| Record Information |
|---|
| Version | 1.0 |
|---|
| Created at | 2020-04-17 19:10:55 UTC |
|---|
| Updated at | 2020-11-18 16:39:23 UTC |
|---|
| CannabisDB ID | CDB005116 |
|---|
| Secondary Accession Numbers | Not Available |
|---|
| Cannabis Compound Identification |
|---|
| Common Name | 5,10-Methylene-THF |
|---|
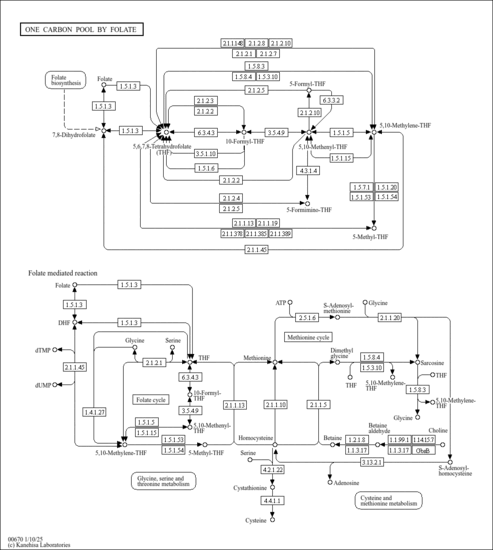
| Description | 5,10-Methylene-THF, also known as CH2H4folate, belongs to the class of organic compounds known as tetrahydrofolic acids. These are heterocyclic compounds based on the 5,6,7,8-tetrahydropteroic acid skeleton conjugated with at least one L-glutamic acid unit. It also acts as a coenzyme in the synthesis of serine from glycine via the enzyme serine hydroxymethyl transferase. More specifically it is the C1-donor in the reactions catalyzed by thymidylate synthase and thymidylate synthase (FAD). 5,10-Methylene-THF is a strong basic compound (based on its pKa). 5,10-Methylene-THF is a substrate for Methylenetetrahydrofolate reductase. 5,10-Methylene-THF exists in all eukaryotes, ranging from yeast to humans. Within humans, 5,10-methylene-THF participates in a number of enzymatic reactions. In particular, dihydrolipoate and 5,10-methylene-THF can be biosynthesized from tetrahydrofolic acid and 8-[(aminomethyl)sulfanyl]-6-sulfanyloctanoic acid; which is catalyzed by the enzyme aminomethyltransferase, mitochondrial. In addition, 5,10-methylene-THF and glycine can be converted into tetrahydrofolic acid and L-serine; which is mediated by the enzyme serine hydroxymethyltransferase, mitochondrial. This enzyme converts 5,10-Methylene-THF to 5-methyltetrahydrofolate. 5,10-Methylene-THF is an intermediate in the metabolism of Methane and the metabolism of Nitrogen. In humans, 5,10-methylene-THF is involved in the metabolic disorder called the dimethylglycine dehydrogenase deficiency pathway. This reaction is required for the multistep process that converts the amino acid homocysteine to methionine. The body uses methionine to make proteins and other important compounds. 5,10-CH2-THF can also be used as a coenzyme in the biosynthesis of thymidine. 5,10-Methylene-THF is an intermediate in glycine, serine and threonine metabolism and one carbon metabolism. 5,10-Methylene-THF is expected to be in Cannabis as all living plants are known to produce and metabolize it. |
|---|
| Structure | |
|---|
| Synonyms | | Value | Source |
|---|
| (6R)-5,10-Methylenetetrahydrofolate | Kegg | | (6R)-5,10-Methylenetetrahydrofolic acid | Generator | | 5,10-Methenyltetrahydropteroylglutamate | HMDB | | 5,10-Methylene-6-hydrofolate | HMDB | | 5,10-Methylene-6-hydrofolic acid | HMDB | | 5,10-Methylenetetrahydrofolate | HMDB | | 5,10-Methylenetetrahydrofolic acid | HMDB | | N5>,N10-methylenetetrahydrofolate | HMDB | | 5,10-Methylene-5,6,7,8-tetrahydrofolate | HMDB | | Tetrahydromethylenefolate | HMDB | | 5,10-Methylenetetrahydrofolate, (D-glu)-isomer | HMDB | | CH2H4Folate | HMDB | | 5,10-Methylenetetrahydrofolate monohydrochloride, (L-glu)-isomer | HMDB | | 5,10-Methylenetetrahydrofolate, (L-glu)-(S)-isomer | HMDB |
|
|---|
| Chemical Formula | C20H23N7O6 |
|---|
| Average Molecular Weight | 457.44 |
|---|
| Monoisotopic Molecular Weight | 457.171 |
|---|
| IUPAC Name | 2-({4-[(6aR)-1-hydroxy-3-imino-3H,4H,5H,6H,6aH,7H,8H,9H-imidazo[1,5-f]pteridin-8-yl]phenyl}formamido)pentanedioic acid |
|---|
| Traditional Name | 2-({4-[(6aR)-1-hydroxy-3-imino-4H,5H,6H,6aH,7H,9H-imidazo[1,5-f]pteridin-8-yl]phenyl}formamido)pentanedioic acid |
|---|
| CAS Registry Number | 31690-11-6 |
|---|
| SMILES | [H][C@@]12CN(CN1C1=C(NC2)N=C(N)NC1=O)C1=CC=C(C=C1)C(=O)NC(CCC(O)=O)C(O)=O |
|---|
| InChI Identifier | InChI=1S/C20H23N7O6/c21-20-24-16-15(18(31)25-20)27-9-26(8-12(27)7-22-16)11-3-1-10(2-4-11)17(30)23-13(19(32)33)5-6-14(28)29/h1-4,12-13H,5-9H2,(H,23,30)(H,28,29)(H,32,33)(H4,21,22,24,25,31)/t12-,13?/m1/s1 |
|---|
| InChI Key | QYNUQALWYRSVHF-PZORYLMUSA-N |
|---|
| Chemical Taxonomy |
|---|
| Description | Belongs to the class of organic compounds known as tetrahydrofolic acids. These are heterocyclic compounds based on the 5,6,7,8-tetrahydropteroic acid skeleton conjugated with at least one L-glutamic acid unit. |
|---|
| Kingdom | Organic compounds |
|---|
| Super Class | Organoheterocyclic compounds |
|---|
| Class | Pteridines and derivatives |
|---|
| Sub Class | Pterins and derivatives |
|---|
| Direct Parent | Tetrahydrofolic acids |
|---|
| Alternative Parents | |
|---|
| Substituents | - Tetrahydrofolic acid
- Glutamic acid or derivatives
- N-acyl-alpha amino acid or derivatives
- N-acyl-alpha-amino acid
- Hippuric acid
- Hippuric acid or derivatives
- Phenylimidazolidine
- Alpha-amino acid or derivatives
- Benzamide
- Benzoic acid or derivatives
- Imidazopyrazine
- Benzoyl
- Dialkylarylamine
- Aniline or substituted anilines
- Pyrimidone
- Aminopyrimidine
- Secondary aliphatic/aromatic amine
- Benzenoid
- Pyrimidine
- Monocyclic benzene moiety
- Dicarboxylic acid or derivatives
- Heteroaromatic compound
- Vinylogous amide
- Imidazolidine
- Secondary carboxylic acid amide
- Amino acid or derivatives
- Amino acid
- Carboxamide group
- Secondary amine
- Azacycle
- Carboxylic acid
- Carboxylic acid derivative
- Primary amine
- Organic nitrogen compound
- Carbonyl group
- Hydrocarbon derivative
- Amine
- Organic oxide
- Organopnictogen compound
- Organic oxygen compound
- Organonitrogen compound
- Organooxygen compound
- Aromatic heteropolycyclic compound
|
|---|
| Molecular Framework | Aromatic heteropolycyclic compounds |
|---|
| External Descriptors | Not Available |
|---|
| Ontology |
|---|
|
| Disposition | Source: Biological location: |
|---|
| Physical Properties |
|---|
| State | Solid |
|---|
| Experimental Properties | | Property | Value | Reference |
|---|
| Melting Point | Not Available | Not Available | | Boiling Point | Not Available | Not Available | | Water Solubility | Not Available | Not Available | | logP | Not Available | Not Available |
|
|---|
| Predicted Properties | [] |
|---|
| EI-MS/GC-MS | | Type | Description | Splash Key | View |
|---|
| Predicted GC-MS | 5,10-Methylene-THF, non-derivatized, Predicted GC-MS Spectrum - 70eV, Positive | splash10-03di-1635900000-4b2ed6618451a7e931b1 | Spectrum | | Predicted GC-MS | 5,10-Methylene-THF, 2 TMS, Predicted GC-MS Spectrum - 70eV, Positive | splash10-001i-2129050000-80e96fb3203322200945 | Spectrum | | Predicted GC-MS | 5,10-Methylene-THF, non-derivatized, Predicted GC-MS Spectrum - 70eV, Positive | Not Available | Spectrum | | Predicted GC-MS | 5,10-Methylene-THF, non-derivatized, Predicted GC-MS Spectrum - 70eV, Positive | Not Available | Spectrum |
|
|---|
| MS/MS | | Type | Description | Splash Key | View |
|---|
| Predicted MS/MS | Predicted LC-MS/MS Spectrum - 10V, Positive | splash10-08fu-0003900000-ce1f75a98c0216d169e2 | 2016-09-12 | View Spectrum | | Predicted MS/MS | Predicted LC-MS/MS Spectrum - 20V, Positive | splash10-03di-0229400000-c8c29bf14ef80a73f864 | 2016-09-12 | View Spectrum | | Predicted MS/MS | Predicted LC-MS/MS Spectrum - 40V, Positive | splash10-02u3-1192000000-ab556f50884c7b4fdcbe | 2016-09-12 | View Spectrum | | Predicted MS/MS | Predicted LC-MS/MS Spectrum - 10V, Negative | splash10-0a4i-0001900000-6ba3ddeb1f652fdc6b6b | 2016-09-12 | View Spectrum | | Predicted MS/MS | Predicted LC-MS/MS Spectrum - 20V, Negative | splash10-08fr-1225900000-82b75909f87dfeb54211 | 2016-09-12 | View Spectrum | | Predicted MS/MS | Predicted LC-MS/MS Spectrum - 40V, Negative | splash10-0006-9553000000-5aca24c246430955de51 | 2016-09-12 | View Spectrum | | Predicted MS/MS | Predicted LC-MS/MS Spectrum - 10V, Positive | splash10-0bt9-0006900000-dd6e195db40930799024 | 2021-09-22 | View Spectrum | | Predicted MS/MS | Predicted LC-MS/MS Spectrum - 20V, Positive | splash10-03di-0109200000-aa6336e6c1c41c98d390 | 2021-09-22 | View Spectrum | | Predicted MS/MS | Predicted LC-MS/MS Spectrum - 40V, Positive | splash10-01q9-1695000000-8b9546bbad524392a35b | 2021-09-22 | View Spectrum | | Predicted MS/MS | Predicted LC-MS/MS Spectrum - 10V, Negative | splash10-000f-0009800000-1ef964ba403eb61f6bf0 | 2021-09-22 | View Spectrum | | Predicted MS/MS | Predicted LC-MS/MS Spectrum - 20V, Negative | splash10-01w3-1407900000-f4b4d0db119b458049fb | 2021-09-22 | View Spectrum | | Predicted MS/MS | Predicted LC-MS/MS Spectrum - 40V, Negative | splash10-0udl-5935200000-5fa8390660d5a558ac8b | 2021-09-22 | View Spectrum |
|
|---|