| Record Information |
|---|
| Version | 1.0 |
|---|
| Created at | 2020-04-17 18:48:22 UTC |
|---|
| Updated at | 2020-12-07 19:11:13 UTC |
|---|
| CannabisDB ID | CDB004899 |
|---|
| Secondary Accession Numbers | Not Available |
|---|
| Cannabis Compound Identification |
|---|
| Common Name | Lanosterol |
|---|
| Description | Lanosterin, also known as botalan base 138 or lanosterol, belongs to the class of organic compounds known as triterpenoids. These are terpene molecules containing six isoprene units. Thus, lanosterin is considered to be a sterol lipid molecule. Lanosterin is a very hydrophobic molecule, practically insoluble (in water), and relatively neutral. Lanosterol is expected to be in Cannabis as all living plants are known to produce and metabolize it. |
|---|
| Structure | |
|---|
| Synonyms | | Value | Source |
|---|
| (3beta)-Lanosta-8,24-dien-3-ol | ChEBI | | (3beta,5alpha)-4,4,14-Trimethylcholesta-8,24-dien-3-ol | ChEBI | | 4,4',14alpha-Trimethyl-5alpha-cholesta-8,24-dien-3beta-ol | ChEBI | | (3b)-Lanosta-8,24-dien-3-ol | Generator | | (3β)-Lanosta-8,24-dien-3-ol | Generator | | (3b,5a)-4,4,14-Trimethylcholesta-8,24-dien-3-ol | Generator | | (3β,5α)-4,4,14-Trimethylcholesta-8,24-dien-3-ol | Generator | | 4,4',14a-Trimethyl-5a-cholesta-8,24-dien-3b-ol | Generator | | 4,4',14α-Trimethyl-5α-cholesta-8,24-dien-3β-ol | Generator | | (3 beta)-Lanosta-8,24-dien-3-ol | HMDB | | (3alpha)-4,4,14-Trimethyl-cholesta-8,24-dien-3-ol | HMDB | | (3beta,5alpha)-4,4,14-Trimethyl-cholesta-8,24-dien-3-ol | HMDB | | Botalan base 138 | HMDB | | Lanosta-8,24-dien-3-ol | HMDB | | Lanosta-8,24-dien-3beta-ol | HMDB | | Lanosta-8,24-dienol | HMDB | | Lanosterol | HMDB | | Lanster | HMDB | | 4,4,14 alpha-Trimethyl-5 alpha-cholesta-8,24-dien-3 beta-ol | MeSH, HMDB | | Kryptosterol | MeSH, HMDB | | Lanosterin | ChEBI | | 3beta-Hydroxy-lansota-8,24-dien-21-oic acid | HMDB | | 3beta-Hydroxylanosta-8,24-diene | HMDB | | 3β-Hydroxy-lansota-8,24-dien-21-oic acid | HMDB | | 3β-Hydroxylanosta-8,24-diene | HMDB | | 4,4,14alpha-Trimethylcholesta-8,24-dien-3beta-ol | HMDB | | 4,4,14α-Trimethylcholesta-8,24-dien-3β-ol | HMDB | | 5alpha-Lanosta-8,24-dien-3beta-ol | HMDB | | 5α-Lanosta-8,24-dien-3β-ol | HMDB | | Lanosta-8,24-dien-3β-ol | HMDB | | Lanostadien-3beta-ol | HMDB | | Lanostadien-3β-ol | HMDB |
|
|---|
| Chemical Formula | C30H50O |
|---|
| Average Molecular Weight | 426.72 |
|---|
| Monoisotopic Molecular Weight | 426.3862 |
|---|
| IUPAC Name | (2S,5S,7R,11R,14R,15R)-2,6,6,11,15-pentamethyl-14-[(2R)-6-methylhept-5-en-2-yl]tetracyclo[8.7.0.0^{2,7}.0^{11,15}]heptadec-1(10)-en-5-ol |
|---|
| Traditional Name | (2S,5S,7R,11R,14R,15R)-2,6,6,11,15-pentamethyl-14-[(2R)-6-methylhept-5-en-2-yl]tetracyclo[8.7.0.0^{2,7}.0^{11,15}]heptadec-1(10)-en-5-ol |
|---|
| CAS Registry Number | 79-63-0 |
|---|
| SMILES | [H][C@@]1(CC[C@@]2(C)C3=C(CC[C@]12C)[C@@]1(C)CC[C@H](O)C(C)(C)[C@]1([H])CC3)[C@H](C)CCC=C(C)C |
|---|
| InChI Identifier | InChI=1S/C30H50O/c1-20(2)10-9-11-21(3)22-14-18-30(8)24-12-13-25-27(4,5)26(31)16-17-28(25,6)23(24)15-19-29(22,30)7/h10,21-22,25-26,31H,9,11-19H2,1-8H3/t21-,22-,25+,26+,28-,29-,30+/m1/s1 |
|---|
| InChI Key | CAHGCLMLTWQZNJ-BQNIITSRSA-N |
|---|
| Chemical Taxonomy |
|---|
| Description | Belongs to the class of organic compounds known as triterpenoids. These are terpene molecules containing six isoprene units. |
|---|
| Kingdom | Organic compounds |
|---|
| Super Class | Lipids and lipid-like molecules |
|---|
| Class | Prenol lipids |
|---|
| Sub Class | Triterpenoids |
|---|
| Direct Parent | Triterpenoids |
|---|
| Alternative Parents | |
|---|
| Substituents | - Triterpenoid
- 14-alpha-methylsteroid
- 3-beta-hydroxysteroid
- Hydroxysteroid
- 3-hydroxysteroid
- Steroid
- Cyclic alcohol
- Secondary alcohol
- Organic oxygen compound
- Hydrocarbon derivative
- Organooxygen compound
- Alcohol
- Aliphatic homopolycyclic compound
|
|---|
| Molecular Framework | Aliphatic homopolycyclic compounds |
|---|
| External Descriptors | |
|---|
| Ontology |
|---|
|
| Disposition | Route of exposure: Source: Biological location: |
|---|
| Role | Biological role: Industrial application: |
|---|
| Physical Properties |
|---|
| State | Solid |
|---|
| Experimental Properties | | Property | Value | Reference |
|---|
| Melting Point | 140.5 °C | Not Available | | Boiling Point | Not Available | Not Available | | Water Solubility | Not Available | Not Available | | logP | Not Available | Not Available |
|
|---|
| Predicted Properties | [] |
|---|
| Spectra |
|---|
| EI-MS/GC-MS | | Type | Description | Splash Key | View |
|---|
| EI-MS | Mass Spectrum (Electron Ionization) | splash10-05mo-9731200000-b1bc6bb68e352e3bd687 | 2015-03-01 | View Spectrum | | GC-MS | Lanosterol, 1 TMS, GC-MS Spectrum | splash10-0a4l-4910000000-0fe7f84c95ab0a849fe9 | Spectrum | | GC-MS | Lanosterol, non-derivatized, GC-MS Spectrum | splash10-03dl-2953300000-ae1832f19369485ff53c | Spectrum | | GC-MS | Lanosterol, non-derivatized, GC-MS Spectrum | splash10-0aou-9820200000-573525b346ad3c9eef59 | Spectrum | | GC-MS | Lanosterol, non-derivatized, GC-MS Spectrum | splash10-0a4l-4910000000-0fe7f84c95ab0a849fe9 | Spectrum | | GC-MS | Lanosterol, non-derivatized, GC-MS Spectrum | splash10-0a4l-2910000000-c67a1f122f33698a14f6 | Spectrum | | Predicted GC-MS | Lanosterol, non-derivatized, Predicted GC-MS Spectrum - 70eV, Positive | splash10-03dj-1009400000-e2b516142f5128a7b5f6 | Spectrum | | Predicted GC-MS | Lanosterol, 1 TMS, Predicted GC-MS Spectrum - 70eV, Positive | splash10-001i-3002900000-35e63c0bf6f033cb847c | Spectrum | | Predicted GC-MS | Lanosterol, non-derivatized, Predicted GC-MS Spectrum - 70eV, Positive | Not Available | Spectrum | | Predicted GC-MS | Lanosterol, non-derivatized, Predicted GC-MS Spectrum - 70eV, Positive | Not Available | Spectrum |
|
|---|
| MS/MS | | Type | Description | Splash Key | View |
|---|
| MS/MS | LC-MS/MS Spectrum - n/a 1V, positive | splash10-0a6r-0110900000-5e42e1f2d9ad534c003c | 2020-07-22 | View Spectrum | | MS/MS | LC-MS/MS Spectrum - Orbitrap 13V, positive | splash10-0a4i-0230900000-10f58fa6e118cd7c5997 | 2020-07-22 | View Spectrum | | MS/MS | LC-MS/MS Spectrum - Orbitrap 17V, positive | splash10-0a4i-1791800000-178153ad92eececb33c4 | 2020-07-22 | View Spectrum | | MS/MS | LC-MS/MS Spectrum - Orbitrap 20V, positive | splash10-0a4i-2980300000-6ce95c7f8b15b35475af | 2020-07-22 | View Spectrum | | MS/MS | LC-MS/MS Spectrum - Orbitrap 23V, positive | splash10-0a4j-2950100000-48f86ff7a22c596aa44d | 2020-07-22 | View Spectrum | | MS/MS | LC-MS/MS Spectrum - Orbitrap 27V, positive | splash10-0592-3940000000-c35d0cb6a2e4e291e8f8 | 2020-07-22 | View Spectrum | | MS/MS | LC-MS/MS Spectrum - Orbitrap 30V, positive | splash10-0592-3920000000-d717fdd3322a881bef99 | 2020-07-22 | View Spectrum | | MS/MS | LC-MS/MS Spectrum - Orbitrap 34V, positive | splash10-0592-4910000000-4c6d07e3a8e48ae88580 | 2020-07-22 | View Spectrum | | MS/MS | LC-MS/MS Spectrum - Orbitrap 41V, positive | splash10-0a4j-5900000000-4c8b72037dbeae9a46d1 | 2020-07-22 | View Spectrum | | MS/MS | LC-MS/MS Spectrum - Orbitrap 52V, positive | splash10-067m-8900000000-522b8688d50362297c41 | 2020-07-22 | View Spectrum | | MS/MS | LC-MS/MS Spectrum - Orbitrap 62V, positive | splash10-0aou-9700000000-70d433827c1a6dab0c41 | 2020-07-22 | View Spectrum | | MS/MS | LC-MS/MS Spectrum - Orbitrap 76V, positive | splash10-05ox-9500000000-5b122dea636d3e3b5916 | 2020-07-22 | View Spectrum | | MS/MS | LC-MS/MS Spectrum - Orbitrap 90V, positive | splash10-05r3-9500000000-0c1c5cbd994f8406f30e | 2020-07-22 | View Spectrum | | MS/MS | LC-MS/MS Spectrum - Orbitrap 111V, positive | splash10-05r3-9600000000-5cb8ae11d071fa19f206 | 2020-07-22 | View Spectrum | | MS/MS | LC-MS/MS Spectrum - n/a 29V, positive | splash10-0ktf-0392000000-03cda521ce6e102d92e4 | 2020-07-22 | View Spectrum | | MS/MS | LC-MS/MS Spectrum - n/a 29V, positive | splash10-059j-2900000000-8086693fb040e5ed7501 | 2020-07-22 | View Spectrum | | MS/MS | LC-MS/MS Spectrum - n/a 29V, positive | splash10-0a4i-4900000000-c58ff2dc28c6ba639997 | 2020-07-22 | View Spectrum | | MS/MS | LC-MS/MS Spectrum - n/a 29V, positive | splash10-01vk-0900000000-805937445cceda0cd70a | 2020-07-22 | View Spectrum | | MS/MS | LC-MS/MS Spectrum - n/a 29V, positive | splash10-0aor-0900000000-22d304b445b737121aa7 | 2020-07-22 | View Spectrum | | Predicted MS/MS | Predicted LC-MS/MS Spectrum - 10V, Positive | splash10-0a6r-0002900000-2cfcf7ce4b5b21140eae | 2016-08-01 | View Spectrum | | Predicted MS/MS | Predicted LC-MS/MS Spectrum - 20V, Positive | splash10-0pdi-2139500000-9a25961000db9796d6da | 2016-08-01 | View Spectrum | | Predicted MS/MS | Predicted LC-MS/MS Spectrum - 40V, Positive | splash10-0uxr-4259100000-9f7f26265abec4122e35 | 2016-08-01 | View Spectrum | | Predicted MS/MS | Predicted LC-MS/MS Spectrum - 10V, Negative | splash10-004i-0000900000-77a57cecc2069bee1926 | 2016-08-03 | View Spectrum | | Predicted MS/MS | Predicted LC-MS/MS Spectrum - 20V, Negative | splash10-004i-0000900000-5b0f06bf8120e780e30c | 2016-08-03 | View Spectrum | | Predicted MS/MS | Predicted LC-MS/MS Spectrum - 40V, Negative | splash10-0a4l-1009400000-421e305d47b29da427d2 | 2016-08-03 | View Spectrum |
|
|---|
| NMR | | Type | Description | | View |
|---|
| 1D NMR | 13C NMR Spectrum (1D, 100 MHz, H2O, predicted) | | Spectrum | | 1D NMR | 1H NMR Spectrum (1D, 100 MHz, H2O, predicted) | | Spectrum | | 1D NMR | 13C NMR Spectrum (1D, 1000 MHz, H2O, predicted) | | Spectrum | | 1D NMR | 1H NMR Spectrum (1D, 1000 MHz, H2O, predicted) | | Spectrum | | 1D NMR | 13C NMR Spectrum (1D, 200 MHz, H2O, predicted) | | Spectrum | | 1D NMR | 1H NMR Spectrum (1D, 200 MHz, H2O, predicted) | | Spectrum | | 1D NMR | 13C NMR Spectrum (1D, 300 MHz, H2O, predicted) | | Spectrum | | 1D NMR | 1H NMR Spectrum (1D, 300 MHz, H2O, predicted) | | Spectrum | | 1D NMR | 13C NMR Spectrum (1D, 400 MHz, H2O, predicted) | | Spectrum | | 1D NMR | 1H NMR Spectrum (1D, 400 MHz, H2O, predicted) | | Spectrum | | 1D NMR | 13C NMR Spectrum (1D, 500 MHz, H2O, predicted) | | Spectrum | | 1D NMR | 1H NMR Spectrum (1D, 500 MHz, H2O, predicted) | | Spectrum | | 1D NMR | 13C NMR Spectrum (1D, 600 MHz, H2O, predicted) | | Spectrum | | 1D NMR | 1H NMR Spectrum (1D, 600 MHz, H2O, predicted) | | Spectrum | | 1D NMR | 13C NMR Spectrum (1D, 700 MHz, H2O, predicted) | | Spectrum | | 1D NMR | 1H NMR Spectrum (1D, 700 MHz, H2O, predicted) | | Spectrum | | 1D NMR | 13C NMR Spectrum (1D, 800 MHz, H2O, predicted) | | Spectrum | | 1D NMR | 1H NMR Spectrum (1D, 800 MHz, H2O, predicted) | | Spectrum | | 1D NMR | 13C NMR Spectrum (1D, 900 MHz, H2O, predicted) | | Spectrum | | 1D NMR | 1H NMR Spectrum (1D, 900 MHz, H2O, predicted) | | Spectrum |
|
|---|
| Pathways |
|---|
| Pathways | | Name | SMPDB/Pathwhiz | KEGG | | Steroid Biosynthesis | 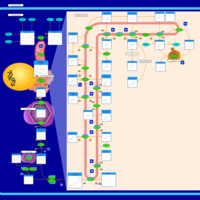 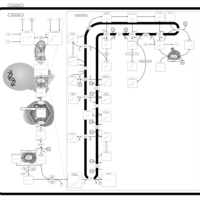 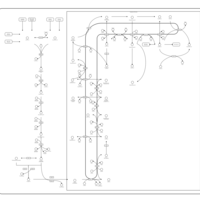 |  | | Smith-Lemli-Opitz Syndrome (SLOS) | 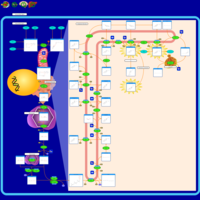 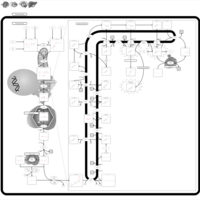  | Not Available | | CHILD Syndrome | 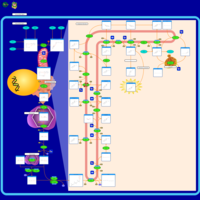 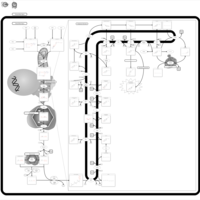 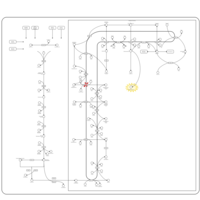 | Not Available | | Desmosterolosis |  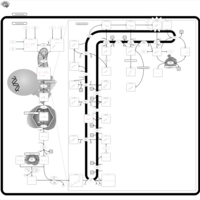 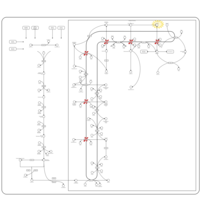 | Not Available | | Chondrodysplasia Punctata II, X Linked Dominant (CDPX2) | 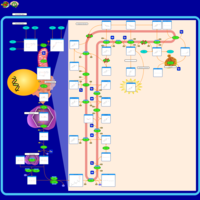 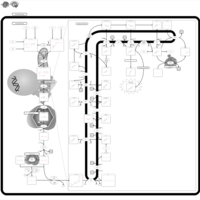 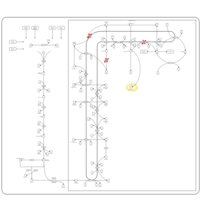 | Not Available |
|
|---|
| Protein Targets |
|---|
| Enzymes | |
|---|
| Transporters | Not Available |
|---|
| Metal Bindings | |
|---|
| Receptors | Not Available |
|---|
| Transcriptional Factors | Not Available |
|---|
| Concentrations Data |
|---|
| Not Available |
|---|
| External Links |
|---|
| HMDB ID | HMDB0001251 |
|---|
| DrugBank ID | DB03696 |
|---|
| Phenol Explorer Compound ID | Not Available |
|---|
| FoodDB ID | FDB013802 |
|---|
| KNApSAcK ID | C00003657 |
|---|
| Chemspider ID | 216175 |
|---|
| KEGG Compound ID | C01724 |
|---|
| BioCyc ID | LANOSTEROL |
|---|
| BiGG ID | 38253 |
|---|
| Wikipedia Link | Lanosterol |
|---|
| METLIN ID | 6108 |
|---|
| PubChem Compound | 246983 |
|---|
| PDB ID | Not Available |
|---|
| ChEBI ID | 16521 |
|---|
| References |
|---|
| General References | Not Available |
|---|
