| Record Information |
|---|
| Version | 1.0 |
|---|
| Created at | 2020-04-17 18:47:56 UTC |
|---|
| Updated at | 2020-11-18 16:38:56 UTC |
|---|
| CannabisDB ID | CDB004895 |
|---|
| Secondary Accession Numbers | Not Available |
|---|
| Cannabis Compound Identification |
|---|
| Common Name | 5-Aminoimidazole ribonucleotide |
|---|
| Description | 5-Aminoimidazole ribonucleotide, also known as 5'-phosphoribosyl-5-aminoimidazole or AIR, belongs to the class of organic compounds known as pentose phosphates. These are carbohydrate derivatives containing a pentose substituted by one or more phosphate groups. 5-Aminoimidazole ribonucleotide is a very strong basic compound (based on its pKa). 5-Aminoimidazole ribonucleotide exists in all living species, ranging from bacteria to humans. Within humans, 5-aminoimidazole ribonucleotide participates in a number of enzymatic reactions. In particular, 5-aminoimidazole ribonucleotide can be biosynthesized from 5-amino-1-(5-phospho-D-ribosyl)imidazole-4-carboxylate; which is mediated by the enzyme multifunctional protein ADE2. In addition, 5-aminoimidazole ribonucleotide can be biosynthesized from phosphoribosylformylglycineamidine through its interaction with the enzyme trifunctional purine biosynthetic protein adenosine-3. A 1-(phosphoribosyl)imidazole that is 5-aminoimidazole in which the proton at position 1 has been replaced by a 5-phospho-beta-D-ribofuranosyl group. In humans, 5-aminoimidazole ribonucleotide is involved in the metabolic disorder called the gout or kelley-seegmiller syndrome pathway. Outside of the human body, 5-Aminoimidazole ribonucleotide has been detected, but not quantified in, several different foods, such as cascade huckleberries, anises, red bell peppers, spirulina, and black mulberries. This could make 5-aminoimidazole ribonucleotide a potential biomarker for the consumption of these foods. 5-Aminoimidazole ribonucleotide is expected to be in Cannabis as all living plants are known to produce and metabolize it. |
|---|
| Structure | |
|---|
| Synonyms | | Value | Source |
|---|
| 1-(5'-Phosphoribosyl)-5-aminoimidazole | ChEBI | | 1-(5-Phospho-D-ribosyl)-5-aminoimidazole | ChEBI | | 5'-Phosphoribosyl-5-aminoimidazole | ChEBI | | 5-Amino-1-beta-D-ribofuranosyl-5'-(dihydrogen phosphate)-imidazole | ChEBI | | 5-Amino-1-ribofuranosylimidazole 5'-phosphate | ChEBI | | AIR | ChEBI | | Aminoimidazole ribonucleotide | ChEBI | | Aminoimidazole ribotide | ChEBI | | Phosphoribosylaminoimidazole | ChEBI | | 5-Amino-1-(5-phospho-D-ribosyl)imidazole | Kegg | | 5-Amino-1-(5-phospho-beta-D-ribosyl)imidazole | Kegg | | 5-Amino-1-b-D-ribofuranosyl-5'-(dihydrogen phosphate)-imidazole | Generator | | 5-Amino-1-b-D-ribofuranosyl-5'-(dihydrogen phosphoric acid)-imidazole | Generator | | 5-Amino-1-beta-D-ribofuranosyl-5'-(dihydrogen phosphoric acid)-imidazole | Generator | | 5-Amino-1-β-D-ribofuranosyl-5'-(dihydrogen phosphate)-imidazole | Generator | | 5-Amino-1-β-D-ribofuranosyl-5'-(dihydrogen phosphoric acid)-imidazole | Generator | | 5-Amino-1-ribofuranosylimidazole 5'-phosphoric acid | Generator | | 5-Amino-1-(5-phospho-b-D-ribosyl)imidazole | Generator | | 5-Amino-1-(5-phospho-β-D-ribosyl)imidazole | Generator | | 5-Aminoimidazole ribotide | HMDB | | Aminoimidazole ribotide, (beta-D-ribofuranosyl)-isomer | HMDB | | Aminoimidazole ribotide, (alpha-D-ribofuranosyl)-isomer | HMDB | | Aminoimidazole ribotide, phosphonoribofuranosyl-isomer | HMDB |
|
|---|
| Chemical Formula | C8H14N3O7P |
|---|
| Average Molecular Weight | 295.19 |
|---|
| Monoisotopic Molecular Weight | 295.0569 |
|---|
| IUPAC Name | {[(2R,3S,4R,5R)-5-(5-amino-1H-imidazol-1-yl)-3,4-dihydroxyoxolan-2-yl]methoxy}phosphonic acid |
|---|
| Traditional Name | 5-aminoimidazole ribotide |
|---|
| CAS Registry Number | 25635-88-5 |
|---|
| SMILES | NC1=CN=CN1[C@@H]1O[C@H](COP(O)(O)=O)[C@@H](O)[C@H]1O |
|---|
| InChI Identifier | InChI=1S/C8H14N3O7P/c9-5-1-10-3-11(5)8-7(13)6(12)4(18-8)2-17-19(14,15)16/h1,3-4,6-8,12-13H,2,9H2,(H2,14,15,16)/t4-,6-,7-,8-/m1/s1 |
|---|
| InChI Key | PDACUKOKVHBVHJ-XVFCMESISA-N |
|---|
| Chemical Taxonomy |
|---|
| Description | Belongs to the class of organic compounds known as pentose phosphates. These are carbohydrate derivatives containing a pentose substituted by one or more phosphate groups. |
|---|
| Kingdom | Organic compounds |
|---|
| Super Class | Organic oxygen compounds |
|---|
| Class | Organooxygen compounds |
|---|
| Sub Class | Carbohydrates and carbohydrate conjugates |
|---|
| Direct Parent | Pentose phosphates |
|---|
| Alternative Parents | |
|---|
| Substituents | - Pentose phosphate
- Pentose-5-phosphate
- Imidazole ribonucleoside
- Glycosyl compound
- N-glycosyl compound
- Monosaccharide phosphate
- Monoalkyl phosphate
- Aminoimidazole
- Alkyl phosphate
- Phosphoric acid ester
- N-substituted imidazole
- Organic phosphoric acid derivative
- Heteroaromatic compound
- Azole
- Tetrahydrofuran
- Imidazole
- 1,2-diol
- Secondary alcohol
- Oxacycle
- Organoheterocyclic compound
- Azacycle
- Organic oxide
- Primary amine
- Organic nitrogen compound
- Alcohol
- Hydrocarbon derivative
- Amine
- Organopnictogen compound
- Organonitrogen compound
- Aromatic heteromonocyclic compound
|
|---|
| Molecular Framework | Aromatic heteromonocyclic compounds |
|---|
| External Descriptors | Not Available |
|---|
| Ontology |
|---|
|
| Disposition | Route of exposure: Source: Biological location: |
|---|
| Physical Properties |
|---|
| State | Solid |
|---|
| Experimental Properties | | Property | Value | Reference |
|---|
| Melting Point | Not Available | Not Available | | Boiling Point | Not Available | Not Available | | Water Solubility | Not Available | Not Available | | logP | Not Available | Not Available |
|
|---|
| Predicted Properties | [] |
|---|
| Spectra |
|---|
| EI-MS/GC-MS | | Type | Description | Splash Key | View |
|---|
| Predicted GC-MS | 5-Aminoimidazole ribonucleotide, non-derivatized, Predicted GC-MS Spectrum - 70eV, Positive | splash10-0002-9310000000-83cf50bc3f675ec17ccb | Spectrum | | Predicted GC-MS | 5-Aminoimidazole ribonucleotide, 2 TMS, Predicted GC-MS Spectrum - 70eV, Positive | splash10-01vk-9314000000-ed8269ba053b2e3a4fc4 | Spectrum | | Predicted GC-MS | 5-Aminoimidazole ribonucleotide, non-derivatized, Predicted GC-MS Spectrum - 70eV, Positive | Not Available | Spectrum |
|
|---|
| MS/MS | | Type | Description | Splash Key | View |
|---|
| Predicted MS/MS | Predicted LC-MS/MS Spectrum - 10V, Positive | splash10-001i-9130000000-143cf7dfaf8cc77d6280 | 2015-09-14 | View Spectrum | | Predicted MS/MS | Predicted LC-MS/MS Spectrum - 20V, Positive | splash10-001i-9000000000-26b4d68163a88586005b | 2015-09-14 | View Spectrum | | Predicted MS/MS | Predicted LC-MS/MS Spectrum - 40V, Positive | splash10-001i-9000000000-87449f0e16a1690c4873 | 2015-09-14 | View Spectrum | | Predicted MS/MS | Predicted LC-MS/MS Spectrum - 10V, Negative | splash10-0036-9160000000-50e60dc03e3bb2050636 | 2015-09-15 | View Spectrum | | Predicted MS/MS | Predicted LC-MS/MS Spectrum - 20V, Negative | splash10-0059-9000000000-797c3e0faf96fe4330aa | 2015-09-15 | View Spectrum | | Predicted MS/MS | Predicted LC-MS/MS Spectrum - 40V, Negative | splash10-004i-9000000000-9e3a71c432cedbcd3892 | 2015-09-15 | View Spectrum | | Predicted MS/MS | Predicted LC-MS/MS Spectrum - 10V, Positive | splash10-0002-2490000000-7dd58fcac26f75e7c46f | 2021-09-25 | View Spectrum | | Predicted MS/MS | Predicted LC-MS/MS Spectrum - 20V, Positive | splash10-001i-9000000000-6dd5f8e7028cd1ab65d2 | 2021-09-25 | View Spectrum | | Predicted MS/MS | Predicted LC-MS/MS Spectrum - 40V, Positive | splash10-001i-9000000000-7542d4d80a10b03631f0 | 2021-09-25 | View Spectrum | | Predicted MS/MS | Predicted LC-MS/MS Spectrum - 10V, Negative | splash10-002f-9080000000-e3666a5af349af6427f9 | 2021-09-25 | View Spectrum | | Predicted MS/MS | Predicted LC-MS/MS Spectrum - 20V, Negative | splash10-004i-9000000000-2dd173dfe9a30642b89d | 2021-09-25 | View Spectrum | | Predicted MS/MS | Predicted LC-MS/MS Spectrum - 40V, Negative | splash10-004i-9000000000-7c5243a801196e080ddb | 2021-09-25 | View Spectrum |
|
|---|
| NMR | Not Available |
|---|
| Pathways |
|---|
| Pathways | | Name | SMPDB/Pathwhiz | KEGG | | Purine Metabolism |  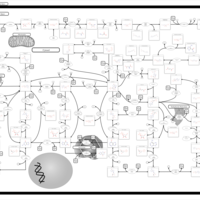 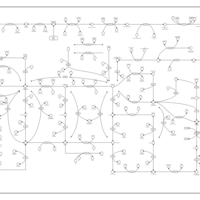 |  | | Adenosine Deaminase Deficiency |  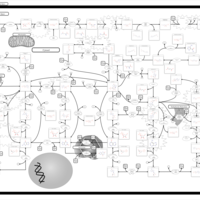 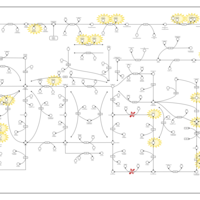 | Not Available | | Adenylosuccinate Lyase Deficiency | 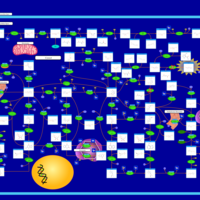 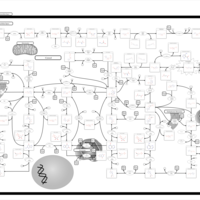 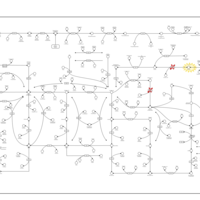 | Not Available | | Gout or Kelley-Seegmiller Syndrome | 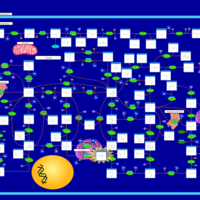 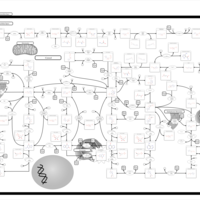 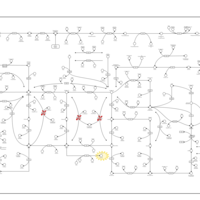 | Not Available | | Lesch-Nyhan Syndrome (LNS) | 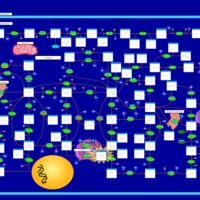 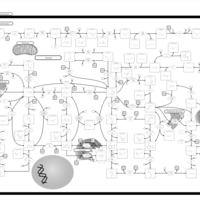  | Not Available |
|
|---|
| Protein Targets |
|---|
| Enzymes | |
| Trifunctional purine biosynthetic protein adenosine-3 | GART | 21q22.11 | P22102 | details | | Multifunctional protein ADE2 | PAICS | 4q12 | P22234 | details |
|
|---|
| Transporters | Not Available |
|---|
| Metal Bindings | |
| Trifunctional purine biosynthetic protein adenosine-3 | GART | 21q22.11 | P22102 | details |
|
|---|
| Receptors | Not Available |
|---|
| Transcriptional Factors | Not Available |
|---|
| Concentrations Data |
|---|
| Not Available |
|---|
| External Links |
|---|
| HMDB ID | HMDB0001235 |
|---|
| DrugBank ID | Not Available |
|---|
| Phenol Explorer Compound ID | Not Available |
|---|
| FoodDB ID | FDB022504 |
|---|
| KNApSAcK ID | C00019655 |
|---|
| Chemspider ID | 141854 |
|---|
| KEGG Compound ID | C03373 |
|---|
| BioCyc ID | 5-PHOSPHORIBOSYL-5-AMINOIMIDAZOLE |
|---|
| BiGG ID | 41727 |
|---|
| Wikipedia Link | 5-Aminoimidazole_ribotide |
|---|
| METLIN ID | 6097 |
|---|
| PubChem Compound | 161500 |
|---|
| PDB ID | Not Available |
|---|
| ChEBI ID | 138560 |
|---|
| References |
|---|
| General References | Not Available |
|---|
