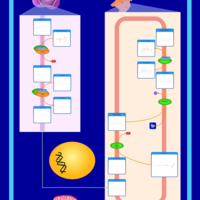 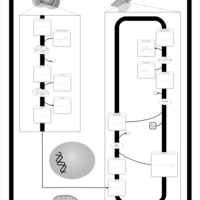 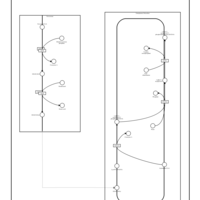 | Not Available |
|
|---|
| Protein Targets |
|---|
| Enzymes | |
|---|
| Transporters | |
|---|
| Metal Bindings | Not Available |
|---|
| Receptors | |
|---|
| Transcriptional Factors | Not Available |
|---|
| Concentrations Data |
|---|
| Not Available |
|---|
| External Links |
|---|
| HMDB ID | HMDB0003424 |
|---|
| DrugBank ID | DB09494 |
|---|
| Phenol Explorer Compound ID | Not Available |
|---|
| FoodDB ID | FDB003051 |
|---|
| KNApSAcK ID | Not Available |
|---|
| Chemspider ID | 2581 |
|---|
| KEGG Compound ID | C00823 |
|---|
| BioCyc ID | CPD-348 |
|---|
| BiGG ID | Not Available |
|---|
| Wikipedia Link | Cetyl_Alcohol |
|---|
| METLIN ID | 6927 |
|---|
| PubChem Compound | 2682 |
|---|
| PDB ID | Not Available |
|---|
| ChEBI ID | 16125 |
|---|
| References |
|---|
| General References | - Turner CE, Elsohly MA, Boeren EG: Constituents of Cannabis sativa L. XVII. A review of the natural constituents. J Nat Prod. 1980 Mar-Apr;43(2):169-234. doi: 10.1021/np50008a001. [PubMed:6991645 ]
- Soga F, Katoh N, Kishimoto S: Contact dermatitis due to lanoconazole, cetyl alcohol and diethyl sebacate in lanoconazole cream. Contact Dermatitis. 2004 Jan;50(1):49-50. doi: 10.1111/j.0105-1873.2004.00271j.x. [PubMed:15059111 ]
|
|---|