| Record Information |
|---|
| Version | 1.0 |
|---|
| Created at | 2020-04-17 19:33:30 UTC |
|---|
| Updated at | 2020-12-07 19:11:59 UTC |
|---|
| CannabisDB ID | CDB005341 |
|---|
| Secondary Accession Numbers | Not Available |
|---|
| Cannabis Compound Identification |
|---|
| Common Name | 3-Keto-4-methylzymosterol |
|---|
| Description | 3-Keto-4-methylzymosterol belongs to the class of organic compounds known as cholesterols and derivatives. Cholesterols and derivatives are compounds containing a 3-hydroxylated cholestane core. 3-Keto-4-methylzymosterol is an extremely weak basic (essentially neutral) compound (based on its pKa). 3-Keto-4-methylzymosterol is expected to be in Cannabis as all living plants are known to produce and metabolize it. |
|---|
| Structure | |
|---|
| Synonyms | | Value | Source |
|---|
| (5a)-4-Methylcholesta-8,24-dien-3-one | HMDB | | (5alpha)-4-Methylcholesta-8,24-dien-3-one | HMDB | | 3-Dehydro-4-methylzymosterol | HMDB | | 4-Methyl-5a-cholesta-8,24-dien-3-one | HMDB | | 4-Methyl-5alpha-cholesta-8,24-dien-3-one | HMDB |
|
|---|
| Chemical Formula | C28H44O |
|---|
| Average Molecular Weight | 396.65 |
|---|
| Monoisotopic Molecular Weight | 396.3392 |
|---|
| IUPAC Name | (2S,15R)-2,6,15-trimethyl-14-[(2R)-6-methylhept-5-en-2-yl]tetracyclo[8.7.0.0^{2,7}.0^{11,15}]heptadec-1(10)-en-5-one |
|---|
| Traditional Name | (2S,15R)-2,6,15-trimethyl-14-[(2R)-6-methylhept-5-en-2-yl]tetracyclo[8.7.0.0^{2,7}.0^{11,15}]heptadec-1(10)-en-5-one |
|---|
| CAS Registry Number | Not Available |
|---|
| SMILES | C[C@H](CCC=C(C)C)C1CCC2C3=C(CC[C@]12C)[C@@]1(C)CCC(=O)C(C)C1CC3 |
|---|
| InChI Identifier | InChI=1S/C28H44O/c1-18(2)8-7-9-19(3)22-12-13-24-21-10-11-23-20(4)26(29)15-17-28(23,6)25(21)14-16-27(22,24)5/h8,19-20,22-24H,7,9-17H2,1-6H3/t19-,20?,22?,23?,24?,27-,28+/m1/s1 |
|---|
| InChI Key | DBPZYKHQDWKORQ-JQDPPCHWSA-N |
|---|
| Chemical Taxonomy |
|---|
| Description | Belongs to the class of organic compounds known as cholesterols and derivatives. Cholesterols and derivatives are compounds containing a 3-hydroxylated cholestane core. |
|---|
| Kingdom | Organic compounds |
|---|
| Super Class | Lipids and lipid-like molecules |
|---|
| Class | Steroids and steroid derivatives |
|---|
| Sub Class | Cholestane steroids |
|---|
| Direct Parent | Cholesterols and derivatives |
|---|
| Alternative Parents | |
|---|
| Substituents | - Cholesterol-skeleton
- Oxosteroid
- 3-oxosteroid
- Cyclic ketone
- Ketone
- Organic oxygen compound
- Organic oxide
- Hydrocarbon derivative
- Organooxygen compound
- Carbonyl group
- Aliphatic homopolycyclic compound
|
|---|
| Molecular Framework | Aliphatic homopolycyclic compounds |
|---|
| External Descriptors | Not Available |
|---|
| Ontology |
|---|
|
| Physiological effect | Health effect: |
|---|
| Disposition | Route of exposure: Source: Biological location: |
|---|
| Role | Industrial application: Biological role: |
|---|
| Physical Properties |
|---|
| State | Solid |
|---|
| Experimental Properties | | Property | Value | Reference |
|---|
| Melting Point | Not Available | Not Available | | Boiling Point | Not Available | Not Available | | Water Solubility | Not Available | Not Available | | logP | Not Available | Not Available |
|
|---|
| Predicted Properties | [] |
|---|
| Spectra |
|---|
| EI-MS/GC-MS | | Type | Description | Splash Key | View |
|---|
| Predicted GC-MS | 3-Keto-4-methylzymosterol, non-derivatized, Predicted GC-MS Spectrum - 70eV, Positive | splash10-02au-2019000000-5173500b11e18ce35b3b | Spectrum | | Predicted GC-MS | 3-Keto-4-methylzymosterol, non-derivatized, Predicted GC-MS Spectrum - 70eV, Positive | Not Available | Spectrum | | Predicted GC-MS | 3-Keto-4-methylzymosterol, non-derivatized, Predicted GC-MS Spectrum - 70eV, Positive | Not Available | Spectrum |
|
|---|
| MS/MS | | Type | Description | Splash Key | View |
|---|
| Predicted MS/MS | Predicted LC-MS/MS Spectrum - 10V, Positive | splash10-0002-0009000000-00d160bd3605d4c8b846 | 2016-09-14 | View Spectrum | | Predicted MS/MS | Predicted LC-MS/MS Spectrum - 20V, Positive | splash10-00pv-1119000000-e5a9171fb6049ee63431 | 2016-09-14 | View Spectrum | | Predicted MS/MS | Predicted LC-MS/MS Spectrum - 40V, Positive | splash10-015i-2119000000-6b021eb86e51393d9798 | 2016-09-14 | View Spectrum | | Predicted MS/MS | Predicted LC-MS/MS Spectrum - 10V, Negative | splash10-0002-0009000000-e91a27c7e4df70239c24 | 2016-09-14 | View Spectrum | | Predicted MS/MS | Predicted LC-MS/MS Spectrum - 20V, Negative | splash10-0002-0009000000-766b7ce92b3bc6de20e7 | 2016-09-14 | View Spectrum | | Predicted MS/MS | Predicted LC-MS/MS Spectrum - 40V, Negative | splash10-01t9-3009000000-b4ba7e91fae9c3e7a4ba | 2016-09-14 | View Spectrum | | Predicted MS/MS | Predicted LC-MS/MS Spectrum - 10V, Positive | splash10-002b-0019000000-f87374bf70bdfa4a699f | 2021-09-24 | View Spectrum | | Predicted MS/MS | Predicted LC-MS/MS Spectrum - 20V, Positive | splash10-0ap0-3259000000-e97d8860c45e22b2a89d | 2021-09-24 | View Spectrum | | Predicted MS/MS | Predicted LC-MS/MS Spectrum - 40V, Positive | splash10-0a4i-6951000000-99c4649de525703330a7 | 2021-09-24 | View Spectrum | | Predicted MS/MS | Predicted LC-MS/MS Spectrum - 10V, Negative | splash10-0002-0009000000-2f6aa9af0bd8878fb6c6 | 2021-09-24 | View Spectrum | | Predicted MS/MS | Predicted LC-MS/MS Spectrum - 20V, Negative | splash10-0002-0009000000-2f6aa9af0bd8878fb6c6 | 2021-09-24 | View Spectrum | | Predicted MS/MS | Predicted LC-MS/MS Spectrum - 40V, Negative | splash10-0007-0009000000-3e9131f464f27a3024a5 | 2021-09-24 | View Spectrum |
|
|---|
| NMR | Not Available |
|---|
| Pathways |
|---|
| Pathways | | Name | SMPDB/Pathwhiz | KEGG | | Steroid Biosynthesis | 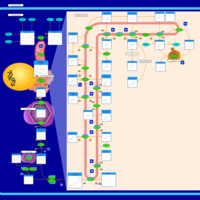 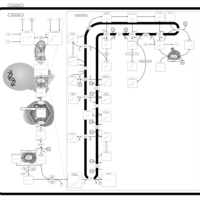 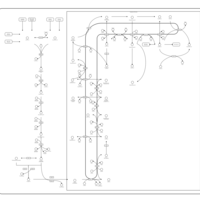 |  | | Smith-Lemli-Opitz Syndrome (SLOS) | 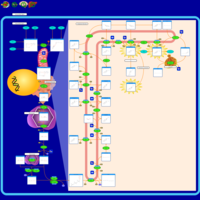 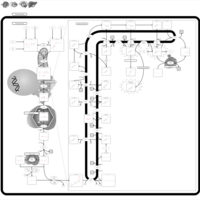 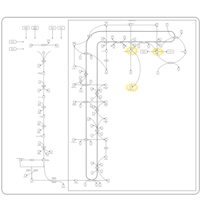 | Not Available | | CHILD Syndrome | 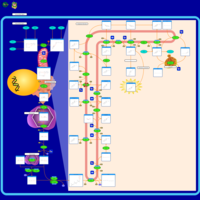 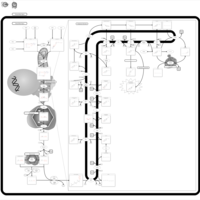 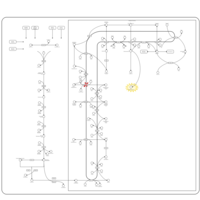 | Not Available | | Desmosterolosis |  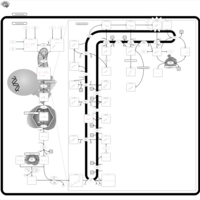 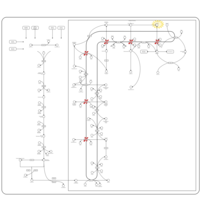 | Not Available | | Chondrodysplasia Punctata II, X Linked Dominant (CDPX2) | 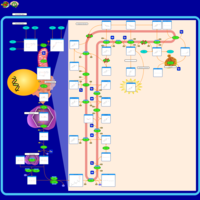 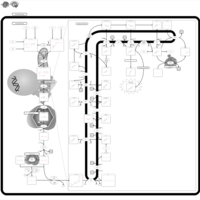 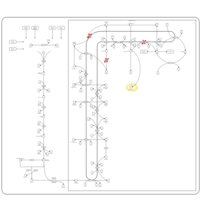 | Not Available |
|
|---|
| Protein Targets |
|---|
| Enzymes | |
| Sterol-4-alpha-carboxylate 3-dehydrogenase, decarboxylating | NSDHL | Xq28 | Q15738 | details | | 3-keto-steroid reductase | HSD17B7 | 1q23 | P56937 | details |
|
|---|
| Transporters | Not Available |
|---|
| Metal Bindings | Not Available |
|---|
| Receptors | Not Available |
|---|
| Transcriptional Factors | Not Available |
|---|
| Concentrations Data |
|---|
| Not Available |
|---|
| External Links |
|---|
| HMDB ID | HMDB0006838 |
|---|
| DrugBank ID | Not Available |
|---|
| Phenol Explorer Compound ID | Not Available |
|---|
| FoodDB ID | FDB024112 |
|---|
| KNApSAcK ID | Not Available |
|---|
| Chemspider ID | Not Available |
|---|
| KEGG Compound ID | C15816 |
|---|
| BioCyc ID | Not Available |
|---|
| BiGG ID | Not Available |
|---|
| Wikipedia Link | Not Available |
|---|
| METLIN ID | Not Available |
|---|
| PubChem Compound | 53477900 |
|---|
| PDB ID | Not Available |
|---|
| ChEBI ID | 505093 |
|---|
| References |
|---|
| General References | Not Available |
|---|
