| Record Information |
|---|
| Version | 1.0 |
|---|
| Created at | 2020-03-19 00:49:59 UTC |
|---|
| Updated at | 2020-12-07 19:07:39 UTC |
|---|
| CannabisDB ID | CDB000666 |
|---|
| Secondary Accession Numbers | Not Available |
|---|
| Cannabis Compound Identification |
|---|
| Common Name | Propylene glycol |
|---|
| Description | Propylene glycol, also known as 1,2-propanediol, belongs to the class of organic compounds known as 1,2-diols. These are polyols containing two adjacent hydroxy groups. Propylene glycol is an organic compound (a diol alcohol), usually a tasteless, odourless, and colourless clear oily liquid that is hygroscopic and miscible with water, acetone, and chloroform. It is manufactured by the hydration of propylene oxide. Propylene glycol is used as a solvent for intravenous, oral, and topical pharmaceutical preparations. It is generally considered safe to be used on humans. However, in large doses, it can be toxic, especially if given over a short period of time. Intravenous lorazepam contains the largest amount of propylene glycol of commonly used drugs. In adults with normal liver and kidney function, the terminal half-life of propylene glycol ranges from 1.4 to 3.3 hours. Propylene glycol is metabolized by the liver to form lactate, acetate, and pyruvate. The nonmetabolized drug is excreted in the urine mainly as the glucuronide conjugate, approximately 12 to 45 percent is excreted unchanged in urine. Renal clearance decreases as the dose administered increases (390 ml/minute/173 m2 at a dose of 5 g/day but only 144 ml/minute/173 m2 at a dose of 21 g/day). These data suggest that renal clearance declines at higher propylene glycol doses because of the saturation of proximal tubular secretion of the drug. As an acceptable level of propylene glycol has not been defined, the clinical implication of a propylene glycol level is unclear. The World Health Organization (WHO) recommends a maximum consumption of 25 mg/kg/day (1.8 g/day for a 75 kg male) of propylene glycol when used as a food additive, but this limit does not address its use as a drug solvent. No maximum dose is recommended in the literature for intravenous therapy with propylene glycol. Intoxication occurs at much higher doses than the WHO dose limit and is exclusive to pharmacologic exposure. Propylene glycol toxicity includes the development of serum hyperosmolality, lactic acidosis, and kidney failure. It has been suggested that proximal tubular necrosis is the cause of acute kidney injury from propylene glycol. Along these lines, proximal tubular cell injury occurs in cultured human cells exposed to propylene glycol. Acute tubular necrosis was described with propylene glycol toxicity in a case of concomitant administration of intravenous lorazepam and trimethoprim sulfamethoxazole. Propylene glycol induced intoxication can also mimic sepsis or systemic inflammatory response syndrome (SIRS). Patients suspected of having sepsis with negative cultures should be evaluated for propylene glycol toxicity if they have been exposed to high dose lorazepam or other medications containing this solvent (PMID: 17555487 ). Propylene glycol is also widely used as a solvent for the carrier liquid for e-cigarettes. Propylene glycol occurs naturally, most likely as a catabolic product of sugars by gut microbiota. It has also been detected in the volatile fraction of Cannabis sativa samples (PMID: 26657499 ). |
|---|
| Structure | |
|---|
| Synonyms | | Value | Source |
|---|
| (R)-1,2-Propanediol | ChEBI | | R-1,2-PROPANEDIOL | ChEBI | | (R)-Propylene glycol | ChEBI, KEGG | | (-)-1,2-Propanediol | HMDB | | (-)-Propylene glycol | HMDB | | (2R)-1,2-Propanediol | HMDB | | (2R)-Propane-1,2-diol | HMDB | | (R)-(-)-1,2-Propanediol | HMDB | | (R)-2-Hydroxy-1-propanol | HMDB | | 1,2-(RS)-Propanediol | HMDB | | 1,2-Dihydroxypropane | HMDB | | 1,2-Propanediol | HMDB | | 1,2-Propylene glycol | HMDB | | 1-Deoxy-sn-glycerol | HMDB | | 2,3-Propanediol | HMDB | | 2-Hydroxypropanol | HMDB | | D-(-)-Propanediol | HMDB | | Isopropylene glycol | HMDB | | Methylethyl glycol | HMDB | | Methylethylene glycol | HMDB | | Monopropylene glycol | HMDB | | Propylene glycol | HMDB | | R-(-)-1,2-Propanediol | HMDB | | R-(-)-Propylene glycol | HMDB | | alpha-Propylene glycol | HMDB | | α-Propylene glycol | HMDB |
|
|---|
| Chemical Formula | C3H8O2 |
|---|
| Average Molecular Weight | 76.09 |
|---|
| Monoisotopic Molecular Weight | 76.0524 |
|---|
| IUPAC Name | (2R)-propane-1,2-diol |
|---|
| Traditional Name | R-1,2-propanediol |
|---|
| CAS Registry Number | 4254-14-2 |
|---|
| SMILES | C[C@@H](O)CO |
|---|
| InChI Identifier | InChI=1S/C3H8O2/c1-3(5)2-4/h3-5H,2H2,1H3/t3-/m1/s1 |
|---|
| InChI Key | DNIAPMSPPWPWGF-GSVOUGTGSA-N |
|---|
| Chemical Taxonomy |
|---|
| Description | Belongs to the class of organic compounds known as 1,2-diols. These are polyols containing an alcohol group at two adjacent positions. |
|---|
| Kingdom | Organic compounds |
|---|
| Super Class | Organic oxygen compounds |
|---|
| Class | Organooxygen compounds |
|---|
| Sub Class | Alcohols and polyols |
|---|
| Direct Parent | 1,2-diols |
|---|
| Alternative Parents | |
|---|
| Substituents | - Secondary alcohol
- 1,2-diol
- Hydrocarbon derivative
- Primary alcohol
- Aliphatic acyclic compound
|
|---|
| Molecular Framework | Aliphatic acyclic compounds |
|---|
| External Descriptors | |
|---|
| Ontology |
|---|
|
| Physiological effect | Health effect: |
|---|
| Disposition | Route of exposure: Source: Biological location: |
|---|
| Role | Indirect biological role: Industrial application: |
|---|
| Physical Properties |
|---|
| State | Solid |
|---|
| Experimental Properties | | Property | Value | Reference |
|---|
| Melting Point | -60 °C | Not Available | | Boiling Point | 188.2 °C | Wikipedia | | Water Solubility | 1000 mg/mL at 20 °C | Not Available | | logP | -0.92 | HANSCH,C ET AL. (1995) |
|
|---|
| Predicted Properties | [] |
|---|
| Spectra |
|---|
| EI-MS/GC-MS | | Type | Description | Splash Key | View |
|---|
| EI-MS | Mass Spectrum (Electron Ionization) | splash10-0002-9000000000-cbc512db1a4c5510015e | 2020-03-26 | View Spectrum | | GC-MS | Propylene glycol, non-derivatized, GC-MS Spectrum | splash10-014j-0900000000-d90655d5a614c4ddf998 | Spectrum | | GC-MS | Propylene glycol, 2 TMS, GC-MS Spectrum | splash10-014i-1900000000-ab4a1ff05d1275711170 | Spectrum | | GC-MS | Propylene glycol, non-derivatized, GC-MS Spectrum | splash10-0002-9000000000-a18bede40461dace657b | Spectrum | | GC-MS | Propylene glycol, non-derivatized, GC-MS Spectrum | splash10-0002-9000000000-6d2345a66e2cf8d2d379 | Spectrum | | GC-MS | Propylene glycol, non-derivatized, GC-MS Spectrum | splash10-014j-0900000000-d90655d5a614c4ddf998 | Spectrum | | GC-MS | Propylene glycol, non-derivatized, GC-MS Spectrum | splash10-014i-1900000000-ab4a1ff05d1275711170 | Spectrum | | Predicted GC-MS | Propylene glycol, non-derivatized, Predicted GC-MS Spectrum - 70eV, Positive | Not Available | Spectrum |
|
|---|
| MS/MS | | Type | Description | Splash Key | View |
|---|
| MS/MS | LC-MS/MS Spectrum - Quattro_QQQ 10V, Positive (Annotated) | splash10-0a7i-9000000000-6c96c59f8ba2e0e5353e | 2020-03-26 | View Spectrum | | MS/MS | LC-MS/MS Spectrum - Quattro_QQQ 25V, Positive (Annotated) | splash10-005d-9000000000-ae07e4049d24341335ec | 2020-03-26 | View Spectrum | | MS/MS | LC-MS/MS Spectrum - Quattro_QQQ 40V, Positive (Annotated) | splash10-00aj-9000000000-f455f8f0f79f9970711a | 2020-03-26 | View Spectrum | | MS/MS | LC-MS/MS Spectrum - EI-B (HITACHI RMU-7M) , Positive | splash10-0002-9000000000-c5bf57086deb14e12495 | 2020-03-26 | View Spectrum | | MS/MS | LC-MS/MS Spectrum - EI-B (HITACHI M-80B) , Positive | splash10-0002-9000000000-6d2345a66e2cf8d2d379 | 2020-03-26 | View Spectrum | | Predicted MS/MS | Predicted LC-MS/MS Spectrum - 10V, Positive | splash10-004i-9000000000-0a1a58ae50ca7bfbc92f | 2015-09-14 | View Spectrum | | Predicted MS/MS | Predicted LC-MS/MS Spectrum - 20V, Positive | splash10-0a6r-9000000000-4f58eaf424aa13185ca2 | 2015-09-14 | View Spectrum | | Predicted MS/MS | Predicted LC-MS/MS Spectrum - 40V, Positive | splash10-052f-9000000000-d0e5def95a7a7879dc71 | 2015-09-14 | View Spectrum | | Predicted MS/MS | Predicted LC-MS/MS Spectrum - 10V, Negative | splash10-004i-9000000000-5bf5d9d3c7cf604af9a3 | 2015-09-15 | View Spectrum | | Predicted MS/MS | Predicted LC-MS/MS Spectrum - 20V, Negative | splash10-056r-9000000000-ea5b27ac0cc585591f63 | 2015-09-15 | View Spectrum | | Predicted MS/MS | Predicted LC-MS/MS Spectrum - 40V, Negative | splash10-0a4i-9000000000-ecc48f7be3304c6cc1e0 | 2015-09-15 | View Spectrum | | Predicted MS/MS | Predicted LC-MS/MS Spectrum - 10V, Negative | splash10-0a6r-9000000000-ea4d9d80ebf7615e6811 | 2021-09-24 | View Spectrum | | Predicted MS/MS | Predicted LC-MS/MS Spectrum - 20V, Negative | splash10-0a4i-9000000000-a77309b9b0e17e94870d | 2021-09-24 | View Spectrum | | Predicted MS/MS | Predicted LC-MS/MS Spectrum - 40V, Negative | splash10-0006-9000000000-7bb23415e61ec73a459c | 2021-09-24 | View Spectrum | | Predicted MS/MS | Predicted LC-MS/MS Spectrum - 10V, Positive | splash10-0a4l-9000000000-5dba4634f494296e1347 | 2021-09-24 | View Spectrum | | Predicted MS/MS | Predicted LC-MS/MS Spectrum - 20V, Positive | splash10-0a4l-9000000000-2174cc6a153316ad8c52 | 2021-09-24 | View Spectrum | | Predicted MS/MS | Predicted LC-MS/MS Spectrum - 40V, Positive | splash10-0006-9000000000-a1a685f935041163641b | 2021-09-24 | View Spectrum |
|
|---|
| NMR | | Type | Description | | View |
|---|
| 1D NMR | 1H NMR Spectrum (1D, 500 MHz, H2O, experimental) | | Spectrum | | 1D NMR | 1H NMR Spectrum (1D, 90 MHz, CDCl3, experimental) | | Spectrum | | 1D NMR | 13C NMR Spectrum (1D, 15.09 MHz, CDCl3, experimental) | | Spectrum | | 1D NMR | 1H NMR Spectrum (1D, D2O, experimental) | | Spectrum | | 1D NMR | 13C NMR Spectrum (1D, D2O, experimental) | | Spectrum | | 1D NMR | 1H NMR Spectrum (1D, 100 MHz, D2O, predicted) | | Spectrum | | 1D NMR | 13C NMR Spectrum (1D, 100 MHz, D2O, predicted) | | Spectrum | | 1D NMR | 1H NMR Spectrum (1D, 200 MHz, D2O, predicted) | | Spectrum | | 1D NMR | 13C NMR Spectrum (1D, 200 MHz, D2O, predicted) | | Spectrum | | 1D NMR | 1H NMR Spectrum (1D, 300 MHz, D2O, predicted) | | Spectrum | | 1D NMR | 13C NMR Spectrum (1D, 300 MHz, D2O, predicted) | | Spectrum | | 1D NMR | 1H NMR Spectrum (1D, 400 MHz, D2O, predicted) | | Spectrum | | 1D NMR | 13C NMR Spectrum (1D, 400 MHz, D2O, predicted) | | Spectrum | | 1D NMR | 1H NMR Spectrum (1D, 500 MHz, D2O, predicted) | | Spectrum | | 1D NMR | 13C NMR Spectrum (1D, 500 MHz, D2O, predicted) | | Spectrum | | 1D NMR | 1H NMR Spectrum (1D, 600 MHz, D2O, predicted) | | Spectrum | | 1D NMR | 13C NMR Spectrum (1D, 600 MHz, D2O, predicted) | | Spectrum | | 1D NMR | 1H NMR Spectrum (1D, 700 MHz, D2O, predicted) | | Spectrum | | 1D NMR | 13C NMR Spectrum (1D, 700 MHz, D2O, predicted) | | Spectrum | | 1D NMR | 1H NMR Spectrum (1D, 800 MHz, D2O, predicted) | | Spectrum | | 1D NMR | 13C NMR Spectrum (1D, 800 MHz, D2O, predicted) | | Spectrum | | 1D NMR | 1H NMR Spectrum (1D, 900 MHz, D2O, predicted) | | Spectrum | | 1D NMR | 13C NMR Spectrum (1D, 900 MHz, D2O, predicted) | | Spectrum | | 1D NMR | 1H NMR Spectrum (1D, 1000 MHz, D2O, predicted) | | Spectrum | | 1D NMR | 13C NMR Spectrum (1D, 1000 MHz, D2O, predicted) | | Spectrum | | 2D NMR | [1H, 13C]-HSQC NMR Spectrum (2D, 500 MHz, H2O, experimental) | | Spectrum |
|
|---|
| Pathways |
|---|
| Pathways | | Name | SMPDB/Pathwhiz | KEGG | | Pyruvate Metabolism |    | 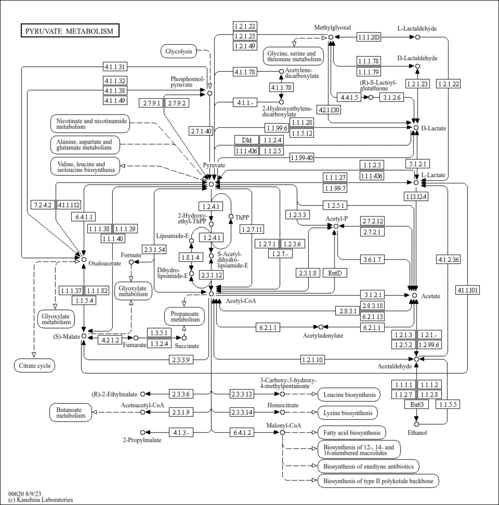 | | Leigh Syndrome |   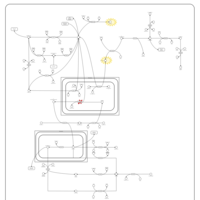 | Not Available | | Pyruvate Decarboxylase E1 Component Deficiency (PDHE1 Deficiency) | 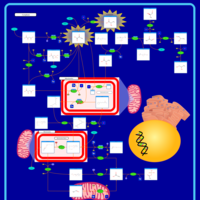 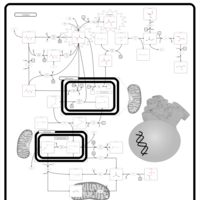 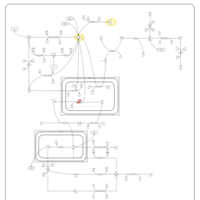 | Not Available | | Pyruvate Dehydrogenase Complex Deficiency | 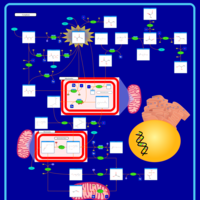 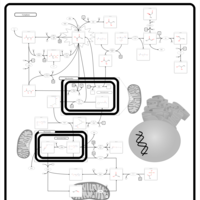 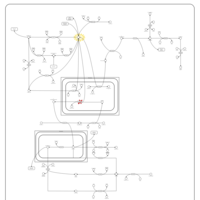 | Not Available | | Primary hyperoxaluria II, PH2 | 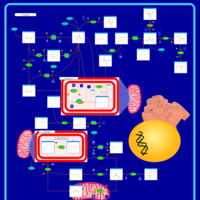   | Not Available |
|
|---|
| Protein Targets |
|---|
| Enzymes | |
|---|
| Transporters | Not Available |
|---|
| Metal Bindings | |
|---|
| Receptors | Not Available |
|---|
| Transcriptional Factors | Not Available |
|---|
| Concentrations Data |
|---|
| Not Available |
|---|
| External Links |
|---|
| HMDB ID | HMDB0001881 |
|---|
| DrugBank ID | DB02159 |
|---|
| Phenol Explorer Compound ID | Not Available |
|---|
| FoodDB ID | FDB008274 |
|---|
| KNApSAcK ID | C00007410 |
|---|
| Chemspider ID | 228188 |
|---|
| KEGG Compound ID | C02912 |
|---|
| BioCyc ID | Not Available |
|---|
| BiGG ID | Not Available |
|---|
| Wikipedia Link | Propylene_glycol |
|---|
| METLIN ID | Not Available |
|---|
| PubChem Compound | 259994 |
|---|
| PDB ID | Not Available |
|---|
| ChEBI ID | 28972 |
|---|
| References |
|---|
| General References | - Zar T, Graeber C, Perazella MA: Recognition, treatment, and prevention of propylene glycol toxicity. Semin Dial. 2007 May-Jun;20(3):217-9. doi: 10.1111/j.1525-139X.2007.00280.x. [PubMed:17555487 ]
- Rice S, Koziel JA: Characterizing the Smell of Marijuana by Odor Impact of Volatile Compounds: An Application of Simultaneous Chemical and Sensory Analysis. PLoS One. 2015 Dec 10;10(12):e0144160. doi: 10.1371/journal.pone.0144160. eCollection 2015. [PubMed:26657499 ]
|
|---|
