| Record Information |
|---|
| Version | 1.0 |
|---|
| Created at | 2020-03-19 00:28:39 UTC |
|---|
| Updated at | 2020-12-07 19:07:19 UTC |
|---|
| CannabisDB ID | CDB000280 |
|---|
| Secondary Accession Numbers | Not Available |
|---|
| Cannabis Compound Identification |
|---|
| Common Name | Malonic acid |
|---|
| Description | Malonic acid, also known as malonate or H2MALO, belongs to the class of organic compounds known as dicarboxylic acids and derivatives. These are organic compounds containing two carboxylic acid groups. Malonic acid is a very hydrophobic molecule, practically insoluble in water. Malonic acid exists in all living species, ranging from bacteria to humans. In humans, malonic acid is involved in fatty acid biosynthesis. Malonic acid has also been detected, but not quantified, in several different foods, such as red beetroots, corns, scarlet beans, common beets, and cow milks. Malonic acid is also one of several organic acids that are known to occur in cannabis plants (PMID: 6991645 ). Industrially, it is used in a number of manufacturing processes as a high value specialty chemical, including the electronics industry, flavors and fragrances industry, specialty solvents, polymer crosslinking, and pharmaceutical industry. |
|---|
| Structure | |
|---|
| Synonyms | | Value | Source |
|---|
| H2Malo | ChEBI | | HOOC-CH2-COOH | ChEBI | | Propanedioic acid | ChEBI | | Propanedioate | Generator | | Malonate | Generator | | alpha,Omega-dicarboxylic acid | HMDB | | Carboxyacetic acid | HMDB | | Dicarboxylate | HMDB | | Dicarboxylic acid | HMDB | | Dicarboxymethane | HMDB | | Kyselina malonova | HMDB | | Malonate dicarboxylic acid | HMDB | | Metahnedicarboxylic acid | HMDB | | Methanedicarbonic acid | HMDB | | Methanedicarboxylic acid | HMDB | | Propanedioic acid dithallium salt | HMDB | | Propanediolic acid | HMDB | | Thallium malonate | HMDB | | Malonic acid, 2-(14)C-labeled | HMDB | | Malonic acid, monocalcium salt | HMDB | | Malonic acid, 1,3-(14)C2-labeled | HMDB | | Malonic acid, diammonium salt | HMDB | | Malonic acid, disodium salt | HMDB | | Malonic acid, dithallium salt | HMDB | | Malonic acid, dipotassium salt | HMDB | | Malonic acid, disodium salt, 1-(14)C-labeled | HMDB | | Malonic acid, monosodium salt | HMDB | | Malonic acid, potassium salt | HMDB | | Malonic acid, sodium salt | HMDB | | Thallous malonate | HMDB | | Dithallium malonate | HMDB | | Monosodium malonate | HMDB | | Malonic acid | Generator |
|
|---|
| Chemical Formula | C3H4O4 |
|---|
| Average Molecular Weight | 104.06 |
|---|
| Monoisotopic Molecular Weight | 104.011 |
|---|
| IUPAC Name | propanedioic acid |
|---|
| Traditional Name | malonic acid |
|---|
| CAS Registry Number | 141-82-2 |
|---|
| SMILES | OC(=O)CC(O)=O |
|---|
| InChI Identifier | InChI=1S/C3H4O4/c4-2(5)1-3(6)7/h1H2,(H,4,5)(H,6,7) |
|---|
| InChI Key | OFOBLEOULBTSOW-UHFFFAOYSA-N |
|---|
| Chemical Taxonomy |
|---|
| Description | Belongs to the class of organic compounds known as dicarboxylic acids and derivatives. These are organic compounds containing exactly two carboxylic acid groups. |
|---|
| Kingdom | Organic compounds |
|---|
| Super Class | Organic acids and derivatives |
|---|
| Class | Carboxylic acids and derivatives |
|---|
| Sub Class | Dicarboxylic acids and derivatives |
|---|
| Direct Parent | Dicarboxylic acids and derivatives |
|---|
| Alternative Parents | |
|---|
| Substituents | - 1,3-dicarbonyl compound
- Dicarboxylic acid or derivatives
- Carboxylic acid
- Organic oxygen compound
- Organic oxide
- Hydrocarbon derivative
- Organooxygen compound
- Carbonyl group
- Aliphatic acyclic compound
|
|---|
| Molecular Framework | Aliphatic acyclic compounds |
|---|
| External Descriptors | |
|---|
| Ontology |
|---|
|
| Disposition | Route of exposure: Source: Biological location: |
|---|
| Role | Industrial application: Biological role: |
|---|
| Physical Properties |
|---|
| State | Solid |
|---|
| Experimental Properties | | Property | Value | Reference |
|---|
| Melting Point | 135 °C | Not Available | | Boiling Point | Not Available | Not Available | | Water Solubility | 763 mg/mL | Not Available | | logP | -0.81 | HANSCH,C ET AL. (1995) |
|
|---|
| Predicted Properties | [] |
|---|
| Spectra |
|---|
| EI-MS/GC-MS | | Type | Description | Splash Key | View |
|---|
| EI-MS | Mass Spectrum (Electron Ionization) | splash10-01ox-9000000000-dd4efef191376724d0f1 | 2015-03-01 | View Spectrum | | GC-MS | Malonic acid, 2 TMS, GC-MS Spectrum | splash10-0002-1900000000-a1463432c138c328557d | Spectrum | | GC-MS | Malonic acid, non-derivatized, GC-MS Spectrum | splash10-0002-0900000000-5e58241137fee0ccc64a | Spectrum | | GC-MS | Malonic acid, 2 TMS, GC-MS Spectrum | splash10-006t-9700000000-54976b3ce8f36ce0676d | Spectrum | | GC-MS | Malonic acid, 2 TMS, GC-MS Spectrum | splash10-001i-4930000000-e7dbed4919870db8dabf | Spectrum | | GC-MS | Malonic acid, 3 TMS, GC-MS Spectrum | splash10-05gl-1943000000-456e387fdf365bf0f8ee | Spectrum | | GC-MS | Malonic acid, non-derivatized, GC-MS Spectrum | splash10-0006-9000000000-6c56a402111059603ba4 | Spectrum | | GC-MS | Malonic acid, non-derivatized, GC-MS Spectrum | splash10-0006-9000000000-f239df8fdd12e9a74445 | Spectrum | | GC-MS | Malonic acid, non-derivatized, GC-MS Spectrum | splash10-0006-9000000000-6865e83f0df5c9831619 | Spectrum | | GC-MS | Malonic acid, non-derivatized, GC-MS Spectrum | splash10-0002-0900000000-a768f0aae86e17a3a6ba | Spectrum | | GC-MS | Malonic acid, non-derivatized, GC-MS Spectrum | splash10-0002-1900000000-a1463432c138c328557d | Spectrum | | GC-MS | Malonic acid, non-derivatized, GC-MS Spectrum | splash10-0002-0900000000-5e58241137fee0ccc64a | Spectrum | | GC-MS | Malonic acid, non-derivatized, GC-MS Spectrum | splash10-006t-9700000000-54976b3ce8f36ce0676d | Spectrum | | GC-MS | Malonic acid, non-derivatized, GC-MS Spectrum | splash10-05gl-1943000000-456e387fdf365bf0f8ee | Spectrum | | GC-MS | Malonic acid, non-derivatized, GC-MS Spectrum | splash10-001i-4930000000-e7dbed4919870db8dabf | Spectrum | | GC-MS | Malonic acid, non-derivatized, GC-MS Spectrum | splash10-0002-0900000000-a45cfb1e2683f8ad8c5c | Spectrum | | GC-MS | Malonic acid, non-derivatized, GC-MS Spectrum | splash10-0002-0911000000-8b0d7393c5dc4462a756 | Spectrum | | Predicted GC-MS | Malonic acid, non-derivatized, Predicted GC-MS Spectrum - 70eV, Positive | splash10-0zfu-9300000000-cbc239877c485e6dff65 | Spectrum | | Predicted GC-MS | Malonic acid, 2 TMS, Predicted GC-MS Spectrum - 70eV, Positive | splash10-00di-9410000000-c30e36ba8e95ef1c83be | Spectrum | | Predicted GC-MS | Malonic acid, non-derivatized, Predicted GC-MS Spectrum - 70eV, Positive | Not Available | Spectrum | | Predicted GC-MS | Malonic acid, TMS_1_1, Predicted GC-MS Spectrum - 70eV, Positive | Not Available | Spectrum | | Predicted GC-MS | Malonic acid, TBDMS_1_1, Predicted GC-MS Spectrum - 70eV, Positive | Not Available | Spectrum | | Predicted GC-MS | Malonic acid, TBDMS_2_1, Predicted GC-MS Spectrum - 70eV, Positive | Not Available | Spectrum |
|
|---|
| MS/MS | | Type | Description | Splash Key | View |
|---|
| MS/MS | LC-MS/MS Spectrum - Quattro_QQQ 10V, Negative (Annotated) | splash10-0a4i-9100000000-c7a1704f8a38ca2d245a | 2012-07-24 | View Spectrum | | MS/MS | LC-MS/MS Spectrum - Quattro_QQQ 25V, Negative (Annotated) | splash10-0a4l-9100000000-9fa84f43f2e19b56035c | 2012-07-24 | View Spectrum | | MS/MS | LC-MS/MS Spectrum - Quattro_QQQ 40V, Negative (Annotated) | splash10-052f-9100000000-8a21753b343cf5dabdcb | 2012-07-24 | View Spectrum | | MS/MS | LC-MS/MS Spectrum - EI-B (Unknown) , Positive | splash10-0006-9000000000-6c56a402111059603ba4 | 2012-08-31 | View Spectrum | | MS/MS | LC-MS/MS Spectrum - EI-B (HITACHI RMU-7M) , Positive | splash10-0006-9000000000-f239df8fdd12e9a74445 | 2012-08-31 | View Spectrum | | MS/MS | LC-MS/MS Spectrum - LC-ESI-QQ (API3000, Applied Biosystems) 10V, Negative | splash10-0udi-3900000000-e0a4c7e792cfd0e60cc3 | 2012-08-31 | View Spectrum | | MS/MS | LC-MS/MS Spectrum - LC-ESI-QQ (API3000, Applied Biosystems) 20V, Negative | splash10-0a4i-9000000000-4c03aa889e6a98ab532f | 2012-08-31 | View Spectrum | | MS/MS | LC-MS/MS Spectrum - LC-ESI-QQ (API3000, Applied Biosystems) 30V, Negative | splash10-0a4l-9000000000-b3d14c986f292bdcf477 | 2012-08-31 | View Spectrum | | MS/MS | LC-MS/MS Spectrum - LC-ESI-QQ (API3000, Applied Biosystems) 40V, Negative | splash10-0006-9000000000-8e1041322f9acda4088a | 2012-08-31 | View Spectrum | | MS/MS | LC-MS/MS Spectrum - LC-ESI-QQ (API3000, Applied Biosystems) 50V, Negative | splash10-0006-9000000000-af4dccf21d68110099a1 | 2012-08-31 | View Spectrum | | MS/MS | LC-MS/MS Spectrum - LC-ESI-QQ , negative | splash10-0udi-3900000000-e0a4c7e792cfd0e60cc3 | 2017-09-14 | View Spectrum | | MS/MS | LC-MS/MS Spectrum - LC-ESI-QQ , negative | splash10-0a4i-9000000000-4c03aa889e6a98ab532f | 2017-09-14 | View Spectrum | | MS/MS | LC-MS/MS Spectrum - LC-ESI-QQ , negative | splash10-0a4l-9000000000-b3d14c986f292bdcf477 | 2017-09-14 | View Spectrum | | MS/MS | LC-MS/MS Spectrum - LC-ESI-QQ , negative | splash10-0006-9000000000-8e1041322f9acda4088a | 2017-09-14 | View Spectrum | | MS/MS | LC-MS/MS Spectrum - LC-ESI-QQ , negative | splash10-0006-9000000000-af4dccf21d68110099a1 | 2017-09-14 | View Spectrum | | MS/MS | LC-MS/MS Spectrum - , negative | splash10-0006-3900000000-2126168f67ddb43e544d | 2017-09-14 | View Spectrum | | MS/MS | LC-MS/MS Spectrum - , positive | splash10-000i-9000000000-e9d5906655e37a471b89 | 2017-09-14 | View Spectrum | | MS/MS | LC-MS/MS Spectrum - 20V, Negative | splash10-0a4i-9000000000-c742107695804ea678f1 | 2021-09-20 | View Spectrum | | MS/MS | LC-MS/MS Spectrum - 40V, Negative | splash10-0006-9000000000-cb15cb8d630a4fbd0750 | 2021-09-20 | View Spectrum | | Predicted MS/MS | Predicted LC-MS/MS Spectrum - 10V, Positive | splash10-0a4r-7900000000-795ca313c7daaf448ede | 2016-09-12 | View Spectrum | | Predicted MS/MS | Predicted LC-MS/MS Spectrum - 20V, Positive | splash10-0bti-9400000000-a6390568ef61fe1a284e | 2016-09-12 | View Spectrum | | Predicted MS/MS | Predicted LC-MS/MS Spectrum - 40V, Positive | splash10-01ox-9100000000-cc17364c6d52c2c208f3 | 2016-09-12 | View Spectrum | | Predicted MS/MS | Predicted LC-MS/MS Spectrum - 10V, Negative | splash10-0zfr-6900000000-b1169abf90c9693f8e30 | 2016-09-12 | View Spectrum | | Predicted MS/MS | Predicted LC-MS/MS Spectrum - 20V, Negative | splash10-0a4i-9300000000-cec6a47ea3eb4a95a957 | 2016-09-12 | View Spectrum | | Predicted MS/MS | Predicted LC-MS/MS Spectrum - 40V, Negative | splash10-0a4i-9000000000-cffa3dea7855764ea854 | 2016-09-12 | View Spectrum |
|
|---|
| NMR | | Type | Description | | View |
|---|
| 1D NMR | 13C NMR Spectrum (1D, 125 MHz, H2O, experimental) | | Spectrum | | 1D NMR | 1H NMR Spectrum (1D, 500 MHz, H2O, experimental) | | Spectrum | | 1D NMR | 1H NMR Spectrum (1D, 90 MHz, DMSO-d6, experimental) | | Spectrum | | 1D NMR | 13C NMR Spectrum (1D, 100 MHz, D2O, predicted) | | Spectrum | | 1D NMR | 1H NMR Spectrum (1D, 100 MHz, D2O, predicted) | | Spectrum | | 1D NMR | 13C NMR Spectrum (1D, 1000 MHz, D2O, predicted) | | Spectrum | | 1D NMR | 1H NMR Spectrum (1D, 1000 MHz, D2O, predicted) | | Spectrum | | 1D NMR | 13C NMR Spectrum (1D, 200 MHz, D2O, predicted) | | Spectrum | | 1D NMR | 1H NMR Spectrum (1D, 200 MHz, D2O, predicted) | | Spectrum | | 1D NMR | 13C NMR Spectrum (1D, 300 MHz, D2O, predicted) | | Spectrum | | 1D NMR | 1H NMR Spectrum (1D, 300 MHz, D2O, predicted) | | Spectrum | | 1D NMR | 13C NMR Spectrum (1D, 400 MHz, D2O, predicted) | | Spectrum | | 1D NMR | 1H NMR Spectrum (1D, 400 MHz, D2O, predicted) | | Spectrum | | 1D NMR | 13C NMR Spectrum (1D, 500 MHz, D2O, predicted) | | Spectrum | | 1D NMR | 1H NMR Spectrum (1D, 500 MHz, D2O, predicted) | | Spectrum | | 1D NMR | 13C NMR Spectrum (1D, 600 MHz, D2O, predicted) | | Spectrum | | 1D NMR | 1H NMR Spectrum (1D, 600 MHz, D2O, predicted) | | Spectrum | | 1D NMR | 13C NMR Spectrum (1D, 700 MHz, D2O, predicted) | | Spectrum | | 1D NMR | 1H NMR Spectrum (1D, 700 MHz, D2O, predicted) | | Spectrum | | 1D NMR | 13C NMR Spectrum (1D, 800 MHz, D2O, predicted) | | Spectrum | | 1D NMR | 1H NMR Spectrum (1D, 800 MHz, D2O, predicted) | | Spectrum | | 1D NMR | 13C NMR Spectrum (1D, 900 MHz, D2O, predicted) | | Spectrum | | 1D NMR | 1H NMR Spectrum (1D, 900 MHz, D2O, predicted) | | Spectrum | | 1D NMR | 13C NMR Spectrum (1D, 400 MHz, H2O, experimental) | | Spectrum | | 1D NMR | 1H NMR Spectrum (1D, 500 MHz, H2O, experimental) | | Spectrum | | 2D NMR | [1H, 13C]-HSQC NMR Spectrum (2D, 600 MHz, H2O, experimental) | | Spectrum |
|
|---|
| Pathways |
|---|
| Pathways | | Name | SMPDB/Pathwhiz | KEGG | | Aspartate Metabolism | 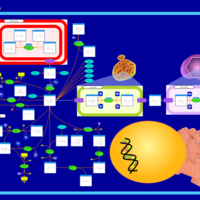 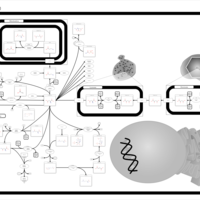  |  | | Fatty Acid Biosynthesis | 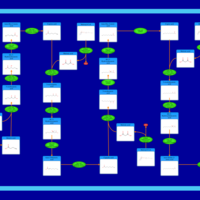 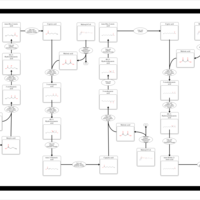 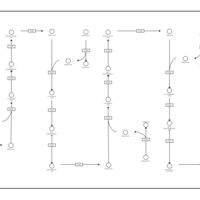 | Not Available |
|
|---|
| Protein Targets |
|---|
| Enzymes | |
| 4-aminobutyrate aminotransferase, mitochondrial | ABAT | 16p13.2 | P80404 | details | | UDP-glucuronosyltransferase 1-1 | UGT1A1 | 2q37 | P22309 | details | | Proto-oncogene tyrosine-protein kinase Src | SRC | 20q12-q13 | P12931 | details |
|
|---|
| Transporters | Not Available |
|---|
| Metal Bindings | Not Available |
|---|
| Receptors | Not Available |
|---|
| Transcriptional Factors | Not Available |
|---|
| Concentrations Data |
|---|
| Not Available |
|---|
| External Links |
|---|
| HMDB ID | HMDB0000691 |
|---|
| DrugBank ID | DB02175 |
|---|
| Phenol Explorer Compound ID | Not Available |
|---|
| FoodDB ID | FDB008117 |
|---|
| KNApSAcK ID | C00001193 |
|---|
| Chemspider ID | 844 |
|---|
| KEGG Compound ID | C04025 |
|---|
| BioCyc ID | MALONATE |
|---|
| BiGG ID | Not Available |
|---|
| Wikipedia Link | Malonic_acid |
|---|
| METLIN ID | 3237 |
|---|
| PubChem Compound | 867 |
|---|
| PDB ID | Not Available |
|---|
| ChEBI ID | 30794 |
|---|
| References |
|---|
| General References | - Turner CE, Elsohly MA, Boeren EG: Constituents of Cannabis sativa L. XVII. A review of the natural constituents. J Nat Prod. 1980 Mar-Apr;43(2):169-234. doi: 10.1021/np50008a001. [PubMed:6991645 ]
|
|---|
