| Record Information |
|---|
| Version | 1.0 |
|---|
| Created at | 2020-03-18 23:22:13 UTC |
|---|
| Updated at | 2022-12-13 23:36:27 UTC |
|---|
| CannabisDB ID | CDB006164 |
|---|
| Secondary Accession Numbers | Not Available |
|---|
| Cannabis Compound Identification |
|---|
| Common Name | Glyceric acid |
|---|
| Description | Glyceric acid is a colourless syrupy acid, obtained via the oxidation of glycerol. Glyceric acid is a weak 3-carbon sugar acid. Several phosphate derivatives of glyceric acid, including 2-phosphoglyceric acid, 3-phosphoglyceric acid, 2,3-bisphosphoglyceric acid, and 1,3-bisphosphoglyceric acid, are important biochemical intermediates in glycolysis. In particular, 3-phosphoglyceric acid is an important molecule for the biosynthesis of the amino acid serine, which in turn can be used in the synthesis of glycine and cysteine. Glyceric acid is not normally found in human biofluids, however high levels are secreted in the urine by patients suffering from D-glyceric aciduria, an inborn error of metabolism. It is also elevated in the serum of individuals suffering from D-glycerate anemia. A deficiency of human glycerate kinase has been shown to lead to D-glycerate acidemia/D-glyceric aciduria ( PMID: 20949620 ). Symptoms of the disease include progressive neurological impairment, hypotonia, seizures, failure to thrive, and metabolic acidosis. At sufficiently high levels, glyceric acid can act as an acidogen and a metabotoxin. An acidogen is an acidic compound that induces acidosis, which has multiple adverse effects on many organ systems. A metabotoxin is an endogenously produced metabolite that causes adverse health effects at chronically high levels. Abnormally high levels of organic acids (glyceric acid is an organic acid) in the blood (organic acidemia), urine (organic aciduria), the brain, and other tissues lead to general metabolic acidosis. Acidosis typically occurs when arterial pH falls below 7.35. In infants with acidosis, the initial symptoms include poor feeding, vomiting, loss of appetite, weak muscle tone (hypotonia), and lack of energy (lethargy). These can progress to heart abnormalities, seizures, coma, and possibly death. These are also the characteristic symptoms of untreated glyceric aciduria. Many affected children with organic acidemias experience intellectual disability or delayed development. In adults, acidosis or acidemia is characterized by headaches, confusion, feeling tired, tremors, sleepiness, and seizures. Elevated values may also be due to microbial sources such as yeast (Aspergillus, Penicillium, probably Candida) or due to dietary sources containing glycerol (glycerine). Glyceric acid can be isolated from various crop plants (e.g. brassicas and pulses) and has also been found in peanuts, artichokes, tomatoes, apples, bananas, broad beans and grapes (PMID: 19621222 ). |
|---|
| Structure | |
|---|
| Synonyms | | Value | Source |
|---|
| alpha,beta-Hydroxypropionic acid | ChEBI | | D-GroA | ChEBI | | R-Glyceric acid | ChEBI | | Glycerate | Kegg | | (R)-Glycerate | Kegg | | a,b-Hydroxypropionate | Generator | | a,b-Hydroxypropionic acid | Generator | | alpha,beta-Hydroxypropionate | Generator | | R-Glycerate | Generator | | (R)-Glyceric acid | Generator | | D-Glycerate | HMDB | | D-Glyceric acid | HMDB | | (2R)-2,3-Dihydroxypropanoic acid | HMDB | | (R)-2,3-Dihydroxypropanoic acid | HMDB | | D-2,3-Dihydroxypropanoic acid | HMDB |
|
|---|
| Chemical Formula | C3H6O4 |
|---|
| Average Molecular Weight | 106.08 |
|---|
| Monoisotopic Molecular Weight | 106.0266 |
|---|
| IUPAC Name | (2R)-2,3-dihydroxypropanoic acid |
|---|
| Traditional Name | glycerate |
|---|
| CAS Registry Number | 6000-40-4 |
|---|
| SMILES | OC[C@@H](O)C(O)=O |
|---|
| InChI Identifier | InChI=1S/C3H6O4/c4-1-2(5)3(6)7/h2,4-5H,1H2,(H,6,7)/t2-/m1/s1 |
|---|
| InChI Key | RBNPOMFGQQGHHO-UWTATZPHSA-N |
|---|
| Chemical Taxonomy |
|---|
| Description | Belongs to the class of organic compounds known as sugar acids and derivatives. Sugar acids and derivatives are compounds containing a saccharide unit which bears a carboxylic acid group. |
|---|
| Kingdom | Organic compounds |
|---|
| Super Class | Organic oxygen compounds |
|---|
| Class | Organooxygen compounds |
|---|
| Sub Class | Carbohydrates and carbohydrate conjugates |
|---|
| Direct Parent | Sugar acids and derivatives |
|---|
| Alternative Parents | |
|---|
| Substituents | - Beta-hydroxy acid
- Glyceric_acid
- Sugar acid
- Alpha-hydroxy acid
- Hydroxy acid
- Monosaccharide
- 1,2-diol
- Secondary alcohol
- Monocarboxylic acid or derivatives
- Carboxylic acid
- Carboxylic acid derivative
- Alcohol
- Carbonyl group
- Organic oxide
- Primary alcohol
- Hydrocarbon derivative
- Aliphatic acyclic compound
|
|---|
| Molecular Framework | Aliphatic acyclic compounds |
|---|
| External Descriptors | |
|---|
| Ontology |
|---|
|
| Physiological effect | Health effect: |
|---|
| Disposition | Route of exposure: Source: Biological location: |
|---|
| Role | Indirect biological role: |
|---|
| Physical Properties |
|---|
| State | Solid |
|---|
| Experimental Properties | | Property | Value | Reference |
|---|
| Melting Point | <25 °C | Wikipedia | | Boiling Point | 412 °C | Wikipedia | | Water Solubility | 1000 mg/mL | Not Available | | logP | -1.723 | Wikipedia |
|
|---|
| Predicted Properties | [] |
|---|
| Spectra |
|---|
| EI-MS/GC-MS | | Type | Description | Splash Key | View |
|---|
| GC-MS | Glyceric acid, non-derivatized, GC-MS Spectrum | splash10-0f6t-0910000000-52b917dbc684454452ae | Spectrum | | GC-MS | Glyceric acid, 3 TMS, GC-MS Spectrum | splash10-0uei-0920000000-4d5f18f14464b7289e4e | Spectrum | | GC-MS | Glyceric acid, non-derivatized, GC-MS Spectrum | splash10-0f72-0920000000-7727c90167e5f1e15f7c | Spectrum | | GC-MS | Glyceric acid, non-derivatized, GC-MS Spectrum | splash10-0f6t-0910000000-52b917dbc684454452ae | Spectrum | | GC-MS | Glyceric acid, non-derivatized, GC-MS Spectrum | splash10-0uei-0920000000-4d5f18f14464b7289e4e | Spectrum | | Predicted GC-MS | Glyceric acid, non-derivatized, Predicted GC-MS Spectrum - 70eV, Positive | splash10-01po-9000000000-e74d8dcadd449f32f28f | Spectrum | | Predicted GC-MS | Glyceric acid, 3 TMS, Predicted GC-MS Spectrum - 70eV, Positive | splash10-0avr-9362000000-4ae43a853db514e46cc7 | Spectrum | | Predicted GC-MS | Glyceric acid, non-derivatized, Predicted GC-MS Spectrum - 70eV, Positive | Not Available | Spectrum | | Predicted GC-MS | Glyceric acid, non-derivatized, Predicted GC-MS Spectrum - 70eV, Positive | Not Available | Spectrum | | Predicted GC-MS | Glyceric acid, TMS_1_1, Predicted GC-MS Spectrum - 70eV, Positive | Not Available | Spectrum | | Predicted GC-MS | Glyceric acid, TMS_1_2, Predicted GC-MS Spectrum - 70eV, Positive | Not Available | Spectrum | | Predicted GC-MS | Glyceric acid, TMS_1_3, Predicted GC-MS Spectrum - 70eV, Positive | Not Available | Spectrum | | Predicted GC-MS | Glyceric acid, TMS_2_1, Predicted GC-MS Spectrum - 70eV, Positive | Not Available | Spectrum | | Predicted GC-MS | Glyceric acid, TMS_2_2, Predicted GC-MS Spectrum - 70eV, Positive | Not Available | Spectrum | | Predicted GC-MS | Glyceric acid, TMS_2_3, Predicted GC-MS Spectrum - 70eV, Positive | Not Available | Spectrum | | Predicted GC-MS | Glyceric acid, TBDMS_1_1, Predicted GC-MS Spectrum - 70eV, Positive | Not Available | Spectrum | | Predicted GC-MS | Glyceric acid, TBDMS_1_2, Predicted GC-MS Spectrum - 70eV, Positive | Not Available | Spectrum | | Predicted GC-MS | Glyceric acid, TBDMS_1_3, Predicted GC-MS Spectrum - 70eV, Positive | Not Available | Spectrum | | Predicted GC-MS | Glyceric acid, TBDMS_2_1, Predicted GC-MS Spectrum - 70eV, Positive | Not Available | Spectrum | | Predicted GC-MS | Glyceric acid, TBDMS_2_2, Predicted GC-MS Spectrum - 70eV, Positive | Not Available | Spectrum | | Predicted GC-MS | Glyceric acid, TBDMS_2_3, Predicted GC-MS Spectrum - 70eV, Positive | Not Available | Spectrum | | Predicted GC-MS | Glyceric acid, TBDMS_3_1, Predicted GC-MS Spectrum - 70eV, Positive | Not Available | Spectrum |
|
|---|
| MS/MS | | Type | Description | Splash Key | View |
|---|
| MS/MS | LC-MS/MS Spectrum - Quattro_QQQ 10V, Negative (Annotated) | splash10-0ufr-9800000000-f68cee7fd839cc0c350a | 2012-07-24 | View Spectrum | | MS/MS | LC-MS/MS Spectrum - Quattro_QQQ 25V, Negative (Annotated) | splash10-0pb9-9300000000-c9741b095b361aa0bf2e | 2012-07-24 | View Spectrum | | MS/MS | LC-MS/MS Spectrum - Quattro_QQQ 40V, Negative (Annotated) | splash10-0udi-8900000000-2ca9bd370ef2f7afdf64 | 2012-07-24 | View Spectrum | | MS/MS | LC-MS/MS Spectrum - LC-ESI-QQ (API3000, Applied Biosystems) 10V, Negative | splash10-0udi-0900000000-2c68fcd2a4c9cc998e14 | 2012-08-31 | View Spectrum | | MS/MS | LC-MS/MS Spectrum - LC-ESI-QQ (API3000, Applied Biosystems) 20V, Negative | splash10-0a4i-9400000000-a3e0c92f667ab9fdfbe5 | 2012-08-31 | View Spectrum | | MS/MS | LC-MS/MS Spectrum - LC-ESI-QQ (API3000, Applied Biosystems) 30V, Negative | splash10-0a4i-9000000000-e2f8962c5a4a4bde459a | 2012-08-31 | View Spectrum | | MS/MS | LC-MS/MS Spectrum - LC-ESI-QQ (API3000, Applied Biosystems) 40V, Negative | splash10-052e-9000000000-08ad44e661510838b17f | 2012-08-31 | View Spectrum | | MS/MS | LC-MS/MS Spectrum - LC-ESI-QQ (API3000, Applied Biosystems) 50V, Negative | splash10-0007-9000000000-b65c29cedef64ad74dfb | 2012-08-31 | View Spectrum | | MS/MS | LC-MS/MS Spectrum - LC-ESI-QTOF (UPLC Q-Tof Premier, Waters) , Negative | splash10-0a4i-1900000000-2d91902f3a95d7e66bba | 2012-08-31 | View Spectrum | | MS/MS | LC-MS/MS Spectrum - LC-ESI-QQ , negative | splash10-0udi-0900000000-2c68fcd2a4c9cc998e14 | 2017-09-14 | View Spectrum | | MS/MS | LC-MS/MS Spectrum - LC-ESI-QQ , negative | splash10-0a4i-9400000000-a3e0c92f667ab9fdfbe5 | 2017-09-14 | View Spectrum | | MS/MS | LC-MS/MS Spectrum - LC-ESI-QQ , negative | splash10-0a4i-9000000000-e2f8962c5a4a4bde459a | 2017-09-14 | View Spectrum | | MS/MS | LC-MS/MS Spectrum - LC-ESI-QQ , negative | splash10-052e-9000000000-08ad44e661510838b17f | 2017-09-14 | View Spectrum | | MS/MS | LC-MS/MS Spectrum - LC-ESI-QQ , negative | splash10-0007-9000000000-b65c29cedef64ad74dfb | 2017-09-14 | View Spectrum | | MS/MS | LC-MS/MS Spectrum - LC-ESI-QTOF , negative | splash10-0a4i-1900000000-2d91902f3a95d7e66bba | 2017-09-14 | View Spectrum | | MS/MS | LC-MS/MS Spectrum - 20V, Negative | splash10-0a4i-9000000000-b4845ca0075939f3095f | 2021-09-20 | View Spectrum | | MS/MS | LC-MS/MS Spectrum - 20V, Negative | splash10-052f-9000000000-f8ad6e1bbc7c43ea1122 | 2021-09-20 | View Spectrum | | MS/MS | LC-MS/MS Spectrum - 10V, Negative | splash10-0a4i-9400000000-9d9a074f07f367d0a0cc | 2021-09-20 | View Spectrum | | MS/MS | LC-MS/MS Spectrum - 10V, Negative | splash10-0a4i-9300000000-0e161d242fe9a38ccf1a | 2021-09-20 | View Spectrum | | Predicted MS/MS | Predicted LC-MS/MS Spectrum - 10V, Positive | splash10-0a4r-9600000000-6c887f0d5bb5b5b43e47 | 2016-09-12 | View Spectrum | | Predicted MS/MS | Predicted LC-MS/MS Spectrum - 20V, Positive | splash10-01p9-9100000000-4dcb939c91e313be7889 | 2016-09-12 | View Spectrum | | Predicted MS/MS | Predicted LC-MS/MS Spectrum - 40V, Positive | splash10-0006-9000000000-0ddbae5fe95cd5c2cc3a | 2016-09-12 | View Spectrum | | Predicted MS/MS | Predicted LC-MS/MS Spectrum - 10V, Negative | splash10-0a4i-7900000000-483a2fcbaa1f9e4282f8 | 2016-09-12 | View Spectrum | | Predicted MS/MS | Predicted LC-MS/MS Spectrum - 20V, Negative | splash10-0a4i-9200000000-c37acde4cab84c418b98 | 2016-09-12 | View Spectrum | | Predicted MS/MS | Predicted LC-MS/MS Spectrum - 40V, Negative | splash10-0a4l-9000000000-c2110468981b90e60c2b | 2016-09-12 | View Spectrum |
|
|---|
| NMR | | Type | Description | | View |
|---|
| 1D NMR | 1H NMR Spectrum (1D, 500 MHz, H2O, experimental) | | Spectrum | | 1D NMR | 1H NMR Spectrum (1D, 100 MHz, D2O, predicted) | | Spectrum | | 1D NMR | 13C NMR Spectrum (1D, 100 MHz, D2O, predicted) | | Spectrum | | 1D NMR | 1H NMR Spectrum (1D, 1000 MHz, D2O, predicted) | | Spectrum | | 1D NMR | 13C NMR Spectrum (1D, 1000 MHz, D2O, predicted) | | Spectrum | | 1D NMR | 1H NMR Spectrum (1D, 200 MHz, D2O, predicted) | | Spectrum | | 1D NMR | 13C NMR Spectrum (1D, 200 MHz, D2O, predicted) | | Spectrum | | 1D NMR | 1H NMR Spectrum (1D, 300 MHz, D2O, predicted) | | Spectrum | | 1D NMR | 13C NMR Spectrum (1D, 300 MHz, D2O, predicted) | | Spectrum | | 1D NMR | 1H NMR Spectrum (1D, 400 MHz, D2O, predicted) | | Spectrum | | 1D NMR | 13C NMR Spectrum (1D, 400 MHz, D2O, predicted) | | Spectrum | | 1D NMR | 1H NMR Spectrum (1D, 500 MHz, D2O, predicted) | | Spectrum | | 1D NMR | 13C NMR Spectrum (1D, 500 MHz, D2O, predicted) | | Spectrum | | 1D NMR | 1H NMR Spectrum (1D, 600 MHz, D2O, predicted) | | Spectrum | | 1D NMR | 13C NMR Spectrum (1D, 600 MHz, D2O, predicted) | | Spectrum | | 1D NMR | 1H NMR Spectrum (1D, 700 MHz, D2O, predicted) | | Spectrum | | 1D NMR | 13C NMR Spectrum (1D, 700 MHz, D2O, predicted) | | Spectrum | | 1D NMR | 1H NMR Spectrum (1D, 800 MHz, D2O, predicted) | | Spectrum | | 1D NMR | 13C NMR Spectrum (1D, 800 MHz, D2O, predicted) | | Spectrum | | 1D NMR | 1H NMR Spectrum (1D, 900 MHz, D2O, predicted) | | Spectrum | | 1D NMR | 13C NMR Spectrum (1D, 900 MHz, D2O, predicted) | | Spectrum | | 2D NMR | [1H, 13C]-HSQC NMR Spectrum (2D, 600 MHz, H2O, experimental) | | Spectrum |
|
|---|
| Pathways |
|---|
| Pathways | | Name | SMPDB/Pathwhiz | KEGG | | Glycine and Serine Metabolism | 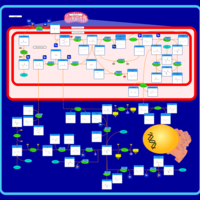 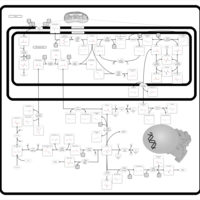 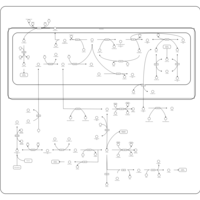 |  | | Glycerolipid Metabolism | 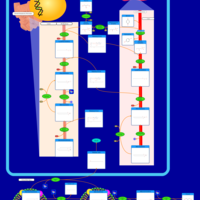   | 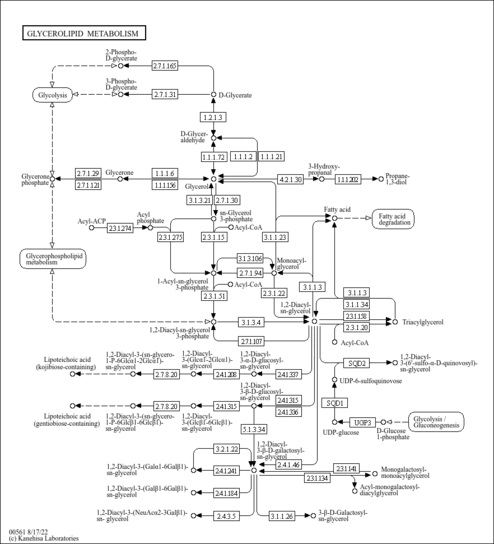 | | Glycerol Kinase Deficiency |  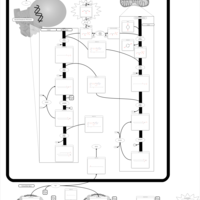 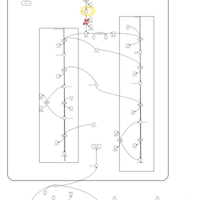 | Not Available | | D-glyceric acidura | 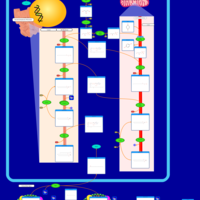 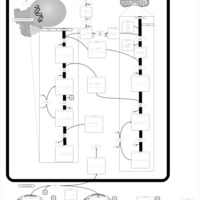 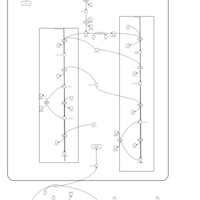 | Not Available | | Familial lipoprotein lipase deficiency |   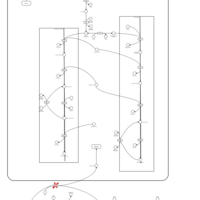 | Not Available |
|
|---|
| Protein Targets |
|---|
| Enzymes | |
|---|
| Transporters | Not Available |
|---|
| Metal Bindings | Not Available |
|---|
| Receptors | Not Available |
|---|
| Transcriptional Factors | Not Available |
|---|
| Concentrations Data |
|---|
| |
| Alien Dawg | Detected and Quantified | 0.158 mg/g dry wt | | details | | Gabriola | Detected and Quantified | 0.0624 mg/g dry wt | | details | | Island Honey | Detected and Quantified | 0.199 mg/g dry wt | | details | | Quadra | Detected and Quantified | 0.246 mg/g dry wt | | details | | Sensi Star | Detected and Quantified | 0.314 mg/g dry wt | | details | | Tangerine Dream | Detected and Quantified | 0.243 mg/g dry wt | | details |
|
|---|
| External Links |
|---|
| HMDB ID | HMDB0000139 |
|---|
| DrugBank ID | Not Available |
|---|
| Phenol Explorer Compound ID | Not Available |
|---|
| FoodDB ID | FDB012242 |
|---|
| KNApSAcK ID | C00001185 |
|---|
| Chemspider ID | 388334 |
|---|
| KEGG Compound ID | C00258 |
|---|
| BioCyc ID | GLYCERATE |
|---|
| BiGG ID | 34424 |
|---|
| Wikipedia Link | Glyceric acid |
|---|
| METLIN ID | 280 |
|---|
| PubChem Compound | 439194 |
|---|
| PDB ID | Not Available |
|---|
| ChEBI ID | 32398 |
|---|
| References |
|---|
| General References | - Habe H, Fukuoka T, Kitamoto D, Sakaki K: Biotechnological production of D-glyceric acid and its application. Appl Microbiol Biotechnol. 2009 Sep;84(3):445-52. doi: 10.1007/s00253-009-2124-3. Epub 2009 Jul 21. [PubMed:19621222 ]
- Sass JO, Fischer K, Wang R, Christensen E, Scholl-Burgi S, Chang R, Kapelari K, Walter M: D-glyceric aciduria is caused by genetic deficiency of D-glycerate kinase (GLYCTK). Hum Mutat. 2010 Dec;31(12):1280-5. doi: 10.1002/humu.21375. Epub 2010 Nov 9. [PubMed:20949620 ]
|
|---|

