| Record Information |
|---|
| Version | 1.0 |
|---|
| Created at | 2020-03-18 23:25:58 UTC |
|---|
| Updated at | 2022-12-13 23:36:23 UTC |
|---|
| CannabisDB ID | CDB000132 |
|---|
| Secondary Accession Numbers | Not Available |
|---|
| Cannabis Compound Identification |
|---|
| Common Name | L-Isoleucine |
|---|
| Description | L-isoleucine or isoleucine, abbreviated as Ile or I, is one of the essential amino acids used in the biosynthesis of proteins. It contains an α-amino group (which is in the protonated −NH3+ form under biological conditions), an α-carboxylic acid group (which is in the deprotonated −COO− form under biological conditions), and a hydrocarbon side chain with a branch (a central carbon atom bound to three other carbon atoms). It is classified as a non-polar, uncharged (at physiological pH), branched-chain, aliphatic amino acid. In plants and microorganisms, it is synthesized via several steps, starting from pyruvate and alpha-ketobutyrate. Isoleucine is one of the three branched chain amino acids (BCAA), together with leucine and valine. Branched chain amino acids (BCAA) are essential amino acids whose carbon structure is marked by a branch point. Isoleucine is less concentrated in foods compared to leucine and valine. A cup of milk contains 800 mg of leucine and only 500 mg of isoleucine and valine. A cup of wheat germ has about 1.6 g of leucine and 1 g of isoleucine and valine. Eggs and cheese have about equal amounts of the BCAA and contain about 400 mg of leucine and 400 mg of valine and isoleucine. Pork contains 7 to 8 g of leucine and 3 to 4 g of isoleucine and valine. L-isoleucine is also found in Cannabis plants (PMID: 6991645 ). These BCAA are critical to human life and are particularly involved in stress, energy and muscle metabolism. BCAA supplementation as therapy, both oral and intravenous, in human health and disease holds great promise. Despite their structural similarities, the branched amino acids have different metabolic routes, with valine going solely to carbohydrates, leucine solely to fats and isoleucine to both. The different metabolism accounts for different requirements for these essential amino acids in humans: 12 mg/kg, 14 mg/kg and 16 mg/kg of valine, leucine, and isoleucine, respectively. Furthermore, these amino acids have different deficiency symptoms. Valine deficiency is marked by neurological defects in the brain, while isoleucine deficiency is marked by muscle tremors. BCAA are decreased in patients with liver disease, such as hepatitis, hepatic coma, cirrhosis, extrahepatic biliary atresia or portacaval shunt; aromatic amino acids (AAA)-tyrosine, tryptophan and phenylalanine, as well as methionine-are increased in these conditions. Valine has been established as a useful supplemental therapy to the ailing liver. All the BCAA probably compete with AAA for absorption into the brain. Supplemental BCAA with vitamin B6 and zinc help normalize the BCAA:AAA ratio. The BCAA are not without side effects. Leucine alone, for example, exacerbates pellagra and can cause psychosis in pellagra patients by increasing excretion of niacin in the urine. Leucine may lower brain serotonin and dopamine whereas isoleucine may have potential as an antipsychotic treatment. A dose of 3 g of isoleucine added to the niacin regime cleared leucine-aggravated psychosis in schizophrenic patients. Moreover, L-isoleucine is associated with maple syrup urine disease (MSUD; PMID: 10234605 ), which is an inborn error of metabolism, so named for the sweet, maple syrup smell of urine. MSUD is an autosomal recessive disorder that affects the metabolism of the BCAA. Mutations occur in the 4 genes that comprise the branched-chain alpha-keto acid dehydrogenase complex, which breaks down the BCAA. |
|---|
| Structure | |
|---|
| Synonyms | | Value | Source |
|---|
| (2S,3S)-2-Amino-3-methylpentanoic acid | ChEBI | | 2-Amino-3-methylvaleric acid | ChEBI | | alpha-Amino-beta-methylvaleric acid | ChEBI | | I | ChEBI | | Ile | ChEBI | | ISOLEUCINE | ChEBI | | (2S,3S)-2-Amino-3-methylpentanoate | Generator | | 2-Amino-3-methylvalerate | Generator | | a-Amino-b-methylvalerate | Generator | | a-Amino-b-methylvaleric acid | Generator | | alpha-Amino-beta-methylvalerate | Generator | | Α-amino-β-methylvalerate | Generator | | Α-amino-β-methylvaleric acid | Generator | | (2S,3S)-2-Amino-3-methyl-pentanoate | HMDB | | (2S,3S)-2-Amino-3-methyl-pentanoic acid | HMDB | | (2S,3S)-a-Amino-b-methyl-N-valerate | HMDB | | (2S,3S)-a-Amino-b-methyl-N-valeric acid | HMDB | | (2S,3S)-a-Amino-b-methylvalerate | HMDB | | (2S,3S)-a-Amino-b-methylvaleric acid | HMDB | | (2S,3S)-Alph-amino-beta-methylvalerate | HMDB | | (2S,3S)-Alph-amino-beta-methylvaleric acid | HMDB | | (2S,3S)-alpha-Amino-beta-merthyl-N-valerate | HMDB | | (2S,3S)-alpha-Amino-beta-merthyl-N-valeric acid | HMDB | | (2S,3S)-alpha-Amino-beta-merthylvalerate | HMDB | | (2S,3S)-alpha-Amino-beta-merthylvaleric acid | HMDB | | (2S,3S)-alpha-Amino-beta-methyl-N-valerate | HMDB | | (2S,3S)-alpha-Amino-beta-methyl-N-valeric acid | HMDB | | (2S,3S)-alpha-Amino-beta-methylvalerate | HMDB | | (2S,3S)-alpha-Amino-beta-methylvaleric acid | HMDB | | (S)-Isoleucine | HMDB | | (S,S)-Isoleucine | HMDB | | 2-Amino-3-methylpentanoate | HMDB | | 2-Amino-3-methylpentanoic acid | HMDB | | 2S,3S-Isoleucine | HMDB | | Erythro-L-isoleucine | HMDB | | Iso-leucine | HMDB | | L-(+)-Isoleucine | HMDB | | L-Ile | HMDB | | [S-(R*,r*)]-2-amino-3-methylpentanoate | HMDB | | [S-(R*,r*)]-2-amino-3-methylpentanoic acid | HMDB | | Isoleucine, L-isomer | HMDB | | Alloisoleucine | HMDB | | Isoleucine, L isomer | HMDB | | L-Isomer isoleucine | HMDB | | 2S-Amino-3S-methylpentanoate | HMDB | | L-Isoleucine | KEGG |
|
|---|
| Chemical Formula | C6H13NO2 |
|---|
| Average Molecular Weight | 131.17 |
|---|
| Monoisotopic Molecular Weight | 131.0946 |
|---|
| IUPAC Name | (2S,3S)-2-amino-3-methylpentanoic acid |
|---|
| Traditional Name | L-isoleucine |
|---|
| CAS Registry Number | 73-32-5 |
|---|
| SMILES | CC[C@H](C)[C@H](N)C(O)=O |
|---|
| InChI Identifier | InChI=1S/C6H13NO2/c1-3-4(2)5(7)6(8)9/h4-5H,3,7H2,1-2H3,(H,8,9)/t4-,5-/m0/s1 |
|---|
| InChI Key | AGPKZVBTJJNPAG-WHFBIAKZSA-N |
|---|
| Chemical Taxonomy |
|---|
| Description | Belongs to the class of organic compounds known as isoleucine and derivatives. Isoleucine and derivatives are compounds containing isoleucine or a derivative thereof resulting from reaction of isoleucine at the amino group or the carboxy group, or from the replacement of any hydrogen of glycine by a heteroatom. |
|---|
| Kingdom | Organic compounds |
|---|
| Super Class | Organic acids and derivatives |
|---|
| Class | Carboxylic acids and derivatives |
|---|
| Sub Class | Amino acids, peptides, and analogues |
|---|
| Direct Parent | Isoleucine and derivatives |
|---|
| Alternative Parents | |
|---|
| Substituents | - Isoleucine or derivatives
- Alpha-amino acid
- L-alpha-amino acid
- Branched fatty acid
- Methyl-branched fatty acid
- Fatty acid
- Fatty acyl
- Amino acid
- Monocarboxylic acid or derivatives
- Carboxylic acid
- Organic oxide
- Organopnictogen compound
- Primary amine
- Organooxygen compound
- Organonitrogen compound
- Primary aliphatic amine
- Carbonyl group
- Organic oxygen compound
- Amine
- Organic nitrogen compound
- Hydrocarbon derivative
- Aliphatic acyclic compound
|
|---|
| Molecular Framework | Aliphatic acyclic compounds |
|---|
| External Descriptors | |
|---|
| Ontology |
|---|
|
| Physiological effect | Health effect: |
|---|
| Disposition | Route of exposure: Source: Biological location: |
|---|
| Role | Industrial application: Biological role: |
|---|
| Physical Properties |
|---|
| State | Solid |
|---|
| Experimental Properties | | Property | Value | Reference |
|---|
| Melting Point | 285.5 °C | Not Available | | Boiling Point | Not Available | Not Available | | Water Solubility | 35 mg/mL | Human Metabolome Project | | logP | -1.70 | HANSCH,C ET AL. (1995) |
|
|---|
| Predicted Properties | [] |
|---|
| Spectra |
|---|
| EI-MS/GC-MS | | Type | Description | Splash Key | View |
|---|
| EI-MS | Mass Spectrum (Electron Ionization) | splash10-004r-9000000000-34d4d4cb7042da231eb4 | 2014-09-20 | View Spectrum | | GC-MS | L-Isoleucine, 2 TMS, GC-MS Spectrum | splash10-0a4i-0930000000-e78f845bb2a8d4736476 | Spectrum | | GC-MS | L-Isoleucine, 2 TMS, GC-MS Spectrum | splash10-0a4i-0910000000-de9162d149073d0e2a37 | Spectrum | | GC-MS | L-Isoleucine, 2 TMS, GC-MS Spectrum | splash10-0a4i-0920000000-599e61f8ccdb6525c7a9 | Spectrum | | GC-MS | L-Isoleucine, non-derivatized, GC-MS Spectrum | splash10-0a4i-0910000000-742e44c426c0d6c2a9ac | Spectrum | | GC-MS | L-Isoleucine, 2 TMS, GC-MS Spectrum | splash10-05fr-8910000000-dbb33e0f02ac2ca5fedd | Spectrum | | GC-MS | L-Isoleucine, non-derivatized, GC-MS Spectrum | splash10-0a4i-0920000000-37690f426455ac41e2c2 | Spectrum | | GC-MS | L-Isoleucine, non-derivatized, GC-MS Spectrum | splash10-0a4i-0930000000-e78f845bb2a8d4736476 | Spectrum | | GC-MS | L-Isoleucine, non-derivatized, GC-MS Spectrum | splash10-0a4i-0910000000-de9162d149073d0e2a37 | Spectrum | | GC-MS | L-Isoleucine, non-derivatized, GC-MS Spectrum | splash10-0a4i-0920000000-599e61f8ccdb6525c7a9 | Spectrum | | GC-MS | L-Isoleucine, non-derivatized, GC-MS Spectrum | splash10-0a4i-0910000000-742e44c426c0d6c2a9ac | Spectrum | | GC-MS | L-Isoleucine, non-derivatized, GC-MS Spectrum | splash10-0udi-2392000000-74cca1eb265b8e3d7f43 | Spectrum | | GC-MS | L-Isoleucine, non-derivatized, GC-MS Spectrum | splash10-05fr-8910000000-dbb33e0f02ac2ca5fedd | Spectrum | | GC-MS | L-Isoleucine, non-derivatized, GC-MS Spectrum | splash10-000i-9100000000-27891dff695c5db53796 | Spectrum | | GC-MS | L-Isoleucine, non-derivatized, GC-MS Spectrum | splash10-0a4i-0910000000-43ec1890f6dbd6aea657 | Spectrum | | Predicted GC-MS | L-Isoleucine, non-derivatized, Predicted GC-MS Spectrum - 70eV, Positive | splash10-00b9-9100000000-40bc9ef43da5f18be883 | Spectrum | | Predicted GC-MS | L-Isoleucine, 1 TMS, Predicted GC-MS Spectrum - 70eV, Positive | splash10-000i-9200000000-ae8694c0f3e4dcc608f9 | Spectrum | | Predicted GC-MS | L-Isoleucine, non-derivatized, Predicted GC-MS Spectrum - 70eV, Positive | Not Available | Spectrum | | Predicted GC-MS | L-Isoleucine, TMS_1_2, Predicted GC-MS Spectrum - 70eV, Positive | Not Available | Spectrum | | Predicted GC-MS | L-Isoleucine, TBDMS_1_1, Predicted GC-MS Spectrum - 70eV, Positive | Not Available | Spectrum | | Predicted GC-MS | L-Isoleucine, TBDMS_1_2, Predicted GC-MS Spectrum - 70eV, Positive | Not Available | Spectrum |
|
|---|
| MS/MS | | Type | Description | Splash Key | View |
|---|
| MS/MS | LC-MS/MS Spectrum - Quattro_QQQ 10V, Positive (Annotated) | splash10-000i-9300000000-8a23b3e62231eb65f80f | 2012-07-24 | View Spectrum | | MS/MS | LC-MS/MS Spectrum - Quattro_QQQ 25V, Positive (Annotated) | splash10-00ko-9000000000-ac7e71578d7e1d0e8ce9 | 2012-07-24 | View Spectrum | | MS/MS | LC-MS/MS Spectrum - Quattro_QQQ 40V, Positive (Annotated) | splash10-052f-9000000000-2586d4a089921dd977ed | 2012-07-24 | View Spectrum | | MS/MS | LC-MS/MS Spectrum - LC-ESI-ITFT (LTQ Orbitrap XL, Thermo Scientfic) , Positive | splash10-001i-0900000000-5b11521ff6a631376d2b | 2012-08-31 | View Spectrum | | MS/MS | LC-MS/MS Spectrum - LC-ESI-ITFT (LTQ Orbitrap XL, Thermo Scientfic) , Positive | splash10-000i-9000000000-3c352f229e4067fcd489 | 2012-08-31 | View Spectrum | | MS/MS | LC-MS/MS Spectrum - LC-ESI-ITFT (LTQ Orbitrap XL, Thermo Scientfic) , Positive | splash10-000i-9000000000-8a7ef48fc0c1b6f845c2 | 2012-08-31 | View Spectrum | | MS/MS | LC-MS/MS Spectrum - LC-ESI-ITFT (LTQ Orbitrap XL, Thermo Scientfic) , Positive | splash10-0002-0920000000-2e11aa1c7a5defc386db | 2012-08-31 | View Spectrum | | MS/MS | LC-MS/MS Spectrum - LC-ESI-ITFT (LTQ Orbitrap XL, Thermo Scientfic) , Positive | splash10-001i-0900000000-b63ac9cf06eda4c54a81 | 2012-08-31 | View Spectrum | | MS/MS | LC-MS/MS Spectrum - LC-ESI-ITFT (LTQ Orbitrap XL, Thermo Scientfic) , Positive | splash10-000i-9000000000-e5b0c8c6b09f541d6dbf | 2012-08-31 | View Spectrum | | MS/MS | LC-MS/MS Spectrum - LC-ESI-ITFT (LTQ Orbitrap XL, Thermo Scientfic) , Positive | splash10-000i-9000000000-d89d16d5c2242e44ec63 | 2012-08-31 | View Spectrum | | MS/MS | LC-MS/MS Spectrum - LC-ESI-ITFT (LTQ Orbitrap XL, Thermo Scientfic) , Positive | splash10-000i-9000000000-5de583142c7bdb381c8f | 2012-08-31 | View Spectrum | | MS/MS | LC-MS/MS Spectrum - LC-ESI-QQ (API3000, Applied Biosystems) 10V, Negative | splash10-001i-0900000000-8c75bde35c4a4073ee65 | 2012-08-31 | View Spectrum | | MS/MS | LC-MS/MS Spectrum - LC-ESI-QQ (API3000, Applied Biosystems) 20V, Negative | splash10-001i-0900000000-233a9862f616afea2d17 | 2012-08-31 | View Spectrum | | MS/MS | LC-MS/MS Spectrum - LC-ESI-QQ (API3000, Applied Biosystems) 30V, Negative | splash10-001i-1900000000-375f35065b82b13e6d6e | 2012-08-31 | View Spectrum | | MS/MS | LC-MS/MS Spectrum - LC-ESI-QQ (API3000, Applied Biosystems) 40V, Negative | splash10-00dl-9000000000-d0d4a9f90fe2aca74483 | 2012-08-31 | View Spectrum | | MS/MS | LC-MS/MS Spectrum - LC-ESI-QQ (API3000, Applied Biosystems) 50V, Negative | splash10-0006-9000000000-0018f47571feaf232ff8 | 2012-08-31 | View Spectrum | | MS/MS | LC-MS/MS Spectrum - LC-ESI-QQ (API3000, Applied Biosystems) 10V, Positive | splash10-001r-7900000000-278dc67396114be331ba | 2012-08-31 | View Spectrum | | MS/MS | LC-MS/MS Spectrum - LC-ESI-QQ (API3000, Applied Biosystems) 20V, Positive | splash10-000i-9000000000-e26c042aa6231eeca071 | 2012-08-31 | View Spectrum | | MS/MS | LC-MS/MS Spectrum - LC-ESI-QQ (API3000, Applied Biosystems) 30V, Positive | splash10-014r-9000000000-b6c1752fd3fbccb3d1c5 | 2012-08-31 | View Spectrum | | MS/MS | LC-MS/MS Spectrum - LC-ESI-QQ (API3000, Applied Biosystems) 40V, Positive | splash10-05mo-9000000000-0569c3162621252ed0a8 | 2012-08-31 | View Spectrum | | MS/MS | LC-MS/MS Spectrum - LC-ESI-QQ (API3000, Applied Biosystems) 50V, Positive | splash10-052f-9000000000-3d52b5d56d3fab45276e | 2012-08-31 | View Spectrum | | MS/MS | LC-MS/MS Spectrum - CE-ESI-TOF (CE-system connected to 6210 Time-of-Flight MS, Agilent) , Positive | splash10-001i-0900000000-720554d58264a9cfdb67 | 2012-08-31 | View Spectrum | | Predicted MS/MS | Predicted LC-MS/MS Spectrum - 10V, Positive | splash10-0019-9500000000-00268b2694cde281641b | 2016-09-12 | View Spectrum | | Predicted MS/MS | Predicted LC-MS/MS Spectrum - 20V, Positive | splash10-000i-9000000000-4014793e17e0290790a4 | 2016-09-12 | View Spectrum | | Predicted MS/MS | Predicted LC-MS/MS Spectrum - 40V, Positive | splash10-0a4i-9000000000-eb9a97a14461e524e76e | 2016-09-12 | View Spectrum |
|
|---|
| NMR | | Type | Description | | View |
|---|
| 1D NMR | 1H NMR Spectrum (1D, 600 MHz, H2O, experimental) | | Spectrum | | 1D NMR | 13C NMR Spectrum (1D, 125 MHz, H2O, experimental) | | Spectrum | | 1D NMR | 13C NMR Spectrum (1D, 22.5 MHz, D2O, experimental) | | Spectrum | | 1D NMR | 1H NMR Spectrum (1D, 100 MHz, D2O, predicted) | | Spectrum | | 1D NMR | 13C NMR Spectrum (1D, 100 MHz, D2O, predicted) | | Spectrum | | 1D NMR | 1H NMR Spectrum (1D, 200 MHz, D2O, predicted) | | Spectrum | | 1D NMR | 13C NMR Spectrum (1D, 200 MHz, D2O, predicted) | | Spectrum | | 1D NMR | 1H NMR Spectrum (1D, 300 MHz, D2O, predicted) | | Spectrum | | 1D NMR | 13C NMR Spectrum (1D, 300 MHz, D2O, predicted) | | Spectrum | | 1D NMR | 1H NMR Spectrum (1D, 400 MHz, D2O, predicted) | | Spectrum | | 1D NMR | 13C NMR Spectrum (1D, 400 MHz, D2O, predicted) | | Spectrum | | 1D NMR | 1H NMR Spectrum (1D, 500 MHz, D2O, predicted) | | Spectrum | | 1D NMR | 13C NMR Spectrum (1D, 500 MHz, D2O, predicted) | | Spectrum | | 1D NMR | 1H NMR Spectrum (1D, 600 MHz, D2O, predicted) | | Spectrum | | 1D NMR | 13C NMR Spectrum (1D, 600 MHz, D2O, predicted) | | Spectrum | | 1D NMR | 1H NMR Spectrum (1D, 700 MHz, D2O, predicted) | | Spectrum | | 1D NMR | 13C NMR Spectrum (1D, 700 MHz, D2O, predicted) | | Spectrum | | 1D NMR | 1H NMR Spectrum (1D, 800 MHz, D2O, predicted) | | Spectrum | | 1D NMR | 13C NMR Spectrum (1D, 800 MHz, D2O, predicted) | | Spectrum | | 1D NMR | 1H NMR Spectrum (1D, 900 MHz, D2O, predicted) | | Spectrum | | 1D NMR | 13C NMR Spectrum (1D, 900 MHz, D2O, predicted) | | Spectrum | | 1D NMR | 1H NMR Spectrum (1D, 1000 MHz, D2O, predicted) | | Spectrum | | 1D NMR | 13C NMR Spectrum (1D, 1000 MHz, D2O, predicted) | | Spectrum | | 1D NMR | 13C NMR Spectrum (1D, 400 MHz, H2O, experimental) | | Spectrum | | 2D NMR | [1H, 1H]-TOCSY. Unexported temporarily by An Chi on Oct 15, 2021 until json or nmrML file is generated. 2D NMR Spectrum (experimental) | | Spectrum | | 2D NMR | [1H, 13C]-HSQC NMR Spectrum (2D, 600 MHz, H2O, experimental) | | Spectrum |
|
|---|
| Pathways |
|---|
| Pathways | | Name | SMPDB/Pathwhiz | KEGG | | Valine, Leucine and Isoleucine Degradation |   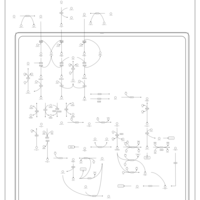 |  | | Transcription/Translation | Not Available | Not Available | | Beta-Ketothiolase Deficiency |  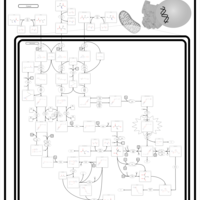  | Not Available | | 2-Methyl-3-Hydroxybutryl CoA Dehydrogenase Deficiency |    | Not Available | | Propionic Acidemia | 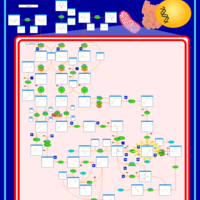 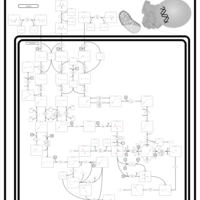 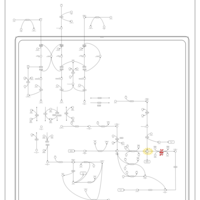 | Not Available |
|
|---|
| Protein Targets |
|---|
| Enzymes | |
| Branched-chain-amino-acid aminotransferase, cytosolic | BCAT1 | 12p12.1 | P54687 | details | | Branched-chain-amino-acid aminotransferase, mitochondrial | BCAT2 | 19q13 | O15382 | details | | Isoleucine--tRNA ligase, cytoplasmic | IARS | 9q21 | P41252 | details | | Short/branched chain specific acyl-CoA dehydrogenase, mitochondrial | ACADSB | 10q26.13 | P45954 | details | | L-amino-acid oxidase | IL4I1 | 19q13.3-q13.4 | Q96RQ9 | details | | Isoleucine--tRNA ligase, mitochondrial | IARS2 | 1q41 | Q9NSE4 | details |
|
|---|
| Transporters | |
|---|
| Metal Bindings | Not Available |
|---|
| Receptors | Not Available |
|---|
| Transcriptional Factors | Not Available |
|---|
| Concentrations Data |
|---|
| |
| Alien Dawg | Detected and Quantified | 0.416 mg/g dry wt | | details | | Gabriola | Detected and Quantified | 0.191 mg/g dry wt | | details | | Island Honey | Detected and Quantified | 0.429 mg/g dry wt | | details | | Quadra | Detected and Quantified | 0.331 mg/g dry wt | | details | | Sensi Star | Detected and Quantified | 0.0613 mg/g dry wt | | details | | Tangerine Dream | Detected and Quantified | 0.120 mg/g dry wt | | details |
|
|---|
| External Links |
|---|
| HMDB ID | HMDB0000172 |
|---|
| DrugBank ID | DB00167 |
|---|
| Phenol Explorer Compound ID | Not Available |
|---|
| FoodDB ID | FDB012397 |
|---|
| KNApSAcK ID | C00001374 |
|---|
| Chemspider ID | 6067 |
|---|
| KEGG Compound ID | C00407 |
|---|
| BioCyc ID | ILE |
|---|
| BiGG ID | 34887 |
|---|
| Wikipedia Link | Isoleucine |
|---|
| METLIN ID | 5193 |
|---|
| PubChem Compound | 6306 |
|---|
| PDB ID | Not Available |
|---|
| ChEBI ID | 17191 |
|---|
| References |
|---|
| General References | - Turner CE, Elsohly MA, Boeren EG: Constituents of Cannabis sativa L. XVII. A review of the natural constituents. J Nat Prod. 1980 Mar-Apr;43(2):169-234. doi: 10.1021/np50008a001. [PubMed:6991645 ]
- Podebrad F, Heil M, Reichert S, Mosandl A, Sewell AC, Bohles H: 4,5-dimethyl-3-hydroxy-2[5H]-furanone (sotolone)--the odour of maple syrup urine disease. J Inherit Metab Dis. 1999 Apr;22(2):107-14. doi: 10.1023/a:1005433516026. [PubMed:10234605 ]
|
|---|
