| Record Information |
|---|
| Version | 1.0 |
|---|
| Created at | 2020-03-18 23:25:25 UTC |
|---|
| Updated at | 2022-12-13 23:36:24 UTC |
|---|
| CannabisDB ID | CDB000117 |
|---|
| Secondary Accession Numbers | Not Available |
|---|
| Cannabis Compound Identification |
|---|
| Common Name | Arginine |
|---|
| Description | Arginine is an essential amino acid that is physiologically active in the L-form. In mammals, arginine is formally classified as a semi-essential or conditionally essential amino acid, depending on the developmental stage and health status of the individual. Infants are unable to effectively synthesize arginine, making it nutritionally essential for infants. Adults, however, are able to synthesize arginine in the urea cycle. Arginine can be considered to be a basic amino acid as the part of the side chain nearest to the backbone is long, carbon-containing, and hydrophobic, whereas the end of the side chain is a complex guanidinium group. With a pKa of 12.48, the guanidinium group is positively charged in neutral, acidic, and even most basic environments. Because of the conjugation between the double bond and the nitrogen lone pairs, the positive charge is delocalized. This group is able to form multiple H-bonds. L-Arginine is an amino acid that has numerous functions in the body. It helps dispose of ammonia, is used to make compounds such as nitric oxide, creatine, L-glutamate, and L-proline, and it can be converted into glucose and glycogen if needed. In large doses, L-arginine also stimulates the release of the hormones growth hormone and prolactin. Arginine is a known inducer of mTOR (mammalian target of rapamycin) and is responsible for inducing protein synthesis through the mTOR pathway. mTOR inhibition by rapamycin partially reduces arginine-induced protein synthesis (PMID: 20841502  ). Catabolic disease states such as sepsis, injury, and cancer cause an increase in arginine utilization, which can exceed normal body production, leading to arginine depletion. Arginine also activates AMP kinase (AMPK) which then stimulates skeletal muscle fatty acid oxidation and muscle glucose uptake, thereby increasing insulin secretion by pancreatic beta-cells (PMID: 21311355  ). Arginine is found in plant and animal proteins, such as dairy products, meat, poultry, fish, and nuts. The ratio of L-arginine to lysine is also important: soy and other plant proteins have more L-arginine than animal sources of protein. Arginine is one of the amino acids that are known in cannabis plant (PMID: 6991645 ). |
|---|
| Structure | |
|---|
| Synonyms | | Value | Source |
|---|
| (2R)-2-Amino-5-(carbamimidamido)pentanoic acid | ChEBI | | (2R)-2-Amino-5-guanidinopentanoic acid | ChEBI | | (R)-2-Amino-5-guanidinopentanoic acid | ChEBI | | D-2-Amino-5-guanidinovaleric acid | ChEBI | | D-Arginin | ChEBI | | DAR | ChEBI | | (2R)-2-Amino-5-(carbamimidamido)pentanoate | Generator | | (2R)-2-Amino-5-guanidinopentanoate | Generator | | (R)-2-Amino-5-guanidinopentanoate | Generator | | D-2-Amino-5-guanidinovalerate | Generator |
|
|---|
| Chemical Formula | C6H14N4O2 |
|---|
| Average Molecular Weight | 174.2 |
|---|
| Monoisotopic Molecular Weight | 174.1117 |
|---|
| IUPAC Name | (2R)-2-amino-5-carbamimidamidopentanoic acid |
|---|
| Traditional Name | D-arginine |
|---|
| CAS Registry Number | 157-06-2 |
|---|
| SMILES | N[C@H](CCCNC(N)=N)C(O)=O |
|---|
| InChI Identifier | InChI=1S/C6H14N4O2/c7-4(5(11)12)2-1-3-10-6(8)9/h4H,1-3,7H2,(H,11,12)(H4,8,9,10)/t4-/m1/s1 |
|---|
| InChI Key | ODKSFYDXXFIFQN-SCSAIBSYSA-N |
|---|
| Chemical Taxonomy |
|---|
| Description | Belongs to the class of organic compounds known as d-alpha-amino acids. These are alpha amino acids which have the D-configuration of the alpha-carbon atom. |
|---|
| Kingdom | Organic compounds |
|---|
| Super Class | Organic acids and derivatives |
|---|
| Class | Carboxylic acids and derivatives |
|---|
| Sub Class | Amino acids, peptides, and analogues |
|---|
| Direct Parent | D-alpha-amino acids |
|---|
| Alternative Parents | |
|---|
| Substituents | - D-alpha-amino acid
- Fatty acid
- Guanidine
- Amino acid
- Carboxylic acid
- Monocarboxylic acid or derivatives
- Carboximidamide
- Propargyl-type 1,3-dipolar organic compound
- Organic 1,3-dipolar compound
- Amine
- Hydrocarbon derivative
- Organic oxide
- Primary amine
- Organooxygen compound
- Organonitrogen compound
- Organopnictogen compound
- Primary aliphatic amine
- Organic oxygen compound
- Organic nitrogen compound
- Carbonyl group
- Aliphatic acyclic compound
|
|---|
| Molecular Framework | Aliphatic acyclic compounds |
|---|
| External Descriptors | |
|---|
| Ontology |
|---|
|
| Disposition | Source: Biological location: |
|---|
| Role | Industrial application: |
|---|
| Physical Properties |
|---|
| State | Solid |
|---|
| Experimental Properties | | Property | Value | Reference |
|---|
| Melting Point | 260 °C | Wikipedia | | Boiling Point | 368 °C | Wikipedia | | Water Solubility | 14.87 g/100 mL at 20 °C | Wikipedia | | logP | 0.0 | Wikipedia |
|
|---|
| Predicted Properties | [] |
|---|
| Spectra |
|---|
| EI-MS/GC-MS | | Type | Description | Splash Key | View |
|---|
| Predicted GC-MS | Arginine, non-derivatized, Predicted GC-MS Spectrum - 70eV, Positive | splash10-00dl-9100000000-60db59de76b9a9b5bfc8 | Spectrum | | Predicted GC-MS | Arginine, 1 TMS, Predicted GC-MS Spectrum - 70eV, Positive | splash10-00dl-9510000000-a5f3be057956f5efb0fd | Spectrum | | Predicted GC-MS | Arginine, non-derivatized, Predicted GC-MS Spectrum - 70eV, Positive | Not Available | Spectrum |
|
|---|
| MS/MS | | Type | Description | Splash Key | View |
|---|
| MS/MS | LC-MS/MS Spectrum - Quattro_QQQ 10V, Positive (Annotated) | splash10-00di-1900000000-a1207a353a5d383e8118 | 2012-07-25 | View Spectrum | | MS/MS | LC-MS/MS Spectrum - Quattro_QQQ 25V, Positive (Annotated) | splash10-00di-9000000000-38faac95afc81e981a08 | 2012-07-25 | View Spectrum | | MS/MS | LC-MS/MS Spectrum - Quattro_QQQ 40V, Positive (Annotated) | splash10-00di-9000000000-776b9e0f6bf6a0ddf5ba | 2012-07-25 | View Spectrum | | Predicted MS/MS | Predicted LC-MS/MS Spectrum - 10V, Positive | splash10-004i-1900000000-0627028bdd33e89ac545 | 2017-07-26 | View Spectrum | | Predicted MS/MS | Predicted LC-MS/MS Spectrum - 20V, Positive | splash10-03mi-8900000000-d00d08306af5359ec3dc | 2017-07-26 | View Spectrum | | Predicted MS/MS | Predicted LC-MS/MS Spectrum - 40V, Positive | splash10-03di-9000000000-09fdbc63550a3fe95638 | 2017-07-26 | View Spectrum | | Predicted MS/MS | Predicted LC-MS/MS Spectrum - 10V, Negative | splash10-00e9-2900000000-4f23a56d70e700301134 | 2017-07-26 | View Spectrum | | Predicted MS/MS | Predicted LC-MS/MS Spectrum - 20V, Negative | splash10-053r-7900000000-02e7df28226010832ffe | 2017-07-26 | View Spectrum | | Predicted MS/MS | Predicted LC-MS/MS Spectrum - 40V, Negative | splash10-0006-9000000000-435f29993f2aaceb0d44 | 2017-07-26 | View Spectrum | | Predicted MS/MS | Predicted LC-MS/MS Spectrum - 10V, Positive | splash10-056r-0900000000-fe2cccae6934dad0053b | 2021-09-22 | View Spectrum | | Predicted MS/MS | Predicted LC-MS/MS Spectrum - 20V, Positive | splash10-00di-9200000000-da3c09ec6a806e5c0777 | 2021-09-22 | View Spectrum | | Predicted MS/MS | Predicted LC-MS/MS Spectrum - 40V, Positive | splash10-00di-9000000000-15e9d13a1d44b86b2a6c | 2021-09-22 | View Spectrum | | Predicted MS/MS | Predicted LC-MS/MS Spectrum - 10V, Negative | splash10-00di-0900000000-a706f1985fb3e9e0adbb | 2021-09-22 | View Spectrum | | Predicted MS/MS | Predicted LC-MS/MS Spectrum - 20V, Negative | splash10-001i-2900000000-b42c24606ae24000888d | 2021-09-22 | View Spectrum | | Predicted MS/MS | Predicted LC-MS/MS Spectrum - 40V, Negative | splash10-0006-9000000000-1c8520c5f7efb1187e43 | 2021-09-22 | View Spectrum |
|
|---|
| NMR | | Type | Description | | View |
|---|
| 1D NMR | 1H NMR Spectrum (1D, 600 MHz, H2O, experimental) | | Spectrum | | 1D NMR | 13C NMR Spectrum (1D, 100 MHz, D2O, predicted) | | Spectrum | | 1D NMR | 1H NMR Spectrum (1D, 100 MHz, D2O, predicted) | | Spectrum | | 1D NMR | 13C NMR Spectrum (1D, 1000 MHz, D2O, predicted) | | Spectrum | | 1D NMR | 1H NMR Spectrum (1D, 1000 MHz, D2O, predicted) | | Spectrum | | 1D NMR | 13C NMR Spectrum (1D, 200 MHz, D2O, predicted) | | Spectrum | | 1D NMR | 1H NMR Spectrum (1D, 200 MHz, D2O, predicted) | | Spectrum | | 1D NMR | 13C NMR Spectrum (1D, 300 MHz, D2O, predicted) | | Spectrum | | 1D NMR | 1H NMR Spectrum (1D, 300 MHz, D2O, predicted) | | Spectrum | | 1D NMR | 13C NMR Spectrum (1D, 400 MHz, D2O, predicted) | | Spectrum | | 1D NMR | 1H NMR Spectrum (1D, 400 MHz, D2O, predicted) | | Spectrum | | 1D NMR | 13C NMR Spectrum (1D, 500 MHz, D2O, predicted) | | Spectrum | | 1D NMR | 1H NMR Spectrum (1D, 500 MHz, D2O, predicted) | | Spectrum | | 1D NMR | 13C NMR Spectrum (1D, 600 MHz, D2O, predicted) | | Spectrum | | 1D NMR | 1H NMR Spectrum (1D, 600 MHz, D2O, predicted) | | Spectrum | | 1D NMR | 13C NMR Spectrum (1D, 700 MHz, D2O, predicted) | | Spectrum | | 1D NMR | 1H NMR Spectrum (1D, 700 MHz, D2O, predicted) | | Spectrum | | 1D NMR | 13C NMR Spectrum (1D, 800 MHz, D2O, predicted) | | Spectrum | | 1D NMR | 1H NMR Spectrum (1D, 800 MHz, D2O, predicted) | | Spectrum | | 1D NMR | 13C NMR Spectrum (1D, 900 MHz, D2O, predicted) | | Spectrum | | 1D NMR | 1H NMR Spectrum (1D, 900 MHz, D2O, predicted) | | Spectrum | | 2D NMR | [1H, 13C]-HSQC NMR Spectrum (2D, 600 MHz, H2O, experimental) | | Spectrum |
|
|---|
| Pathways |
|---|
| Pathways | | Name | SMPDB/Pathwhiz | KEGG | | D-Arginine and D-Ornithine Metabolism |    | 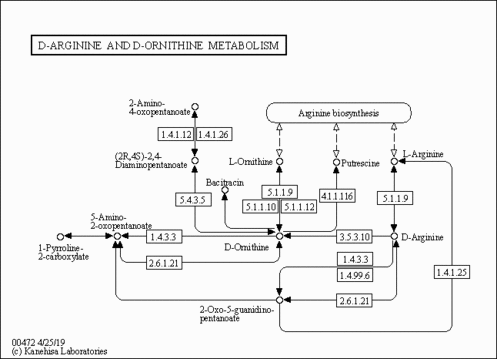 |
|
|---|
| Protein Targets |
|---|
| Enzymes | |
|---|
| Transporters | Not Available |
|---|
| Metal Bindings | Not Available |
|---|
| Receptors | Not Available |
|---|
| Transcriptional Factors | Not Available |
|---|
| Concentrations Data |
|---|
| |
| Alien Dawg | Detected and Quantified | 7.909 mg/g dry wt | | details | | Gabriola | Detected and Quantified | 4.771 mg/g dry wt | | details | | Island Honey | Detected and Quantified | 6.114 mg/g dry wt | | details | | Quadra | Detected and Quantified | 4.578 mg/g dry wt | | details | | Sensi Star | Detected and Quantified | 3.474 mg/g dry wt | | details | | Tangerine Dream | Detected and Quantified | 4.651 mg/g dry wt | | details |
|
|---|
| External Links |
|---|
| HMDB ID | HMDB0003416 |
|---|
| DrugBank ID | DB04027 |
|---|
| Phenol Explorer Compound ID | Not Available |
|---|
| FoodDB ID | FDB023167 |
|---|
| KNApSAcK ID | Not Available |
|---|
| Chemspider ID | 64224 |
|---|
| KEGG Compound ID | C00792 |
|---|
| BioCyc ID | CPD-220 |
|---|
| BiGG ID | 36012 |
|---|
| Wikipedia Link | Arginine |
|---|
| METLIN ID | 6924 |
|---|
| PubChem Compound | 71070 |
|---|
| PDB ID | Not Available |
|---|
| ChEBI ID | 15816 |
|---|
| References |
|---|
| General References | - Turner CE, Elsohly MA, Boeren EG: Constituents of Cannabis sativa L. XVII. A review of the natural constituents. J Nat Prod. 1980 Mar-Apr;43(2):169-234. doi: 10.1021/np50008a001. [PubMed:6991645 ]
- Bauchart-Thevret C, Cui L, Wu G, Burrin DG: Arginine-induced stimulation of protein synthesis and survival in IPEC-J2 cells is mediated by mTOR but not nitric oxide. Am J Physiol Endocrinol Metab. 2010 Dec;299(6):E899-909. doi: 10.1152/ajpendo.00068.2010. Epub 2010 Sep 14. [PubMed:20841502 ]
- Linden KC, Wadley GD, Garnham AP, McConell GK: Effect of l-arginine infusion on glucose disposal during exercise in humans. Med Sci Sports Exerc. 2011 Sep;43(9):1626-34. doi: 10.1249/MSS.0b013e318212a317. [PubMed:21311355 ]
|
|---|
